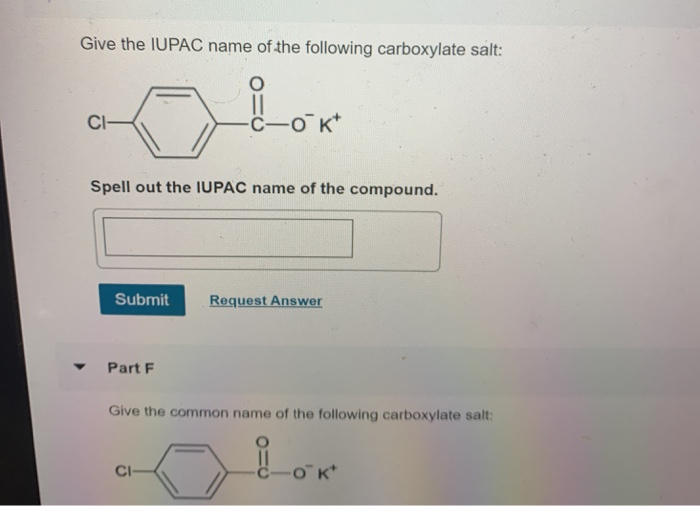
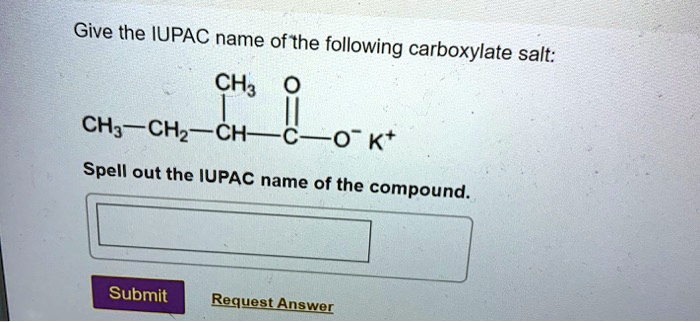
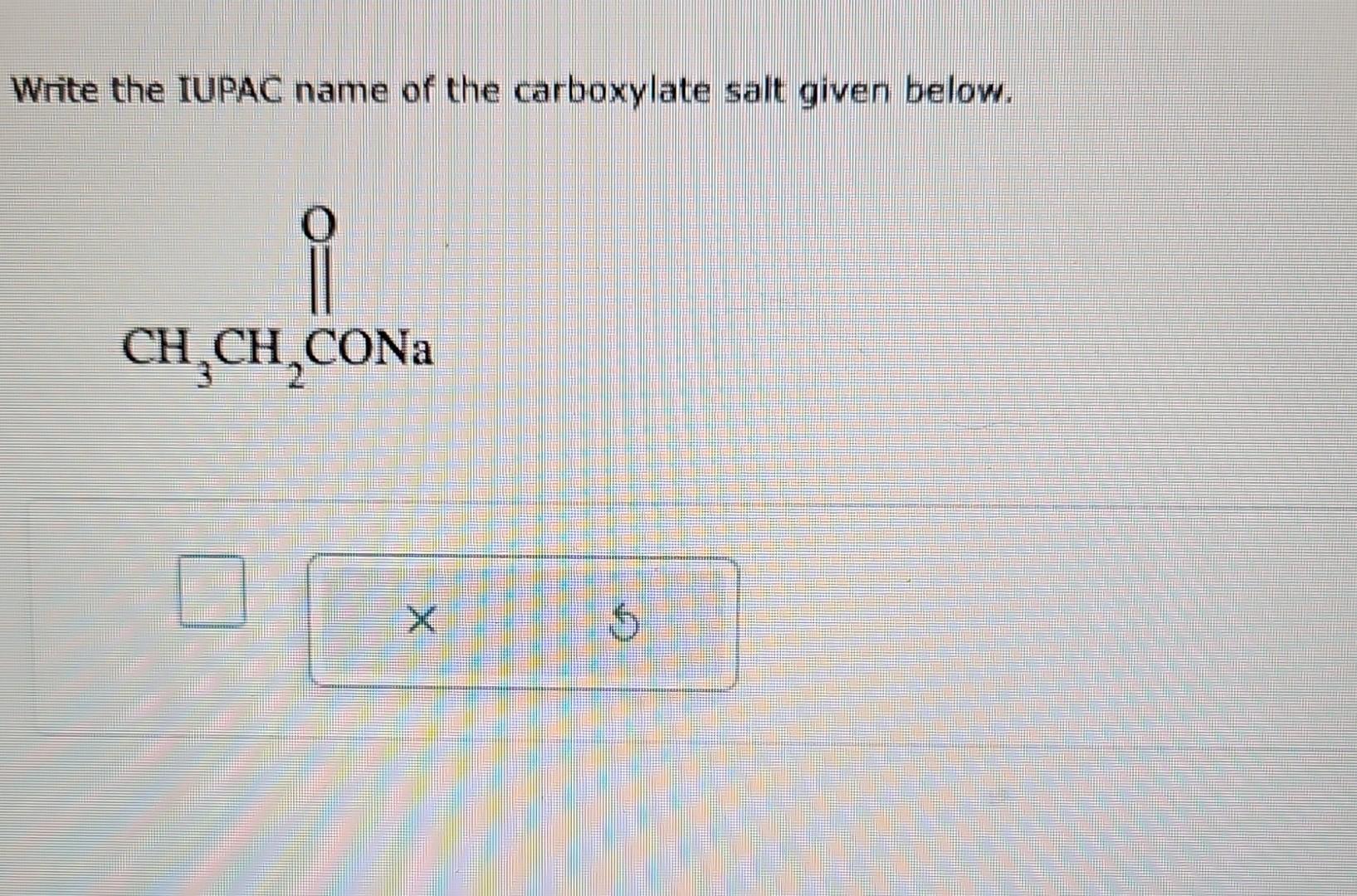
Write The Iupac Name Of The Following Carboxylate Salts:
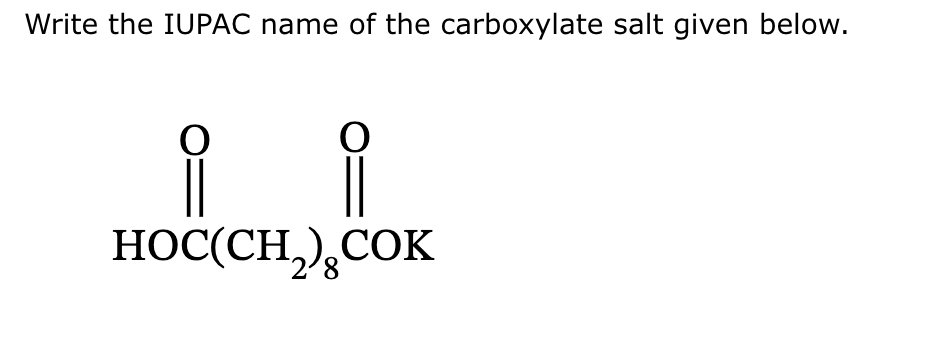
So, I was rummaging through my grandma’s attic the other day, you know, the usual Saturday morning adventure. Dust bunnies the size of small rodents, forgotten treasures, and that distinct smell of aged paper and mothballs. I stumbled upon this old, leather-bound chemistry textbook. Seriously, the pages were so brittle, I was half expecting them to turn into confetti when I touched them. It belonged to my great-uncle, apparently a bit of a whiz with… well, chemicals. Mostly, I just remember him making really impressive, albeit slightly alarming, fireworks. Anyway, I flipped it open, and there it was, a whole chapter dedicated to something called “carboxylate salts.” My eyes immediately glazed over. I mean, who names these things? It sounds like something out of a sci-fi movie where they’re trying to synthesize a new alien super-weapon. But then, I got thinking. We use these things everywhere, probably without even realizing it.
Like, think about it. That lovely, smooth lotion you use after a shower? Chances are, it’s got some fancy-sounding ingredient that’s secretly a carboxylate salt. That slightly tangy taste in some of your favorite snacks? Yep, you guessed it. And don't even get me started on cleaning products. They’d be a lot less effective without these chemical superheroes. So, while the names might sound intimidating, the reality is, these are the unsung heroes of our everyday lives. And as I was leafing through this ancient tome, I realized something else: naming them isn't just some nerdy academic exercise. It's actually pretty logical, once you get the hang of it. It’s like learning a secret handshake for the world of chemistry. So, let’s dive in, shall we? No need for lab coats or goggles, just your trusty curiosity and maybe a comfy chair. We’re going on a journey into the wonderfully (and sometimes weirdly) named world of carboxylate salts.
The Humble Beginnings: What Even Is a Carboxylate Salt?
Before we can even think about naming things, we need to know what we’re dealing with. So, let’s break it down. A carboxylate is basically what you get when a carboxylic acid loses a hydrogen atom. Imagine a carboxylic acid as a molecule with a special little group on it, a bit like a grumpy old man with a cane. This grumpy old man is the hydrogen atom. When he finally decides to take a break and wander off, the rest of the molecule, the carboxylic acid part, becomes a carboxylate ion. It's now carrying a negative charge, like it’s a bit miffed about losing its companion. Don't worry, this is a perfectly normal chemical event!
