Writing The Net Equation For A Sequence Of Reactions

Ever felt like you’re juggling flaming chainsaws while trying to bake a soufflé? That’s kind of what writing a net equation for a sequence of reactions can feel like sometimes. You know, those multi-step chemical processes where one thing happens, then another, then poof, something totally new shows up? Yeah, those. It’s like trying to follow a recipe that has about a dozen steps, and each step involves its own mini-recipe. But don't sweat it! We're going to break this down, make it as painless as possible, and hopefully, you’ll walk away feeling like you’ve just conquered Mount Everest… or at least found your car keys after a frantic search.
Think about it like this: you’re assembling IKEA furniture. You’ve got a box full of bits, a cryptic instruction booklet, and a vague sense of impending doom. Step one: attach piece A to piece B using screw type C. Okay, got it. Then, step two: take the thing you just made and somehow connect it to piece D, but only after you’ve wrestled with that ridiculously fiddly little dowel. Each step is its own little adventure, and if you mess up one, the whole wobbly monstrosity might just… collapse.
A sequence of reactions, or a reaction mechanism, is pretty similar. Instead of wooden dowels and Allen wrenches, we’re dealing with molecules bumping into each other, breaking bonds, and forming new ones. These individual steps are called elementary reactions. They’re the bread and butter of the overall transformation. And just like with IKEA, sometimes there are intermediates – these are molecules that show up for a bit, do their job, and then disappear, only to be reformed later or turned into something else. They're like that one rogue screw that you find after you've finished, but it turns out you didn't need it anyway. Or, worse, it’s the one screw you actually needed and now your bookshelf is leaning like the Leaning Tower of Pisa.
So, what’s the big deal with the net equation? Well, the net equation is essentially the Cliff's Notes version of the whole shebang. It tells you what you start with (the reactants) and what you end up with (the products), without getting bogged down in all the messy, in-between stuff. It’s the difference between showing someone your entire chaotic kitchen after you’ve baked that soufflé, versus just presenting them with the perfectly risen masterpiece on a plate. Nobody needs to see the flour explosions or the rogue eggshells, right?
Imagine you’re making a sandwich. That’s your overall reaction. You start with bread, cheese, and ham. You end with a sandwich. Simple enough. But the sequence of making that sandwich might be: step one, get bread; step two, open cheese wrapper; step three, place cheese on bread; step four, open ham wrapper; step five, place ham on cheese; step six, place other bread slice on top. See? Lots of little steps! And maybe you even have an intermediate like a “cheesy bread slice” or a “ham-and-cheesy bread slice” before the final sandwich is formed.
When we write the net equation, we’re basically saying, “Okay, I’m starting with bread, cheese, and ham, and I’m ending with a sandwich. The universe doesn’t need to know about the intricate choreography of me fumbling with the cheese wrapper.” It’s the grand finale, the curtain call, the… well, you get the picture. It’s the big, important summary.
Now, how do we actually get this magical net equation from all those individual elementary reactions? It’s kind of like solving a mystery. We have all these clues (the elementary reactions), and we need to piece them together to get the whole story (the net equation).
The key principle is that what goes in must come out, in one form or another. This is like a cosmic accounting rule. If you add up all the reactants from all the individual steps, and then you add up all the products from all the individual steps, they should, in theory, balance out. But here’s the clever part: intermediates are the ghosts of reactions past. They get created in one step and then consumed in a later step. Because they’re there and then they’re gone, they don’t show up in the final net equation. They’re like that friend who always brings snacks to a party but leaves before the main event – you appreciate them, but they’re not part of the closing credits.
Let’s take a super simplified, made-up example. Imagine we have a two-step reaction to make… let’s call it “Sparkle Juice.”

