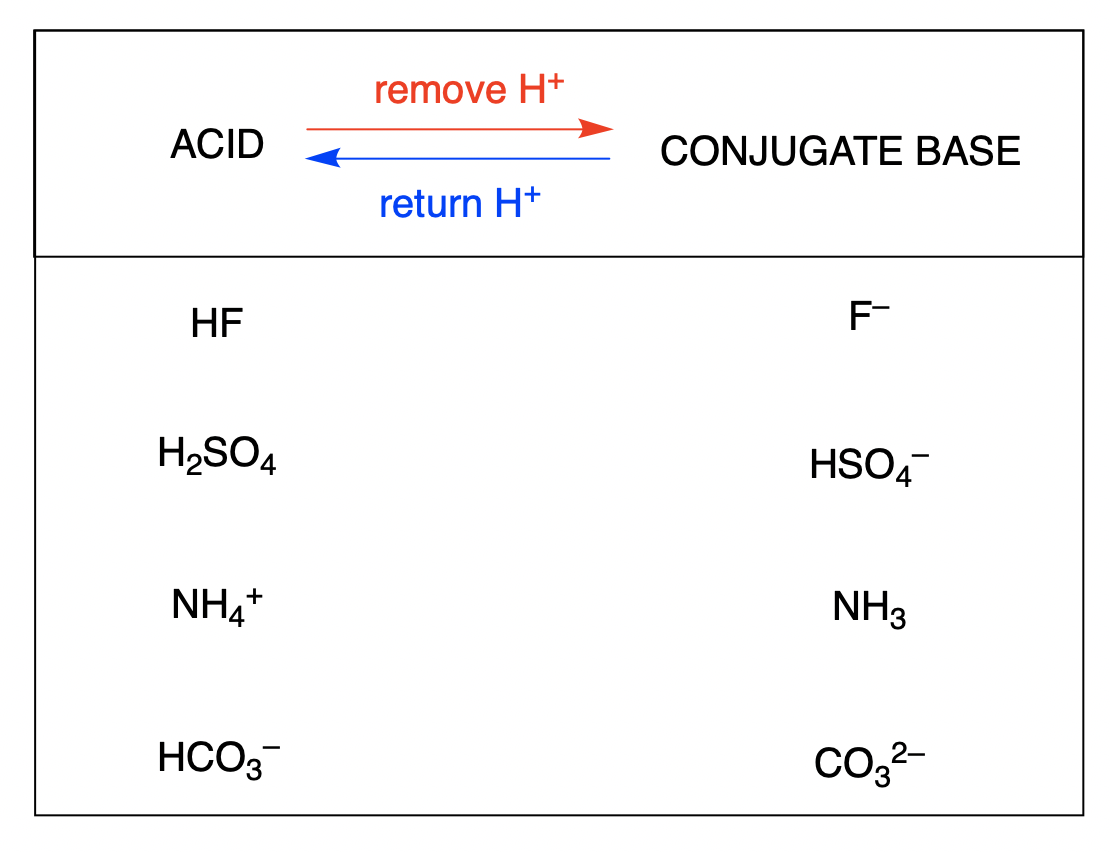
Write The Formula For The Conjugate Acid Of Each Base
Ever thought about the secret lives of molecules? It turns out, even the most ordinary-looking ones have fascinating relationships, kind of like a quirky family tree! Today, we’re going to peek behind the curtain and see how some of these molecular buddies help each other out.
Think of it like this: when a molecule is feeling a bit down, needing a boost or a friendly handshake, its closest pal might step in. This pal is what we call its conjugate acid. It’s like a supportive friend who’s always there to lend a helping atom!
Now, the fun part is figuring out who these pals are. It's not rocket science, but it does involve a little bit of chemical matchmaking. We’re going to look at a few common molecules and see who their super supportive conjugate acid friends are.
The Humble Water: More Than Just a Drink!
Let's start with something we all know and love: water. H₂O, the stuff that makes up oceans, rivers, and your morning coffee. When water plays the role of a base (which it loves to do!), it’s like a gracious host ready to accept a guest.
So, what happens when this hospitable water molecule decides to welcome a little something extra? It picks up a tiny, positively charged particle, a little spark of goodness called a proton (which is just a hydrogen atom with a positive charge, H⁺).