Now, salts, in the chemical sense, are generally formed when an acid and a base react. Think of it like a little chemical dance where they neutralize each other. In the case of carboxylate salts, our carboxylate ion (which is essentially the leftover, charged part of a carboxylic acid) teams up with a positive ion, usually a metal ion. This positive ion is called a cation. So, you have your negatively charged carboxylate and your positively charged cation, and voila! They form an ionic bond, sticking together like magnets. This is your carboxylate salt. Pretty neat, huh?
Think of it this way: If carboxylic acids are like families with a hydrogen child, carboxylate ions are those families after the child has moved out to college. And carboxylate salts? They're like those families adopting a new pet (the cation) to fill the void. It’s all about balancing charges and making stable compounds. So, when you see a carboxylate salt, picture a negatively charged organic bit (the carboxylate) happily bonded to a positively charged metal or other ion. It’s a partnership!
The Naming Convention: Where the Magic (and Confusion) Happens
Alright, so now we know what a carboxylate salt is. The next big question is: how on earth do we give these things names? And more importantly, how do we make them sound less like they belong in a mad scientist's lab and more like… well, useful things?
This is where the International Union of Pure and Applied Chemistry, or IUPAC, comes in. They’re the ultimate word on chemical nomenclature. Their rules are designed to be unambiguous, meaning that no matter where you are in the world, if you say the IUPAC name of a compound, everyone knows exactly what molecule you’re talking about. It’s like having a universal chemical language. Pretty handy, if you ask me.
The naming of carboxylate salts follows a pretty straightforward pattern, once you get the hang of it. It's essentially a two-part name. The first part tells you what the positive ion is, and the second part tells you what the negative ion (the carboxylate part) is. Easy peasy, right? Well, almost. The tricky bit, as with most things in chemistry, is remembering the root names and how they change.
Let’s break down that second part – the carboxylate. Remember how I said it comes from a carboxylic acid? Well, the naming of the carboxylate ion is directly linked to the naming of its parent carboxylic acid. For simple, straight-chain carboxylic acids, the IUPAC name ends in -oic acid. For example, the simplest one, with one carbon atom, is methanoic acid. The next one, with two carbon atoms, is ethanoic acid. Got it so far?
Now, here’s the crucial transformation: when that carboxylic acid becomes a carboxylate ion, the -oic acid ending gets replaced with -oate. So, methanoic acid becomes methanoate, and ethanoic acid becomes ethanoate. See? The “acid” part is gone, and the “ate” part takes its place, signaling that it’s now an ion. This is where you start to see the connection to common names too. For instance, ethanoic acid is also known as acetic acid, and its salt, sodium ethanoate, is commonly known as sodium acetate. You've probably heard of that one!
The first part of the name is for the cation. If it’s a simple metal cation like sodium (Na+), potassium (K+), or calcium (Ca2+), you just use the name of the metal. So, sodium ethanoate, potassium methanoate, calcium propanoate. If it’s a more complex cation, the naming gets a bit more involved, but for the most common carboxylate salts, it's usually just the metal name.
Let's Practice! Some Examples to Get Your Chemistry Brain Buzzing
Okay, theory is great and all, but let's get our hands dirty (metaphorically, of course). Here are a few examples, and we'll walk through how to name them using our newfound IUPAC superpowers. Imagine you’re looking at a chemical formula. You see the positive ion first, then the negative ion. We’re going to focus on the negative, the carboxylate part, and then tack on the positive part.
Example 1: CH3COO- Na+
First, let’s look at the anion: CH3COO-. This is the ethanoate ion. How do we know? Well, the CH3-COO- part comes from ethanoic acid. It has two carbon atoms in total (one in the CH3 and one in the COO-). So, the parent acid is ethanoic acid, and the ion is ethanoate.
Now, what’s the cation? It’s Na+, which is the sodium ion.
Putting it all together, the IUPAC name for CH3COO- Na+ is sodium ethanoate. See? Not so scary after all. It just tells you you’ve got sodium, and it’s bonded to the ethanoate part of ethanoic acid.
Example 2: C2H5COO- K+
Let’s tackle this one. The anion is C2H5COO-. How many carbon atoms are there in the parent acid? We have two carbons in the C2H5 chain, and one carbon in the COO- group. That makes a total of three carbon atoms.
What’s the IUPAC name for a carboxylic acid with three carbon atoms? It’s propanoic acid. So, the anion, C2H5COO-, is the propanoate ion.
And the cation? It’s K+, which is the potassium ion.
Therefore, the IUPAC name for C2H5COO- K+ is potassium propanoate. You’re officially a carboxylate salt naming whiz! Keep it up!
