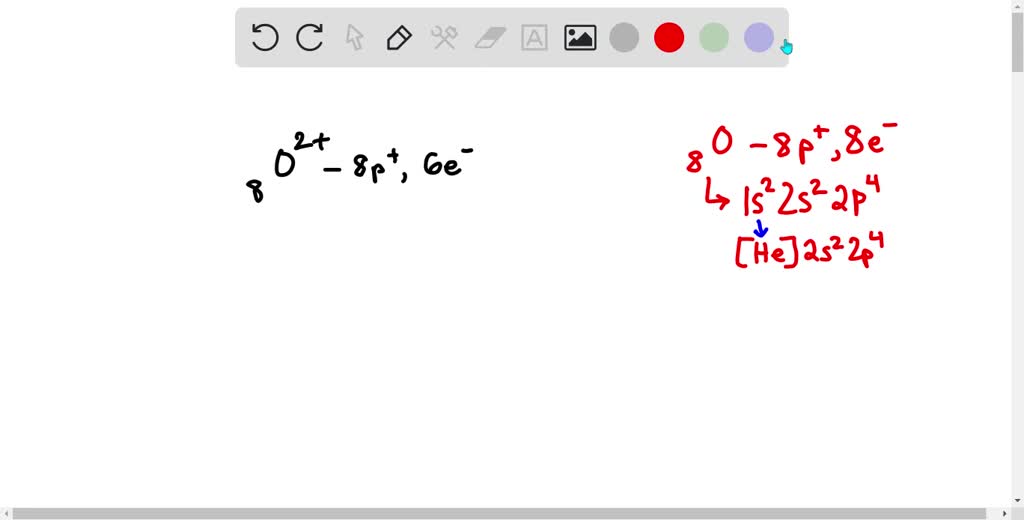
Write The Full Ground State Electron Configuration Of O2+
Hey there, ever wondered what makes the air we breathe, well, breathable? It's all thanks to oxygen, that invisible superhero that keeps us going. But what happens when oxygen gets a little bit... charged up? Today, we're going to dive into something a little bit science-y, but I promise, it's going to be a fun ride! We're talking about the full ground state electron configuration of O2+. Sounds complicated? Think of it like figuring out the perfect seating arrangement for a group of very particular friends at a party.
Now, before you click away thinking this is way too much like chemistry class, let's break it down. Imagine electrons are like little energetic party guests. They have specific rules about where they like to hang out, and they prefer to be as far away from each other as possible, or snuggled up in pairs in comfortable spots. These spots are called "orbitals," and they have different energy levels, kind of like different floors in a fancy hotel. The ground state just means everyone is in their most relaxed, lowest-energy seating arrangement. No jumping around or getting too excited!
So, we're talking about O2+. What does that even mean? Well, "O2" is simply two oxygen atoms hanging out together, like a dynamic duo. Think of it as a couple of best friends always together. Oxygen, bless its heart, usually likes to have a certain number of these little electron guests. A neutral oxygen atom has 8 electrons. So, two oxygen atoms would have a total of 16 electrons.
But then there's that little plus sign, the "+". This is where things get interesting! That little plus sign is like a cosmic sigh of relief for one of the electrons. It means our O2 duo has lost one electron. So, instead of having 16 electrons, our O2+ is now a bit leaner, with only 15 electrons to arrange. It's like one friend at the party decided to leave a little early, maybe to grab some extra snacks or just needed some personal space.
Now, the electron configuration is basically the address book for these electrons. It tells us which orbital each of the 15 electrons is calling home. We fill these orbitals in a specific order, starting from the lowest energy level and moving up. It’s like filling up a parking garage, starting with the most convenient spots on the ground floor and then moving to higher levels only when the lower ones are full.
The first "hotel floor" or orbital type is the 1s orbital. This is the coziest, lowest-energy spot. It can hold a maximum of two electrons. So, our O2+ has 15 electrons, and the first two comfortably settle into the 1s orbital. We write this as 1s2. The '1s' is the location, and the '2' is the number of electrons chilling there.

Next up is the 2s orbital. This is like the next floor in our hotel. It can also hold a maximum of two electrons. So, we place another two electrons here, and our configuration becomes 1s22s2. We've now accounted for 4 electrons (2 + 2).
Now, things get a little more crowded, and we move to the 2p orbitals. These are like a set of three smaller rooms on the same floor, each capable of holding two electrons. Think of them as a suite of rooms! But here’s the catch: electrons are a bit like shy people in a crowded room; they’d rather have their own space if possible. So, before they start pairing up, they spread out into these three rooms (let's call them 2px, 2py, and 2pz) first, with one electron in each, if there are enough electrons. These 2p orbitals have slightly different energies, but for our ground state configuration, we group them together. Each of these three 2p orbitals can hold up to two electrons, meaning the whole 2p subshell can hold a total of 6 electrons.
So, we put 6 electrons into the 2p orbitals. Our configuration now looks like 1s22s22p6. That's 2 + 2 + 6 = 10 electrons placed. We're getting closer!

We've got 15 electrons in total, and we've placed 10. That leaves us with 5 more electrons to find a home for. Where do they go? They move to the next available energy level, which is the 3s orbital. Similar to the 1s and 2s, the 3s orbital can hold a maximum of two electrons. So, we pop two electrons in there. Our configuration is now 1s22s22p63s2. That’s 10 + 2 = 12 electrons accounted for.
We still have 3 more electrons to place (15 total - 12 placed = 3 remaining). These last three electrons will also head to the 3p orbitals, which, just like the 2p orbitals, consist of three smaller orbitals. Remember how electrons like their own space? Since we have three electrons and three 3p orbitals, they will each take up one of these orbitals. So, we’ll have one electron in each of the 3p orbitals. Our final configuration for O2+ is 1s22s22p63s23p3.
Let's count them up one last time to be sure: 2 (in 1s) + 2 (in 2s) + 6 (in 2p) + 2 (in 3s) + 3 (in 3p) = 15 electrons. Perfect! This is the full ground state electron configuration of O2+. It’s like a perfectly organized closet where every item has its designated spot.

Now, you might be thinking, "Why should I care about where these tiny electrons hang out?" Well, it's actually pretty darn important! Understanding electron configurations helps scientists predict how molecules will behave. It's like knowing your friends' personalities; you can guess how they'll react in different situations.
For example, knowing the electron configuration of O2+ helps us understand things like: * Reactivity: How likely is this charged oxygen molecule to react with other things? Is it going to be a gentle handshake or a full-on embrace? This matters in everything from chemical reactions in industry to biological processes in our bodies.
* Bonding: How will this O2+ molecule interact with other molecules? Can it form stable partnerships? This is key to understanding how materials are formed and how they hold together.
Think about it: the very air we breathe is made of oxygen molecules, O2. Even though O2+ isn't what we're breathing directly, understanding its behavior helps us understand the broader world of chemistry that impacts our lives in countless ways. It’s the foundation for creating new medicines, developing advanced materials, and even understanding the atmospheres of distant planets!
So, the next time you take a deep breath, spare a thought for those little electrons. They're not just floating around randomly; they're organized, following rules, and ultimately, contributing to the incredible world around us. And who knew figuring out the electron configuration of O2+ could be a little bit like a fun puzzle? It’s a reminder that even the most complex scientific concepts have relatable analogies, and a little bit of curiosity can unlock a whole universe of understanding.