And voilà! The conjugate acid of water is born. It’s now become hydronium, which has the formula H₃O⁺. Isn't that neat? Water, in its most common form, is also a great host for another H⁺.
Imagine water as a cozy little house with two hydrogen atoms and one oxygen atom. When it acts as a base, it opens its door and another hydrogen ion, a little traveler, knocks. The house welcomes it in, becoming a slightly bigger, slightly more charged dwelling!

Ammonia: The Busybody of the Molecular World
Next up, let’s meet ammonia. This molecule, with the formula NH₃, is like a super social butterfly in the chemical world. It's known for being a bit of a base, always ready to share its space.
Just like our friend water, ammonia also has a soft spot for accepting a proton (that H⁺ traveler). It’s like ammonia is always looking for someone to chat with and is happy to give them a temporary home.
When ammonia (NH₃) grabs a proton, it transforms into its conjugate acid. This new, slightly more complex molecule is called ammonium, and its formula is NH₄⁺. It’s like ammonia got a little friend attached to it, making it a slightly more crowded but equally friendly entity.
Think of ammonia as a busy kitchen with three hydrogen atoms buzzing around an nitrogen atom. When a proton arrives, it finds a spot, and suddenly, it’s a four-hydrogen kitchen! The kitchen is still functional, just a bit more bustling.
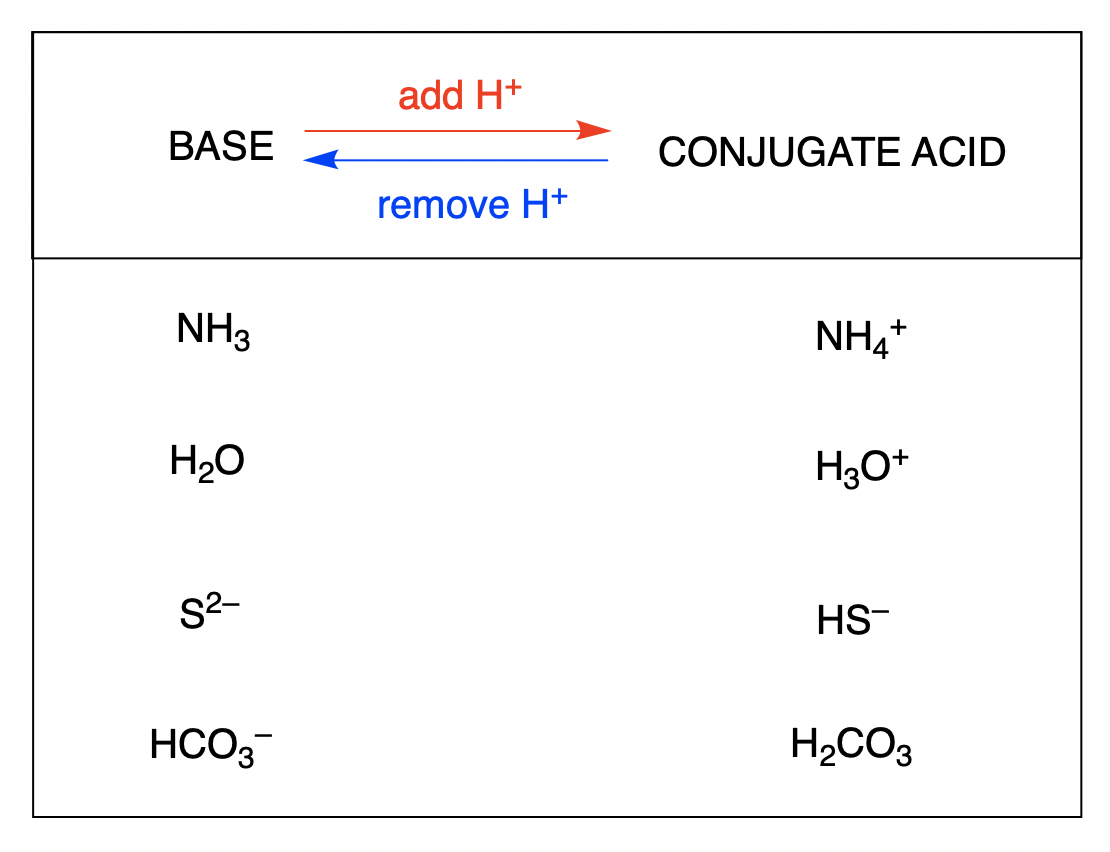
Hydroxide Ion: The Gentle Giant
Now, let's talk about the hydroxide ion. This one looks a little different, with the formula OH⁻. It's already carrying a bit of a negative vibe, which makes it a super strong base, eager to give away that extra bit of "stuff" it's holding.
