Zinc + Hydrogen Chloride Yields Zinc Chloride And Hydrogen
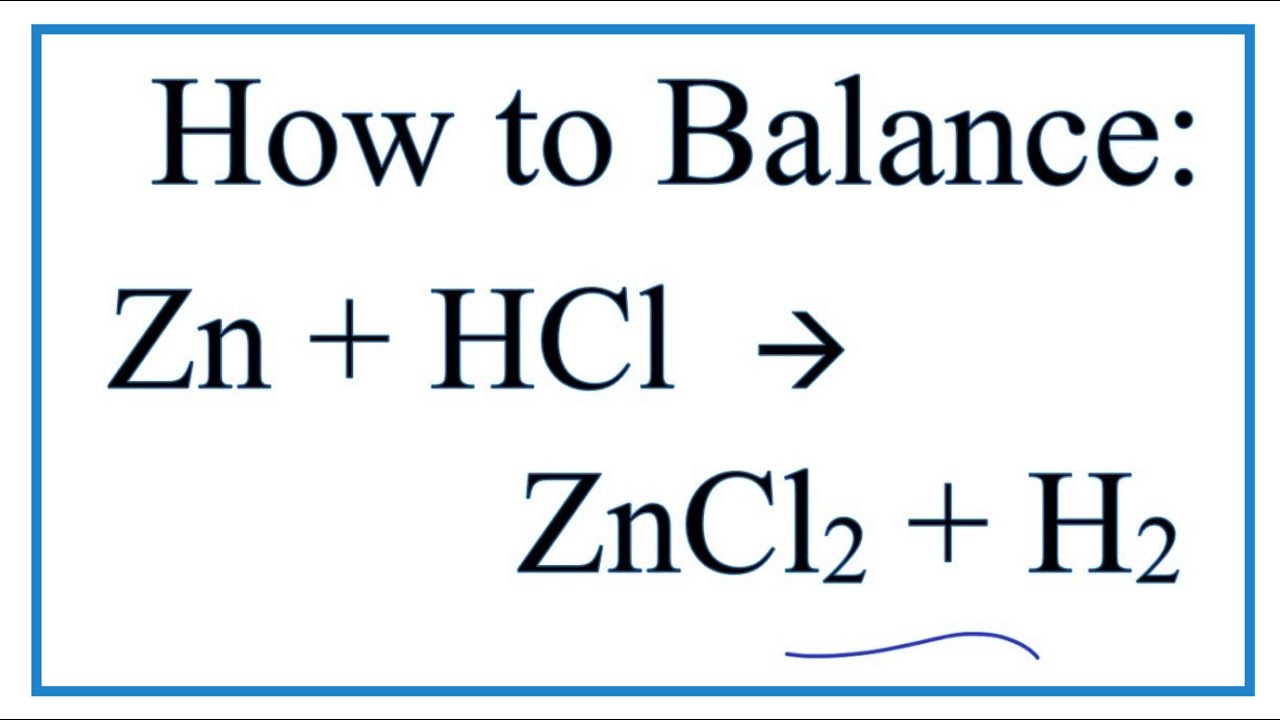
Ever found yourself staring at a bubbling beaker, mesmerized by the sheer science unfolding before your eyes? Or maybe you're that friend who’s always tinkering with something, creating a little magic in their garage? Well, today we’re diving into a reaction that’s both visually engaging and surprisingly practical: the dance between Zinc and Hydrogen Chloride. It might sound like something out of a mad scientist's lab, but understanding this chemical tango can actually shed light on a whole host of everyday phenomena and even offer some cool DIY opportunities (with the right safety precautions, of course!).
So, why is this particular chemical reaction worth talking about? For starters, it’s a fantastic demonstration of a fundamental concept in chemistry: acid-metal reactions. When you combine these two substances, you get a lively fizzing as hydrogen gas is released, and a new compound, Zinc Chloride, is formed. This seemingly simple exchange is the bedrock of many industrial processes and has surprising applications that impact our lives more than we might realize. Think about it – the ability to predict and control chemical reactions allows us to create new materials, generate energy, and develop essential products. This specific reaction is a gateway to understanding how elements can transform into entirely new substances with different properties.
But how does this translate to your everyday life? Well, Zinc Chloride itself is a pretty handy chemical. It’s a strong Lewis acid, which means it’s excellent at catalyzing reactions, particularly in the production of plastics and dyes. Ever used a dry cell battery? The electrolyte in many of them contains Zinc Chloride, helping to conduct electricity. Beyond that, it has uses in wood preservation to protect against decay and insect infestation, and even in some soldering fluxes to help clean metal surfaces for better bonding. The hydrogen gas produced, while often seen as a byproduct in this demonstration, is a clean fuel source that’s gaining traction for the future of transportation and energy.
Now, if you’re feeling inspired and want to explore this reaction further (responsibly, of course!), there are ways to enhance the experience. The most crucial tip is safety first. Hydrogen Chloride is a corrosive acid, and hydrogen gas is flammable. Always conduct experiments in a well-ventilated area, wear appropriate safety goggles and gloves, and never attempt this without proper supervision and knowledge of chemical handling. For a more visually engaging experience, consider using different forms of zinc – zinc powder will react much faster and more vigorously than a solid chunk. You can also observe the reaction in a clear container to better see the gas bubbles forming. And if you’re interested in collecting the hydrogen gas (again, with extreme caution and proper equipment), it can be a fascinating way to demonstrate its presence.
Ultimately, the reaction of Zinc with Hydrogen Chloride isn't just a textbook example; it's a glimpse into the power of chemistry to transform matter and create useful products. Whether you’re a seasoned experimenter or just curious about the world around you, understanding these fundamental reactions can open up a whole new perspective on the science that shapes our modern lives. So, next time you see something bubbling or hear about a new material, remember the simple yet profound interactions happening at the atomic level, like the satisfying fizz of zinc meeting acid!