Example 3: (CH3)2CHCOO- Ca2+
This one looks a little more complex with those parentheses, but don't let it intimidate you! Let's figure out the number of carbons in the parent acid first. Inside the parentheses, we have (CH3)2, which means two CH3 groups. That's two carbons. Then we have another CH group. So, that’s 2 + 1 = 3 carbons from the (CH3)2CH part. And finally, we have the carbon in the COO- group. So, in total, we have 3 + 1 = four carbon atoms.
A carboxylic acid with four carbon atoms is butanoic acid. So, the anion (CH3)2CHCOO- is the butanoate ion. You might also recognize this as the anion of isobutyric acid, but we're sticking to IUPAC here for maximum clarity!
Now for the cation: Ca2+. This is the calcium ion.

Here's a little curveball: calcium has a 2+ charge. This means we need two butanoate ions to balance out one calcium ion. This is reflected in the chemical formula (Ca(CH3)2CHCOO)2, though sometimes for naming, we focus on the simplest ratio. So, the name for this salt is calcium butanoate. When you see a metal with a variable charge (like iron, copper, etc.), you'd usually specify the charge in Roman numerals, but for metals like calcium, sodium, and potassium, it's generally understood. So, no need for “calcium(II) butanoate” here!
Example 4: HCOO- Mg2+
Let's look at the anion: HCOO-. This is the simplest carboxylate ion. It comes from a carboxylic acid with just one carbon atom. What’s that called again? Ah yes, methanoic acid. So, the ion is methanoate.
The cation is Mg2+. This is the magnesium ion.
Similar to calcium, magnesium has a fixed charge. So, we need two methanoate ions to balance the Mg2+. The IUPAC name is magnesium methanoate. You might know methanoic acid by its common name, formic acid (hence why you might see this referred to as magnesium formate). But for our IUPAC purposes, it's magnesium methanoate!
The Beauty of Branching Chains and Other Quirks
So far, we've looked at pretty straightforward, straight-chain carboxylic acids. But what happens when things get a little more… branched? This is where IUPAC really shines in its ability to provide clear, descriptive names. For branched-chain carboxylic acids, you find the longest continuous carbon chain that includes the carboxyl group. That chain determines the parent name. Then, you number the carbons in that chain starting from the carboxyl carbon (which is always carbon #1).
Let's say you have a structure like this: CH3-CH(CH3)-COOH. The longest continuous chain containing the COOH group has three carbons. So, the parent acid is propanoic acid. However, there's a methyl group (CH3) attached to the second carbon atom of that propanoic acid chain. So, the IUPAC name of the acid is 2-methylpropanoic acid.
Now, if this acid loses its hydrogen to form a salt, the anion would be 2-methylpropanoate. If it were to form a salt with sodium, the IUPAC name would be sodium 2-methylpropanoate. See how the numbering and the substituent name are incorporated?
This system allows for naming even incredibly complex organic molecules in a systematic way. It’s like giving every molecule a unique address. It might seem a bit overwhelming at first, with all the numbers and prefixes, but it's incredibly powerful.
What about dicarboxylic acids? These have two carboxyl groups. Their salts are named accordingly. For example, the salt of ethanedioic acid (oxalic acid), which has two carboxyl groups, would be an ethanedioate salt. So, if you had sodium ethanedioate, it would be Na2C2O4. The naming here just extends the principle. You're still looking at the parent hydrocarbon chain and then adding the "-dioate" ending to indicate two carboxylate groups.
Why Does This Even Matter (Besides Impressing Your Friends at Parties)?
Honestly, the naming is a bit like learning grammar. It’s the foundation for understanding and communicating chemical information. When a chemist anywhere in the world sees the IUPAC name of a carboxylate salt, they know its exact structure, its potential properties, and how it might react. This is crucial for research, for developing new medicines, for creating new materials, and for ensuring safety in chemical processes.
And as I mentioned at the beginning, these compounds are all around us. Sodium stearate is a common soap. Calcium propionate is used as a food preservative. Ammonium citrate is used in soft drinks. The list goes on and on! Understanding their names allows us to understand their roles and origins.
So, the next time you pick up a product and see a long, complex ingredient list, take a peek. You might just spot a carboxylate salt name that you now understand. It’s a little bit of chemistry knowledge that can demystify the world around you. It’s like having a secret decoder ring for everyday products. Pretty cool, right? It's not just about memorizing names; it's about understanding the language of chemistry and appreciating the intricate, often invisible, world of molecules that shape our lives.
And who knows, maybe next time you’re in your grandma’s attic, you’ll find an even older chemistry book with even more bizarre-sounding compounds. But with the tools you've gained today, you'll be a little less intimidated and a lot more curious. That, my friends, is the true magic of learning. Now, if you'll excuse me, I think I’ll go find some sodium ethanoate… I mean, sodium acetate. You know, for science. Or maybe just for a bit of flavor in my popcorn. 😉