Step 1: A + B → C
Step 2: C + D → Sparkle Juice
Here, A and B are our starting reactants. C is an intermediate. It gets formed in step 1, and then it’s used up in step 2. D is also a reactant, and Sparkle Juice is our final product.
To get the net equation, we simply add all the reactants and products from each step together:
(A + B) + (C + D) → C + Sparkle Juice
Now, for the magic trick: we cancel out anything that appears on both sides of the arrow. See that C chilling on the left and the right? It’s like it’s got a ticket to go in both directions, but ultimately, it just cancels itself out. It’s like having a receipt for a purchase and then returning the item – the transaction effectively didn’t happen in the grand scheme of your bank account.
So, after cancelling out C, we’re left with:
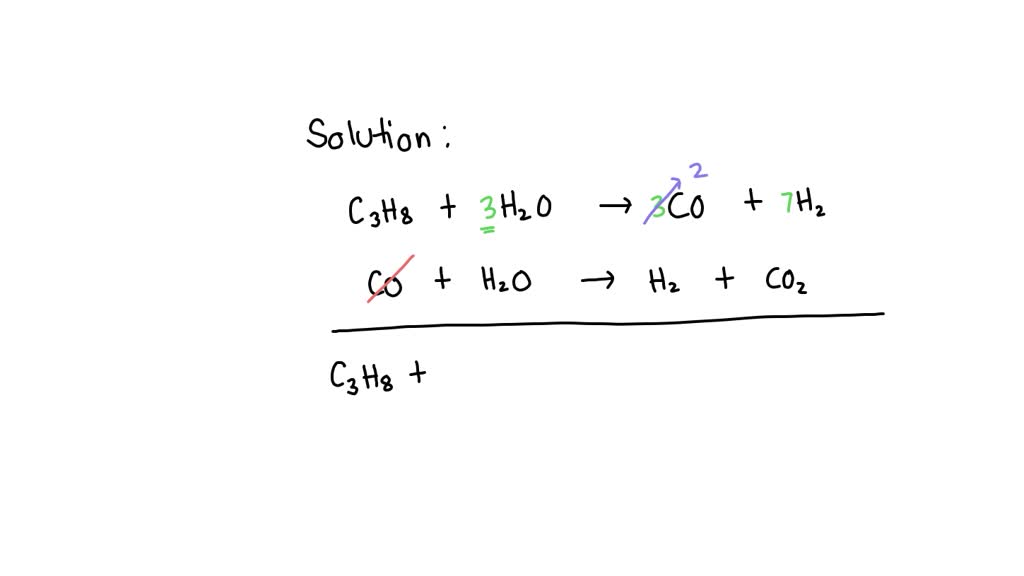
A + B + D → Sparkle Juice
Ta-da! That’s our net equation. It tells us that if we mix A, B, and D together, we’ll eventually get Sparkle Juice. We don’t need to know about the fleeting existence of C to understand the overall transformation.
It’s a bit like following a recipe for a fancy layered cake. You have instructions for making the cake layers, then for making the frosting, and then for assembling. The net equation is like the final picture of the finished cake. You don't see the separate bowls of batter or the whisk covered in cream. You just see the delicious end result.
Sometimes, these reaction sequences can be a bit more complex. You might have multiple intermediates, or a reactant might be used in one step and then regenerated in another. It can get a little dicey, like trying to untangle a knot made of spaghetti. But the principle remains the same: if a species appears on both the reactant and product side of the overall sum of the elementary steps, it gets cancelled out.
Let's try another one, a little more involved. Suppose we're trying to make "Glo-Mist":
Elementary Reaction 1: 2X + Y → Z
Elementary Reaction 2: Z + X → W

Elementary Reaction 3: W → Glo-Mist
First, we add up all the reactants and products:
(2X + Y) + (Z + X) + W → Z + W + Glo-Mist
Now, let’s see what cancels. We have a Z on both sides. Poof! Gone. We also have a W on both sides. Poof! Also gone.
What’s left? On the reactant side, we have 2X and Y, plus another X. So, that’s a total of 3X and Y. On the product side, we're left with Glo-Mist.
So the net equation is:
3X + Y → Glo-Mist
See? It’s like simplifying a complex math problem. You look for common terms and get rid of them to find the elegant, core truth.
The trickiest part is usually correctly identifying the intermediates. They are the species that are formed in one step and consumed in a subsequent step. They are not starting reactants, and they are not final products. They are the supporting actors, the ones who have a brief moment in the spotlight and then exit stage left.
Think of it like a relay race. You have runners passing a baton. The baton is the intermediate. Runner 1 hands off the baton to Runner 2. Runner 2 runs with it and hands it off to Runner 3. Runner 3 carries it across the finish line. In the overall race, what matters is who started the race (the initial reactants) and who finished it with the baton (the final products). The individual runners and the act of passing the baton are the elementary steps and intermediates. The net equation is just the starting line and the finish line, with a clear indication of the overall journey.
One common pitfall is accidentally including a reactant or product that looks like an intermediate but isn’t. For example, if a substance is used in one step but not fully consumed until the very end, it might still show up in the net equation. Or, if a substance is produced in one step and then also produced in another step as a final product, it should be accounted for differently. It’s like trying to sort laundry – you have to be careful about what’s delicates, what’s colors, and what’s whites. You can’t just toss it all in together.
Sometimes, a catalyst also pops up in these reaction sequences. A catalyst is like a helpful friend who encourages a reaction but doesn't actually get used up in the process. They show up, facilitate the reaction, and then are regenerated at the end. So, if you see a catalyst in one of the elementary steps, it will appear on both the reactant and product side of the overall sum, and thus, it will also cancel out in the net equation. Catalysts are the ultimate social butterflies of the chemical world – they facilitate everything but never get stuck with the bill.
The main takeaway here is that writing a net equation is about simplifying complexity. It’s about seeing the forest for the trees, or in this case, the delicious sandwich for all the fiddly bits of bread-handling. By summing up all the individual steps and then strategically cancelling out the intermediates (and catalysts), you arrive at a clean, concise representation of what’s happening on a macroscopic level.
So, next time you’re faced with a multi-step reaction and asked to write the net equation, take a deep breath. Think of your IKEA furniture, your sandwich-making prowess, or your relay race analogy. Remember that intermediates are temporary visitors, and the net equation is just the highlight reel. With a little practice and a good dose of patience, you’ll be writing net equations like a pro, or at least like someone who can assemble a moderately stable Billy bookcase without too many existential crises. Happy reacting!
