You Wish To Substitute Bromine In The Following Molecules

Hey there, coffee buddy! So, you've got this vibe going on, right? You're looking at some molecules, and your brain’s just doing that little happy dance, thinking, “What if… what if we tossed in some bromine?” I get it. It’s like wanting to spice up your usual Tuesday night with a little something extra. Bromine, that’s our guest star today. It’s a bit of a drama queen, isn’t it? Big, clunky, and definitely makes its presence known. So, let’s dive into this, shall we? Imagine we’re just chilling, sketching out some molecular diagrams on a napkin. No pressure, just pure, unadulterated chemical curiosity.
The thing about substituting elements, it’s like swapping out a key ingredient in your favorite recipe. You know, sometimes you swap out sugar for honey, and it’s a whole new ballgame. Other times, you swap out, say, a tiny pinch of salt for a whole tablespoon of… well, something that’s definitely not salt. And that, my friend, can go hilariously, spectacularly wrong. So, when we’re talking about bromine, we’re talking about a major ingredient swap. It’s not a subtle change; it’s a whole personality shift for the molecule. We’re going from, maybe, a quiet, well-behaved hydrogen atom to this flashy, somewhat intimidating bromine. Big shoes to fill, or perhaps, big atoms to replace!
First up, let’s imagine we’re looking at something super simple. Like, really simple. Think methane, CH4. It’s the OG of alkanes, the friendly neighborhood hydrocarbon. Four little hydrogens, all cozy around a carbon. So cute, right? Now, you’re thinking, “What if we kick one of those hydrogens out? And shove in a bromine instead?” Boom! You get bromomethane. CH3Br. It’s still methane-ish, but now it’s got this… edge. It’s like your shy friend who suddenly shows up wearing a leather jacket and dark sunglasses. Still your friend, but you know, different vibe. You can totally see it, can’t you? That little CH3 group is still there, but this bromine atom is just… there. Making a statement.
And the implications? Oh, the implications! Bromomethane, while still relatively simple, has some very different properties from methane. Methane is a gas, pretty inert, used for heating and all that jazz. Bromomethane? It’s a liquid at room temperature, and it’s got some serious oomph when it comes to reactivity. It’s used as a fumigant, which sounds… intense. So, this one little swap? It changes everything. From being the life of the party (methane, powering your stove) to being the guy who cleans up the mess (bromomethane, tackling pests). It’s a career change, basically. A radical one.
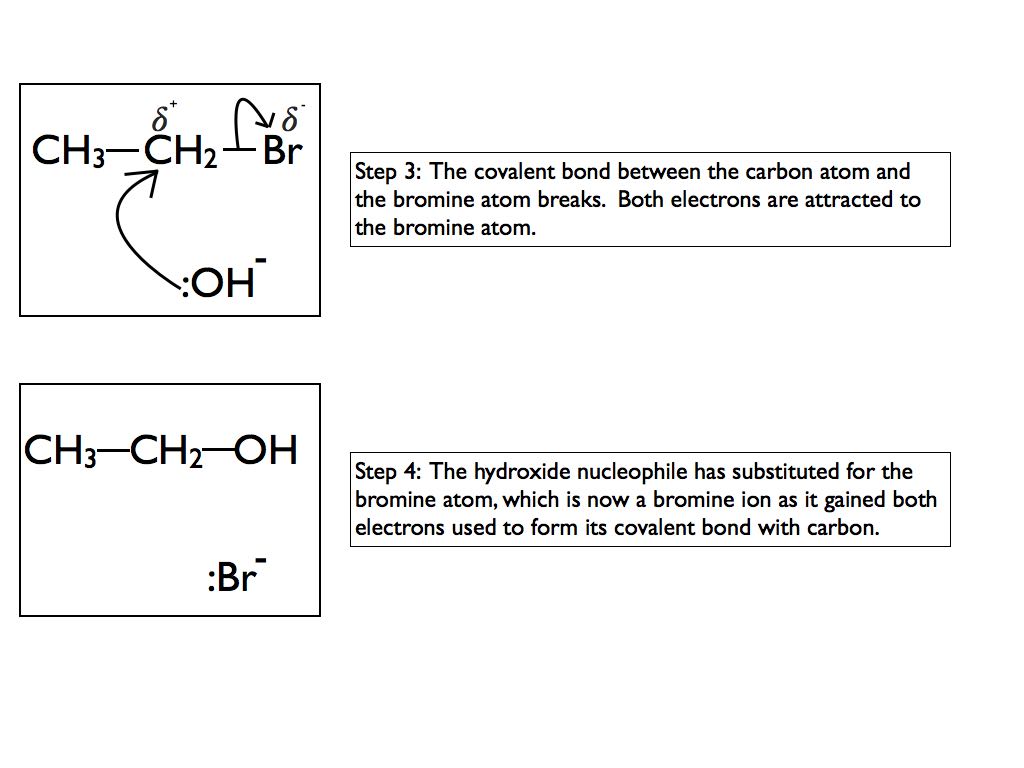
Now, let’s crank it up a notch. What about something a little more complex? Let’s say we have an alcohol. Like ethanol. C2H5OH. That’s the stuff in your fancy evening beverage. Two carbons, five hydrogens, and that all-important hydroxyl group (-OH). It’s pretty stable, right? Now, imagine we decide to replace… hmm… maybe one of the hydrogens on the ethyl part. So, not the hydrogen on the OH, that’s a whole other story. We’re talking about one of those hydrogens attached to the carbon. We swap it for bromine. So, instead of C2H5OH, we get something like C2H4BrOH. We’ve got ourselves a brominated alcohol. Fancy!
