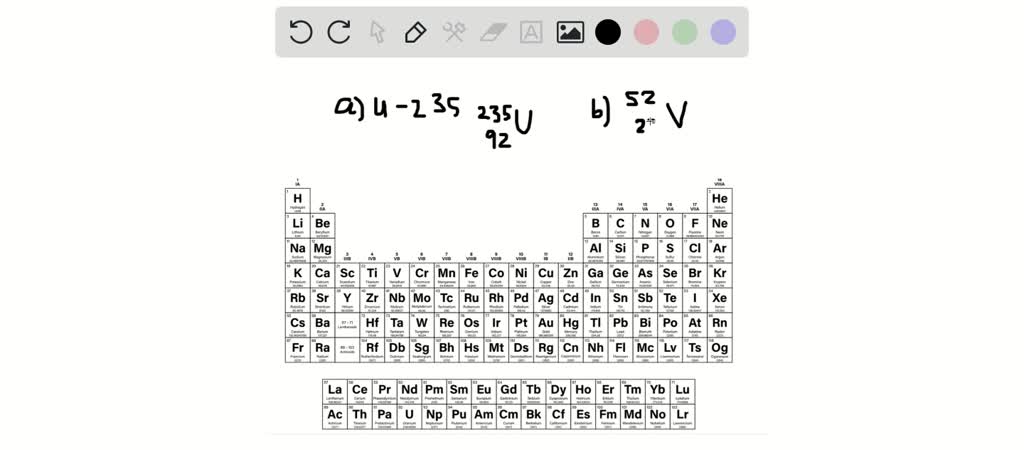
Write The Symbol For Each Isotope In The Form Azx

Hey there, future atom whisperers! Get ready to unlock a secret code that atoms use to tell us who they are and what makes them special. It's like a secret handshake, but for tiny particles! And guess what? You’re already practically an expert.
Think about your favorite cookie recipe. You’ve got flour, sugar, and maybe some chocolate chips, right? Each ingredient is super important. Well, atoms have their own special ingredients too, and they use a super cool symbol to let us know exactly which version of an element we’re dealing with. It’s not as complicated as it sounds, promise!
Imagine you have a special superhero. This superhero has a name, like "Captain Awesome." But sometimes, Captain Awesome has different versions of his powers, maybe one is super fast and the other is super strong. We'd need a way to tell them apart, right? Atoms do the same thing, and their special symbol is their way of showing off their unique "power levels."
This magical symbol is written in a very specific way, like a secret formula. It’s called the isotope notation, and it’s going to be your new best friend in the world of chemistry. Don’t worry, we’re not doing calculus here. We’re just learning how to read a tiny tag that tells a big story.
So, how does this amazing symbol look? It’s like a little puzzle with numbers and a letter. The letter is easy-peasy – it's the normal, everyday name of the element. Think of it as the superhero’s first name. Like, if we’re talking about the element that makes up your bones, that’s calcium, and its symbol is Ca. Simple, right?
Now for the really fun part, the numbers! These numbers are like the superhero's power stats. They tell us something super important about that specific version of the atom. The first number you’ll see is a big, bold number sitting up high, like a crown. This number is called the mass number.
This mass number is basically a count of how much "stuff" is packed inside the atom’s core, its nucleus. Think of it as the total number of building blocks in the atom’s little house. And the nucleus is where all the action happens, like the engine room of a spaceship!
But what are these building blocks? They’re called protons and neutrons. Protons are like the positive, energetic cheerleaders of the atom, always shouting out “We are element X!” The number of protons is what defines an element. It’s the element’s unique ID card, so to speak.
Then we have neutrons. These guys are the chill, neutral friends. They don’t have a charge, they just hang out and add weight. They’re like the sturdy walls of the atom’s house, giving it some extra oomph. So, the mass number is just the total count of these energetic protons and chill neutrons. Easy math!
The second number is a bit smaller and hides down low, almost like it’s peeking out. This number is called the atomic number. And this, my friends, is the true identity of the element. It is the number of protons!
Think of it this way: the atomic number is like your social security number for atoms. It’s unique to each element and never, ever changes for that element. If an atom has 6 protons, it’s always going to be carbon, no matter what else is going on. It’s the element’s ultimate identifier, its DNA.
So, putting it all together, the symbol looks like this: mass numberatomic numberElement Symbol. For example, let's talk about that awesome element carbon again. Pure, unadulterated carbon has 6 protons. So its atomic number is 6.

Now, carbon can come in different "flavors" or isotopes. Some carbon atoms might have 6 neutrons, making their total building blocks (protons + neutrons) equal to 12. This super common version of carbon would be written as 126C. See? The 12 is the mass number, and the 6 is the atomic number.
But wait, there's more! Sometimes carbon can be a little more hefty. Imagine a version of carbon that decided to eat a few extra donuts and gained 8 neutrons. That would give it a total of 14 building blocks (6 protons + 8 neutrons). This heavier version is called carbon-14, and its symbol is 146C.
Notice how the atomic number (the number of protons) is still 6 for both? That's the magic of the atomic number! It’s the anchor that keeps the element firmly in its place. The mass number is the part that can change, creating these different isotopes. It's like having the same type of car, but with different optional packages.
So, 126C and 146C are both carbon, but they are different isotopes of carbon. They have the same number of protons, which makes them the same element, but they have a different number of neutrons, which changes their mass number and makes them slightly different versions. It’s like having twins – they look alike, but they might have different personalities or favorite colors.

Let's try another one. Take the element oxygen, with the symbol O. All oxygen atoms have 8 protons. So, their atomic number is always 8.
Most oxygen atoms are pretty standard and have 8 neutrons. That makes their mass number 16 (8 protons + 8 neutrons). So, this common form of oxygen is written as 168O. Easy peasy, lemon squeezy!
But sometimes, oxygen atoms might be a little more adventurous and have 9 neutrons. This would give them a mass number of 17 (8 protons + 9 neutrons). This isotope of oxygen would be written as 178O.
And you guessed it, there's another one! Some oxygen atoms are even heavier, packing 10 neutrons. That brings their mass number to 18 (8 protons + 10 neutrons). This hefty isotope of oxygen is represented as 188O.
So, 168O, 178O, and 188O are all different isotopes of oxygen. They all have 8 protons, but they have 8, 9, and 10 neutrons respectively. It’s like having three siblings who all have the same last name but are different individuals with their own unique quirks.

Why is this important, you ask? Well, these different isotopes can have wildly different personalities, even though they're the same element. Some isotopes are stable, like a comfy armchair. They’re happy to just hang out.
But other isotopes are a bit more… excitable. They’re what we call radioactive isotopes. These guys are like popcorn kernels before they pop – they’re full of energy and eventually, they’ll release it in a big burst, changing into a different element altogether! This is the stuff of exciting science and even some cool medical treatments.
So, the next time you see a symbol like 23892U, don't be intimidated! You’ll know that U stands for uranium, the 92 down low tells you it has 92 protons (making it definitively uranium), and the 238 up high tells you it has a total of 238 building blocks (protons and neutrons) in its nucleus. This particular isotope of uranium is quite famous for its radioactivity!
The notation AZX is your universal key to understanding these atomic variations. A is the mass number (protons + neutrons), Z is the atomic number (just protons – the element’s ID!), and X is the element's symbol. It’s a simple system, but it unlocks a universe of information about the building blocks of everything around us.
So go forth and conquer! You've just learned a fundamental skill that will make chemistry so much more understandable and, dare I say, fun! It's like you've been given a decoder ring for the atomic world. Isn't that just the coolest? Keep exploring, and remember, every atom has a story to tell, and now you know how to read its name tag!
