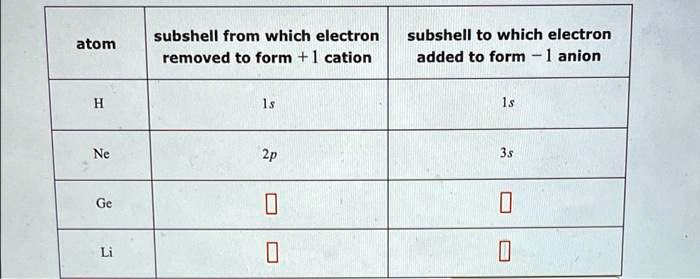
Xe Subshell From Which Electron Removed To Form Cation

Alright, settle in, grab your latte, and let's talk about something that sounds super fancy but is actually, like, the atom's version of a break-up. We're diving into the mysterious world of electron removal and figuring out where these little rascals go when they decide to ditch their atomic squad.
Imagine an atom is a super popular kid in school. It's got all these electrons hanging around it, like a posse. They're chilling in different areas, right? We've got the inner circle, the super-close friends, and then some folks who are a bit further out. These "areas" where the electrons hang out are called subshells. Think of them like different cliques or hangouts within the school. You've got the "S" clique, which is pretty chill and just hangs out in one spot. Then there's the "P" clique, a bit more social and has three different hangouts. The "D" clique is even bigger, with five hangouts, and the "F" clique is like the super-exclusive VIP section with seven spots. It's like a whole social hierarchy, but with way less drama (usually!).
Now, sometimes, an atom decides it's had enough. Maybe it wants to be more stable, like when you decide to declutter your closet and toss out all those clothes you haven't worn since 2008. To become more stable, atoms sometimes kick out an electron. This is how they become cations, which is just a fancy word for a positively charged atom. It's like the atom put on its "cool, independent" hat. And when this happens, we need to know which electron got the boot, right? Which hangout did it leave behind?
This is where the xe subshell comes into play. Now, "xe" isn't a real, official subshell name like S, P, D, or F. Nope. "Xe" is actually the symbol for the element Xenon. And Xenon? It's a noble gas. These guys are the ultimate introverts of the periodic table. They're already so stable, they practically never want to interact with anyone. They're the ones at the party who are just standing in the corner, perfectly content, while everyone else is trying to start a mosh pit.
So, when we talk about the "xe subshell from which an electron is removed to form a cation," it's a bit of a trick question, or maybe a really specific scenario. Usually, when an atom forms a cation, it loses an electron from its outermost shell, the one that's the furthest away from the nucleus (that's the atom's core, the brainy part). Think of it like taking out the trash – you usually toss out the stuff that's been sitting around the longest, right?
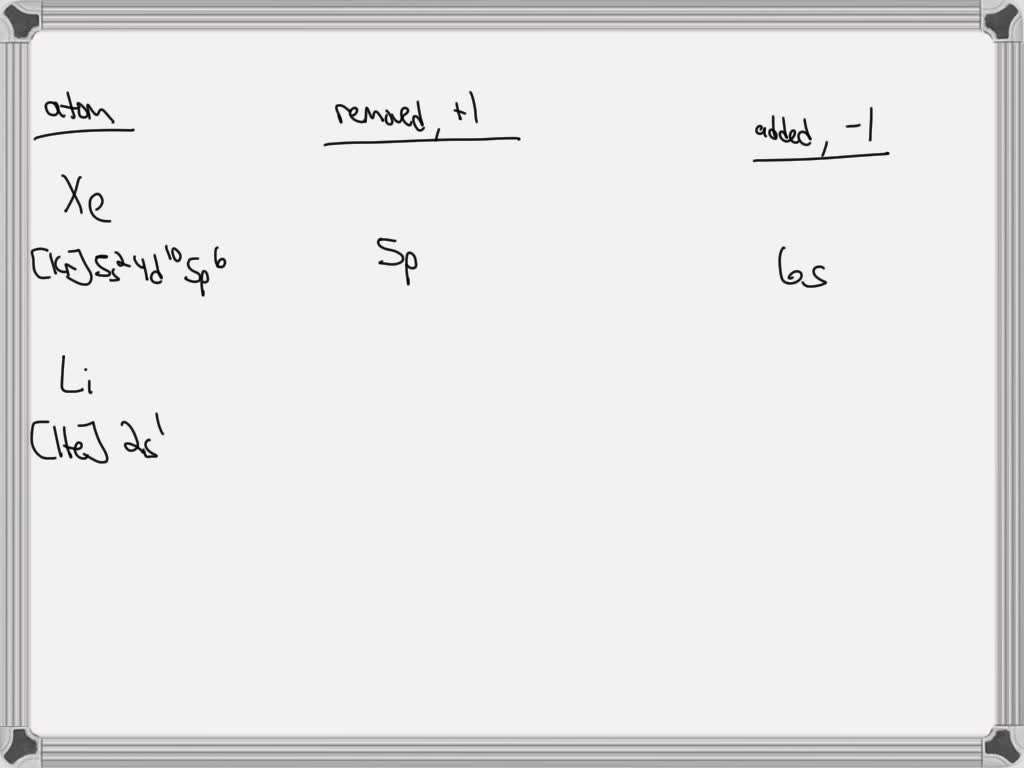
But what if we're talking about a very special kind of cation formation, or perhaps we're using "xe subshell" as a placeholder for the outermost or highest energy subshell? This is where it gets fun. The electrons in the outermost subshell are the most accessible. They're the ones most exposed to the outside world, so to speak. They're the most eager to, you know, leave the party. They have the least amount of energy holding them in place. It's like the person who's been eyeing the exit all night – they're the first one out when the opportunity arises.