What does this brominated alcohol do? Well, it’s going to behave differently. The bromine atom is electron-withdrawing. That means it likes to pull electrons towards itself. This can make nearby bonds a bit… wobbly. It can also make the molecule more susceptible to certain types of reactions. Think of it like adding a really opinionated person to a group discussion. They might steer the conversation in a completely unexpected direction. This brominated alcohol might be more reactive, maybe prone to elimination reactions, or nucleophilic substitutions. It’s less of a gentle sipper and more of a… chemistry experiment waiting to happen.

And that hydroxyl group, the -OH? If we were to replace that hydrogen with bromine, well, that’s not really an alcohol anymore, is it? That would be like trying to make water into something else by just swapping out a hydrogen. That's more in the realm of… a hypobromous acid, maybe? HOBr. It’s a very different beast. So, context, my friend, context is key. Where you put that bromine matters. It’s not just about the ingredient; it’s about where you sprinkle it on your molecular dish.
Let’s talk about aromatic compounds. You know, the ones with the neat little rings. Benzene, C6H6. Six carbons in a hexagon, with alternating double bonds. It’s the poster child for aromaticity. Pretty stable, right? Like a well-constructed building. Now, you’re feeling adventurous. You’re thinking, “What if we replace one of those hydrogens on the benzene ring with bromine?” So, one of those hydrogens is unceremoniously evicted, and in strolls a bromine atom. You get bromobenzene. C6H5Br. One bromine, clinging to that aromatic ring.
This is a classic reaction, you know. Electrophilic aromatic substitution. And bromine is a pretty good electrophile, especially with a little help from a Lewis acid catalyst like iron(III) bromide. The bromine atom, once attached, changes the game for the benzene ring. It’s still aromatic, of course, but the bromine atom can influence where other substituents will go. It's a deactivating group, meaning it makes the ring slightly less reactive towards further electrophilic substitution. But it's also an ortho, para director. So, if you try to add something else, it's more likely to pop up next to the bromine (ortho) or opposite it (para). It's like the bromine is saying, "Okay, everyone else, you can join the party, but try to stay out of my personal space. And maybe hang out over there."

Bromobenzene itself is a pretty useful intermediate in organic synthesis. It can be used to make Grignard reagents (phenylmagnesium bromide), which are super handy for forming new carbon-carbon bonds. So, that one bromine atom, it doesn’t just sit there looking pretty; it opens up a whole world of possibilities. It’s like giving your molecule a superpower. A somewhat menacing, halogenated superpower.
What if we go wild and put more than one bromine on the benzene ring? We could have dibromobenzenes, tribromobenzenes, even hexabromobenzene! Each bromine you add makes the ring even less reactive to electrophilic substitution. It’s like plastering your building with so much lead paint, nothing new is going to stick. And the physical properties change too. These polybrominated compounds can become solids, often with high melting points. They can also become quite toxic and persistent in the environment. So, you know, be careful what you wish for. More bromine isn't always more better.