But here's where the story gets interesting. When hydroxide wants to act like a base, it’s not looking to take a proton. Instead, it’s more like it’s passing on something it doesn’t need, making its conjugate acid actually a bit simpler!
So, when hydroxide ion (OH⁻) gives away its extra bit (effectively losing a proton from its perspective, even though it's not directly receiving one in this process), it leaves behind... you guessed it, water (H₂O)! This might seem backward, but in the world of acids and bases, sometimes the "acid" form is actually a simpler molecule.
It's like hydroxide is a balloon with a little extra air. To become more stable, it lets out some of that air. What's left is a perfectly deflated balloon, which in this case, is water! The formula for its conjugate acid is simply H₂O.
Chloride Ion: The Quiet Observer
Let's look at the chloride ion, Cl⁻. This one is often found hanging around in your salt shaker. It's a rather stable character, and when it decides to play the base role, it's looking for someone to hold onto.
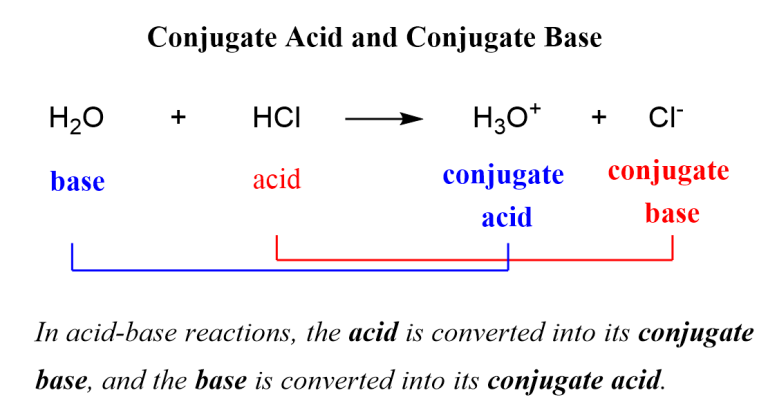
Much like our previous examples, the chloride ion also has a penchant for protons. It’s like it’s a bit shy and wants a proton to feel more complete and stable.
When chloride ion (Cl⁻) accepts a proton (H⁺), it forms its conjugate acid. This new molecule is hydrochloric acid, with the formula HCl. This is a famous acid, often found in our stomachs helping us digest our food.
Think of chloride as a single sock. It feels a bit incomplete. When it finds its matching sock, a proton, they become a pair, a cozy outfit of hydrochloric acid.
Carbonate Ion: The Versatile Player
Finally, let's explore the carbonate ion, CO₃²⁻. This one has a bit more negative charge to play with, making it a strong contender for accepting protons. It’s like it’s got plenty of room for guests!

When the carbonate ion acts as a base, it can actually accept not one, but two protons! This makes its conjugate acid formation a bit more layered.
First, it accepts one proton (H⁺) to become bicarbonate (or hydrogen carbonate), with the formula HCO₃⁻. This molecule is also quite common and plays important roles in our bodies.
But wait, there's more! If the bicarbonate molecule then decides to accept another proton, its final conjugate acid form is carbonic acid, with the formula H₂CO₃. It’s like a chain reaction of welcoming!
Imagine carbonate as a welcome mat with space for two people. The first person to arrive is a proton, making it bicarbonate. Then, another proton comes along and joins them, creating the friendly trio of carbonic acid.
So, the next time you encounter these molecules, remember their little secrets. They’re not just random collections of atoms; they’re characters in a never-ending story of chemical connection and support. Who knew chemistry could be so heartwarming?
