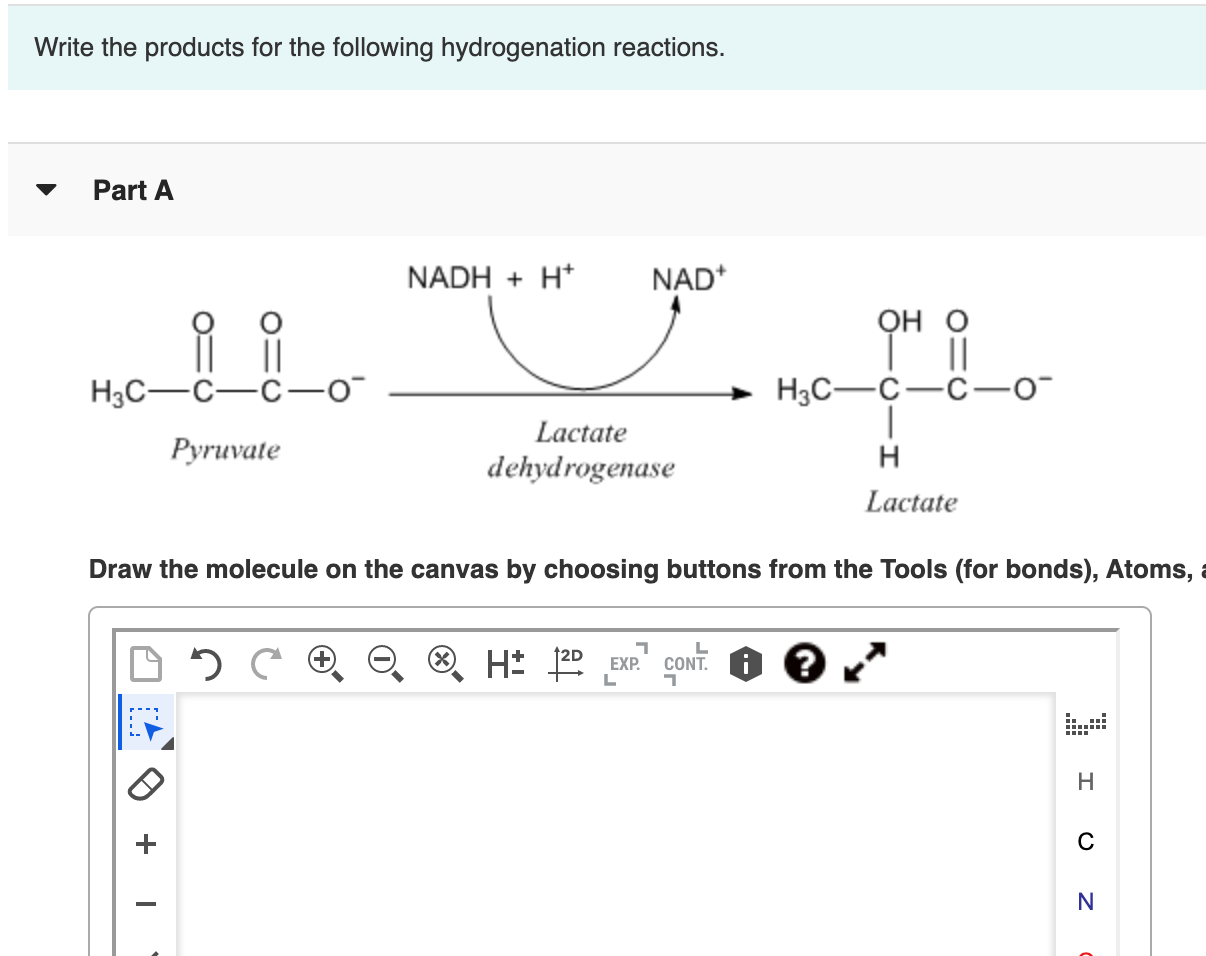
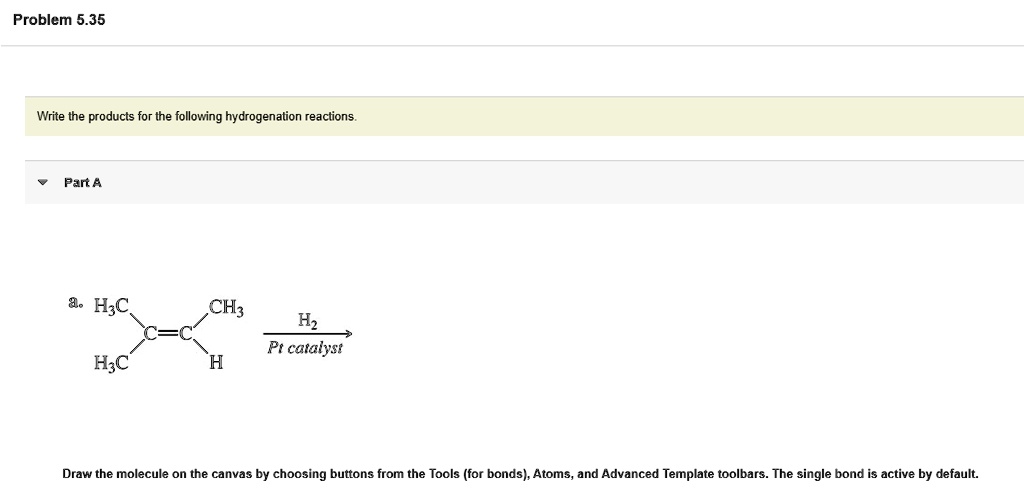
Write The Products For The Following Hydrogenation Reactions
Hey there, fellow science explorers! Ever feel like you’re drowning in a sea of chemical reactions? Yeah, me too sometimes. But fear not! Today, we’re diving into a super cool part of chemistry that’s actually kinda fun: hydrogenation reactions. Think of it as giving molecules a little hydrogen spa treatment. And guess what? We’re going to figure out what awesome new products pop out after this treatment!