Let's get a little nerdy, but in a fun way. The names of subshells (S, P, D, F) actually correspond to the shape of the electron's orbital, which is just the region of space where an electron is most likely to be found. S orbitals are spherical, like a perfectly round bubble. P orbitals are dumbbell-shaped, like a tiny, elegant bowtie. D and F orbitals get much more complicated, with cloverleaf shapes and even more lobes. It’s like the atom’s got its own little collection of origami.
When an atom forms a cation, it's usually looking to achieve a more stable electron configuration. This often means emptying its outermost occupied subshell or half-filling it. Imagine you've got a bunch of boxes, and you're trying to pack them in the most efficient way possible. You'll start with the biggest, most accessible boxes first. That's what the atom does with its electrons.
So, if we're talking about a typical cation formation, the electron is removed from the valence shell. The valence shell is the outermost shell of an atom. And the subshells within that valence shell are where the action happens. For most elements, this will be an S or P subshell. For example, Sodium (Na), that friendly metal that loves to react, loses an electron from its 3s subshell to become Na+. That 3s subshell is its outermost hangout.

But here’s a curveball: what if the atom is already really stable, like Xenon? Xenon has a full outermost shell, a beautiful, complete set of electrons. It’s like the kid who’s already got all the coolest toys and doesn't need anything else. To force Xenon to give up an electron (which is super hard to do), you'd need a ridiculous amount of energy. And if you did manage it, you'd be pulling it from its outermost occupied subshell, which in Xenon's case is the 5p subshell. So, in a roundabout way, if you had to remove an electron from Xenon to make a cation (which is practically a superpower achievement in chemistry), you'd be taking it from the 5p subshell.
The key takeaway here, my friends, is that atoms, like us, sometimes want to simplify. They shed what's no longer serving them to achieve a better state of being. And when they shed an electron to become a cation, it's almost always the electron from the furthest, most accessible subshell that gets the heave-ho. It's the electron with the least commitment, the one ready to explore the vast, positive expanse of the universe. Or, you know, just bond with something else.
So, next time you hear about electron removal and cations, remember it's not just abstract science. It's atoms making choices, decluttering their electron closets, and striving for a more harmonious existence. And sometimes, that involves a little bit of breaking up with an electron. It's chemistry, but it's also, in a way, a tiny, atomic drama unfolding before our very eyes!
