Write The Formula For The Conjugate Base Of Each Acid
Ever feel like you're just trying to keep it all together, a delicate balancing act between your morning latte and that never-ending to-do list? Well, in the wild world of chemistry, acids are kind of the same way. They're these energetic molecules, always ready to lend a hand – or, more precisely, a proton. But what happens when they give up that proton? That’s where the magic of the conjugate base comes in, and honestly, it’s not as scary as it sounds. Think of it as a chemical support system, a chill evolution from their more assertive acid selves.
We're diving into the serene waters of acid-base chemistry, but with a vibe that’s more beach read than textbook cram session. Forget those intimidating equations for a sec. We're talking about the fundamental concept of what's left behind when an acid does its thing. It’s like when your favorite artist releases a new album – the original is amazing, but the acoustic version (the conjugate base, in this analogy!) offers a whole new, often more intimate, perspective.
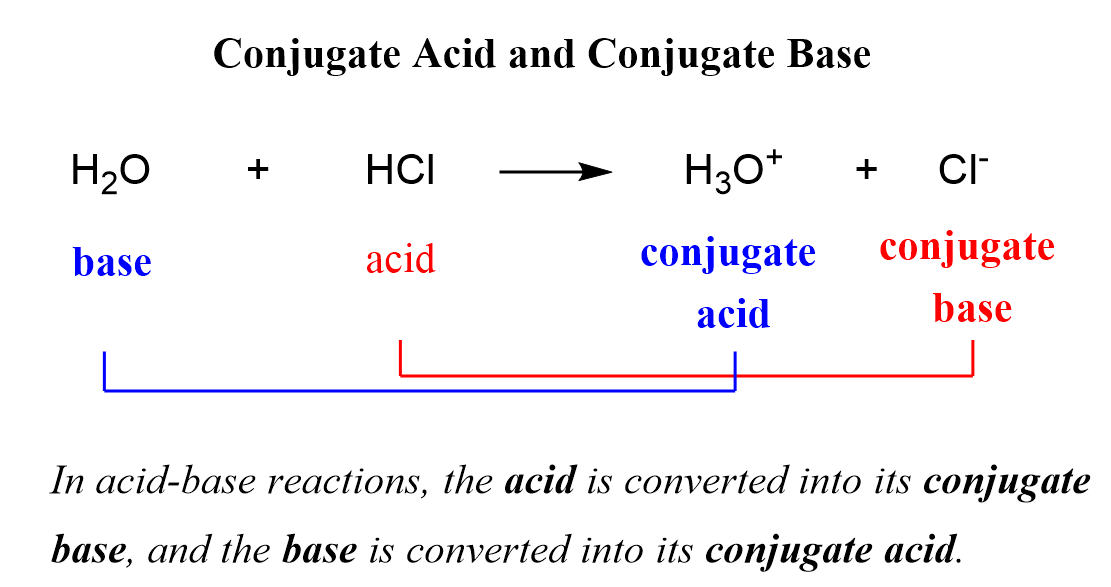
So, what exactly is a conjugate base? In the simplest terms, it's what remains after an acid has donated a proton (a little positively charged hydrogen ion, H⁺). It’s like that feeling after a really good workout – you’re a bit lighter, a bit different, but ready for what’s next. This concept is the bedrock of so much chemistry, from the fizz in your soda to the buffering systems in your body.
The Proton, The Star of the Show
Let's get friendly with the proton, the H⁺. This tiny guy is the key player in acid-base reactions. When we talk about an acid, we’re usually talking about a molecule that’s eager to get rid of a proton. Think of it as a generous friend who’s always willing to share their last slice of pizza. The acid, by donating this proton, becomes its conjugate base.
This process is often described using the Brønsted-Lowry definition of acids and bases. An acid is a proton donor, and a base is a proton acceptor. When the acid donates its proton, it transforms into its conjugate base. This newly formed conjugate base is then perfectly poised to accept a proton itself, making it a base.
Decoding the Formula: The Art of the Conjugate Base
Writing the formula for a conjugate base is surprisingly straightforward, once you get the hang of it. It’s all about that missing proton. For every acid, its conjugate base is simply the acid minus one H⁺. It’s like deconstructing a recipe: you know the ingredients of the original dish, and when you remove one key element, you get a variation.
Let’s break it down with some common examples. These are the molecules you'll encounter in your lab coat dreams (or, you know, when you're trying to figure out why your homemade pickles are a bit too sour).
The Mighty Hydrochloric Acid (HCl)
Hydrochloric acid is a strong acid, like a perfectly brewed espresso – powerful and to the point. When HCl decides to donate its proton, it’s like that espresso shot giving you a jolt of energy. What’s left behind?
The acid is HCl.
It donates a proton (H⁺).
So, the formula for its conjugate base is Cl⁻.