So, what exactly is hydrogenation? Basically, it’s adding hydrogen (that’s H₂ for all you chemistry nerds out there) to a molecule. Usually, this happens with the help of a catalyst. Catalysts are like the wingmen of chemistry – they help the reaction happen faster without getting used up themselves. Think of them as the DJs of the molecular dance floor, getting things moving!
Why is this cool? Because it changes things! It can take molecules that are a bit… well, bendy and make them straighter and more stable. It’s like taking a noodle and making it a bit more rigid. Sounds simple, right? But the products we get are everywhere!
Let’s Get Our Hands (Figuratively!) Dirty
We’re not going to get into super complex jargon. We’re going to look at some common examples. Imagine we have some starting materials, and we hit them with hydrogen and a catalyst. What do we get? Let’s find out!
Alkenes: The Unsaturated Fun Lovin' Molecules
First up, let’s talk about alkenes. These guys have a special double bond (C=C). Think of that double bond as a party waiting to happen. Hydrogenation loves crashing these parties. The hydrogen molecules sneak in and break up the double bond, making it a single bond. It’s like turning a two-lane highway into a one-lane country road, but way cooler.
So, if you have an alkene, say, propene (that’s three carbons with a double bond), and you hydrogenate it, what do you get? You get propane! Propane, as in the stuff that fuels your BBQ! How neat is that? From a gas you can’t even see to something that makes your burgers sizzle. Pretty neat, huh?
Let’s draw it out (in our heads, of course!):
Alkene (with a double bond) + H₂ (with a catalyst) → Alkane (with only single bonds)
The double bond is the place where the magic happens. It’s a bit more reactive, like the energetic friend at a party who’s always up for something new. The hydrogen molecules, with the catalyst’s encouragement, jump in and fill the gaps created by breaking that double bond. Boom! You’ve got a saturated molecule. Saturated means it’s holding onto as many hydrogen atoms as it possibly can. It’s like a fully loaded snack pack.
Think about the industrial side of this. Many important chemicals start as alkenes. Hydrogenating them gives us more stable, useful alkanes. It’s a fundamental step in making lots of everyday products. So next time you see a plastic bottle or use something made of rubber, remember, hydrogenation might have played a starring role!
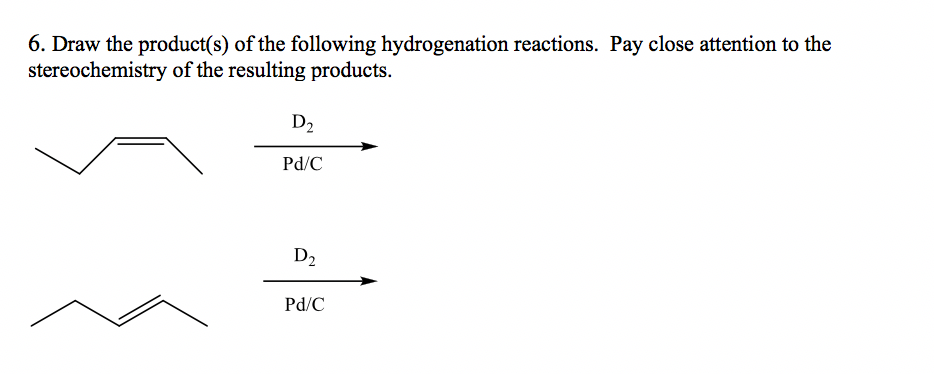
Alkynes: The Double Double Trouble Molecules
Now, let’s crank it up a notch. We have alkynes. These guys have a triple bond (C≡C). That’s like the alkene party, but with a triple-decker sandwich of bonding! These are even more eager for hydrogen to join the fun.
Hydrogenation can happen in stages with alkynes. You can add one molecule of H₂ and break one of the bonds, turning the alkyne into an alkene. Or, you can go all out and add two molecules of H₂, breaking both bonds and turning it into an alkane.