Let’s consider another scenario. What about something like an alkene with a double bond? Ethene, C2H4. Two carbons, double bond, two hydrogens on each carbon. Very reactive, that double bond. It’s like a handshake waiting to happen. Now, what if we decide to add bromine across that double bond? This is a super common reaction! You take bromine (Br2) and ethene, and bam! The double bond breaks, and you get 1,2-dibromoethane. CH2Br-CH2Br. Two bromines, one on each carbon that used to be part of the double bond. It’s like the double bond decided to take a break and invited bromine over for a two-for-one deal.
This reaction is so characteristic of alkenes, it's often used as a test to see if something is an alkene. The reddish-brown color of bromine disappears as it reacts with the alkene. Poof! Gone! 1,2-dibromoethane is a liquid, and it's actually used as a lead scavenger in leaded gasoline (though that’s less common now). It’s another example of how adding bromine can totally transform a molecule’s properties and its applications. From a simple gas (ethene) to a liquid with a specific industrial purpose.

What about alkynes? Like ethyne (acetylene), C2H2. Triple bond. Even more reactive than a double bond. If you react ethyne with bromine, you can add two molecules of bromine, first to form 1,2-dibromoethene (which can exist as cis and trans isomers, by the way), and if you keep going, you can end up with 1,1,2,2-tetrabromoethane. Wowza! So, alkynes are like super-inviters for bromine. They’re practically begging to be brominated.
Think about it, we’re going from a simple molecule with just carbon and hydrogen to something loaded with halogens. It’s like going from a minimalist apartment to a fully furnished mansion, but the furniture is all… bromine. And that changes everything. The polarity of the molecule changes, its solubility changes, its boiling point changes, and most importantly, its reactivity changes. It’s not just a cosmetic change; it’s a fundamental alteration of the molecule’s personality and its place in the chemical world.
Let’s not forget about functional groups that already contain elements other than carbon and hydrogen. Imagine you have a carboxylic acid. Like acetic acid, CH3COOH. It’s got that -COOH group, the one that makes things acidic. Now, you’re thinking, “Can I replace a hydrogen on that methyl group with bromine?” Yep, you can! You’d get bromoacetic acid, CH2BrCOOH. This is a really interesting one. The bromine atom, being electronegative, pulls electron density away from the carboxyl group. This makes the hydrogen on the carboxyl group more acidic. So, bromoacetic acid is a stronger acid than acetic acid. It’s like giving your already opinionated friend a megaphone. They’re going to be heard even louder!
Bromoacetic acid is also a pretty nasty chemical. It’s a potent alkylating agent, which means it can add an alkyl group (in this case, a bromoacetyl group) to other molecules. This makes it useful in some organic synthesis, but also means it's quite toxic and corrosive. So, again, that little bromine atom, it’s not playing around. It brings a certain… intensity to the party. It can make things more reactive, more acidic, and often, more dangerous. It’s the chemical equivalent of adding a dash of super-hot chili to your mild salsa. Suddenly, it’s got a kick you weren’t expecting!
What if we had an amine? Like methylamine, CH3NH2. It’s got that nitrogen atom with a lone pair, making it basic. Now, if we try to brominate the methyl group, we’d get bromomethylamine, CH2BrNH2. This is a bit trickier because the nitrogen atom itself is reactive. It’s possible the bromine could react with the nitrogen or the nitrogen could attack the bromine. It’s a bit of a dance of attraction and repulsion. And if you try to replace the hydrogen on the nitrogen with bromine? That’s… well, that’s not really an amine anymore, is it? That's heading into uncharted territory. Things get messy fast.
The key takeaway here, my friend, is that bromine is a powerful substituent. It’s big, it’s electronegative, and it loves to react. When you decide to swap it into a molecule, you’re not just making a minor tweak. You’re fundamentally changing its character. You’re altering its electron distribution, its polarity, its steric bulk, and its overall reactivity. It’s like taking a quiet librarian and turning them into a rockstar. The same person, but a completely different performance.
And the exact outcome? It all depends on where you put that bromine. Is it on a saturated carbon? An aromatic ring? Next to a double bond? Near a functional group? Each location presents a different set of opportunities and challenges for our new brominated friend. It’s a puzzle, really. And the more you play with these molecular puzzles, the more you appreciate the subtle, and sometimes not-so-subtle, ways that elements can transform a molecule. So, next time you’re sipping your coffee and staring at a chemical structure, feel free to let your mind wander. Imagine all the possibilities, all the ways you could spice things up with a little bit of bromine. Just, you know, do it on paper first. For safety’s sake. And for the sake of your molecular sanity!