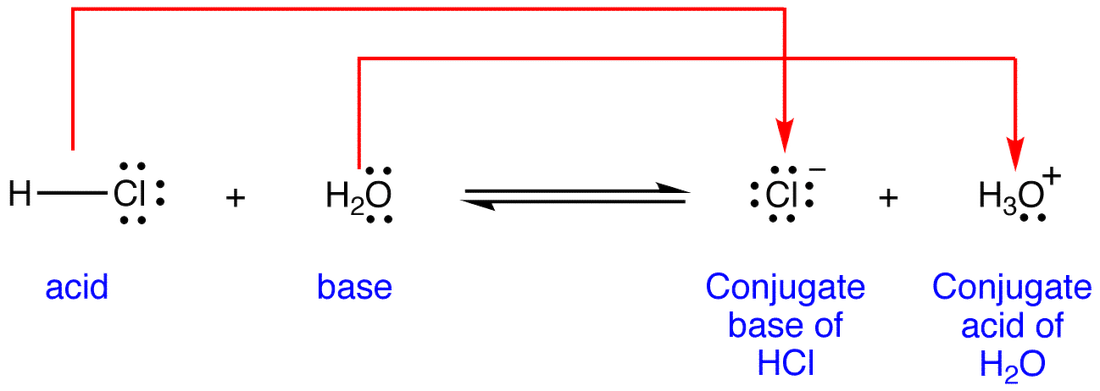
This is the chloride ion. See? No more hydrogen to give away. It’s the chill, slightly negatively charged aftermath. Think of it as the quiet hum after the intense burst of caffeine. It’s still a part of the story, just in a different role.
The Versatile Acetic Acid (CH₃COOH)
Acetic acid is the star of vinegar, that tangy delight that elevates everything from salad dressings to marinades. It’s a bit more complex than HCl, with a whole methyl group (CH₃) hanging around. But the principle is the same.
The acid is CH₃COOH.
It donates its acidic proton (the one attached to the oxygen).
So, the formula for its conjugate base is CH₃COO⁻.
This is the acetate ion. It still has that methyl group, but the proton that was previously attached to the -COOH part is gone. This is where things get interesting. The acetate ion is what gives vinegar its characteristic bite, and it’s also a key player in many biological processes.
Fun Fact: The acetate ion is so common that it's even found in some synthetic flavorings, giving that distinct "vinegar-y" note without the actual vinegar. It's like the distilled essence of tang!
The Classic Sulfuric Acid (H₂SO₄)
Sulfuric acid is a powerhouse, used in everything from fertilizer production to refining metals. It’s the industrial titan of acids. It can even donate two protons, but we’re focusing on the first one for its primary conjugate base.
The acid is H₂SO₄.

It donates one proton (H⁺).
So, the formula for its conjugate base is HSO₄⁻.
This is the bisulfate ion (or hydrogen sulfate ion). Notice it still has a hydrogen, but it's now negatively charged because it lost one positively charged proton. This bisulfate ion can itself act as an acid, donating its remaining proton to form the sulfate ion (SO₄²⁻).
This step-wise donation is super common with polyprotic acids (acids with more than one acidic proton). It’s like a chemical relay race, passing the baton (the proton) one at a time.
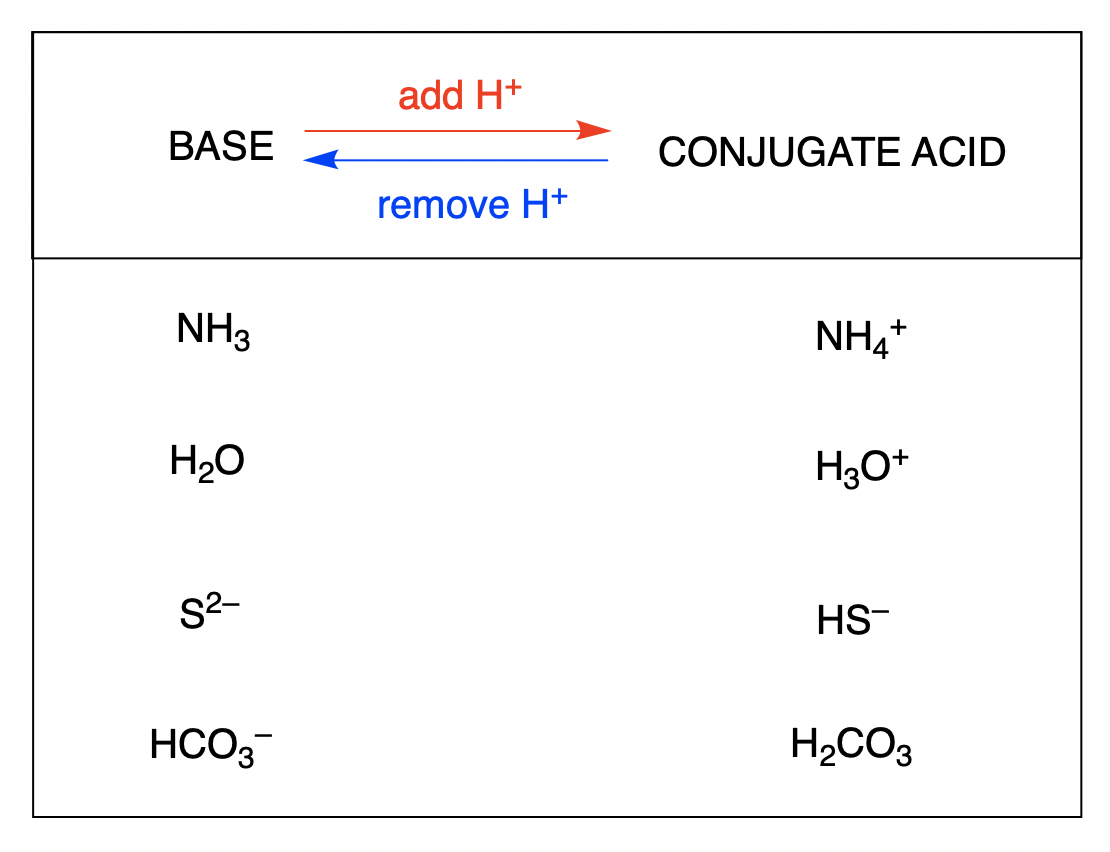
The Building Block: Water (H₂O)
You might be surprised to see water on this list, but it can act as an acid! It’s a bit of a chameleon in the chemical world, often acting as a base, but it can donate a proton.