So, if you have ethyne (that’s two carbons with a triple bond, also known as acetylene, the stuff in welding torches), and you hydrogenate it carefully with just one H₂, you get ethene (that’s our old friend, the alkene). If you go wild and add two H₂ molecules, you get ethane (the simple alkane).
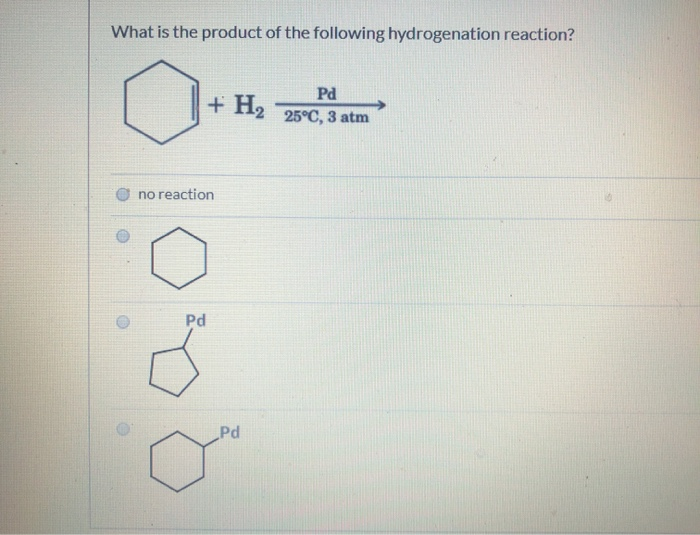
This control is super important. Sometimes you want the alkene. Sometimes you want the full alkane. Chemists are like molecular chefs, adjusting the heat (catalyst) and ingredients (hydrogen) to get exactly the dish (product) they’re craving.
Alkyne (with a triple bond) + H₂ (with a catalyst) → Alkene (with a double bond)
Alkyne (with a triple bond) + 2H₂ (with a catalyst) → Alkane (with single bonds)
It’s like a choose-your-own-adventure for molecules! The type of catalyst you use and the conditions you set can dictate whether you stop at the alkene stage or go all the way to the alkane. It’s a subtle art, but incredibly powerful.
The Quirky Facts and Fun Bits
Did you know that hydrogenation is how we turn liquid vegetable oils into solid or semi-solid fats like margarine? Yep, those long, wiggly unsaturated fat chains in oils get hydrogenated to become more stable and solid. This process is called partial hydrogenation, and it’s responsible for creating those handy solid fats. It’s like taking a floppy spaghetti noodle and giving it a bit of a freeze-dry, making it hold its shape!
However, there’s a quirky (and sometimes not-so-fun) side effect. Sometimes, during partial hydrogenation, the hydrogen doesn’t add cleanly. It can rearrange the double bonds, creating trans fats. These are the ones your doctor tells you to avoid. So, even in this simple reaction, there are layers of complexity and unintended consequences. It’s a reminder that chemistry, while amazing, is also a delicate dance.
Another fun tidbit? Many pharmaceuticals are made using hydrogenation. That’s right, some of the drugs that help us feel better have gone through this hydrogen spa treatment on their way to being made. It’s a key step in building complex drug molecules, ensuring they have the right structure and stability to do their job.
Think about it: from the fuel in your grill to the butter on your toast, to life-saving medicines, hydrogenation is working behind the scenes. It’s like the unsung hero of the chemical world. It doesn’t get much fanfare, but it’s essential!
Beyond the Double and Triple Bonds
Hydrogenation isn’t just for alkenes and alkynes. It can also happen with other functional groups that contain double bonds, like ketones and aldehydes. These reactions often produce alcohols.
For example, if you have acetone (a ketone, the stuff in nail polish remover!) and you hydrogenate it, you get isopropanol (isopropyl alcohol), also known as rubbing alcohol. So, with a bit of hydrogen and a catalyst, you can transform one common chemical into another!

Ketone/Aldehyde + H₂ (with a catalyst) → Alcohol
It’s amazing how adding just a few atoms of hydrogen can completely change the properties and uses of a molecule. It’s a testament to the power of chemical transformation.
Why is This Fun to Talk About?
Because it’s everywhere! It’s not some abstract concept confined to a dusty textbook. Hydrogenation is a real, tangible process that impacts our lives every single day. It’s the magic that makes things happen, often without us even realizing it.
Plus, the idea of adding hydrogen to make things more stable and useful is just inherently satisfying. It’s like giving molecules a little boost, a bit of encouragement. And the products? They’re the tangible results of that little boost. It’s a story of transformation, of potential realized.
So, the next time you’re enjoying a crispy fried food, using a plastic container, or taking a medication, give a little nod to hydrogenation. It’s been busy at work, making our world a little bit different, and often, a lot better. Keep exploring, and keep asking questions. Chemistry is full of these fun little secrets!