The acid is H₂O.
It donates a proton (H⁺).
So, the formula for its conjugate base is OH⁻.
This is the hydroxide ion. This is a super important ion in chemistry and biology. In fact, water itself is in equilibrium with hydroxide ions and hydronium ions (H₃O⁺), the conjugate acid of water. This delicate balance is what gives pure water its neutral pH.
Cultural Connection: The hydroxide ion is the key ingredient in many cleaning products, like drain cleaner. It's a strong base because it readily accepts protons. So, while it’s fundamental to life, it also has its powerful industrial uses!
The Slightly More Complex: Phosphoric Acid (H₃PO₄)
Phosphoric acid is another polyprotic acid, found in sodas and also important in biological systems (think DNA and ATP!).
The acid is H₃PO₄.
It donates one proton (H⁺).
So, the formula for its conjugate base is H₂PO₄⁻.
This is the dihydrogen phosphate ion. Like sulfuric acid, phosphoric acid can lose more protons sequentially, forming HPO₄²⁻ (hydrogen phosphate ion) and finally PO₄³⁻ (phosphate ion).
These different phosphate ions are absolutely vital in biochemistry. They’re part of the backbone of DNA, they carry energy in the form of ATP, and they act as buffers in our cells to maintain a stable pH. They’re the unsung heroes of cellular life!
The Organic Touch: Phenol (C₆H₅OH)
Phenol is an organic compound that’s a bit acidic, though not as strong as many inorganic acids. It’s got that benzene ring, which adds a whole aromatic flair.
The acid is C₆H₅OH.

It donates its proton (the one attached to the oxygen).
So, the formula for its conjugate base is C₆H₅O⁻.
This is the phenoxide ion. The benzene ring makes the proton on the oxygen slightly more acidic because it can delocalize the negative charge. It’s like the ring helps to spread out the "discomfort" of being negatively charged, making it more stable.
Lifestyle Tip: While phenol itself is a bit harsh, its derivatives are used in many everyday products, including some antiseptics and even as flavoring agents (though in very small, safe amounts!). The chemistry of organic molecules is all around us.
The Practical Magic: Why Does This Matter?
Understanding conjugate bases isn't just an academic exercise. It's the key to understanding how so many natural processes and chemical reactions work. Buffering systems, for instance, rely on the interplay between acids and their conjugate bases to maintain a stable pH. Our blood, for example, uses a bicarbonate buffering system to keep our pH within a very narrow, life-sustaining range.
Think about it: if your body produces too much acid, the conjugate base in the buffer system is there to soak it up, preventing a drastic pH change. It’s like having a really good shock absorber for your internal chemistry. This is how we can go from intense exercise (which produces lactic acid) to a relaxed state without our cells shutting down.
Even in the kitchen, understanding acid-base reactions can help. Why does adding a pinch of baking soda (a base) calm down a bubbling tomato sauce that’s too acidic? Because the baking soda neutralizes the excess acid, and the resulting compounds are less reactive and less tart.
In essence, the conjugate base is the stable, often less reactive, product that results from an acid’s generosity. It’s the yin to the acid’s yang, the calm after the storm, the foundation upon which many chemical interactions are built.
A Moment of Reflection
In our fast-paced world, it's easy to feel like we're constantly giving, constantly donating our energy and our protons, so to speak. We juggle work, family, social lives, and personal passions, and sometimes we feel depleted. But just like these acids have their conjugate bases, we too have our moments of rest, our periods of replenishment, our "conjugate base" selves. It's in these quieter moments, when we're not actively giving, that we can recharge and prepare for our next contribution. So, the next time you’re feeling a bit drained, remember the chemistry of it all. Allow yourself to be the conjugate base for a while – it’s not a sign of weakness, but a crucial part of the cycle, essential for continued vibrancy and strength. It's the natural, easy-going rhythm of existence, both in our test tubes and in our lives.
