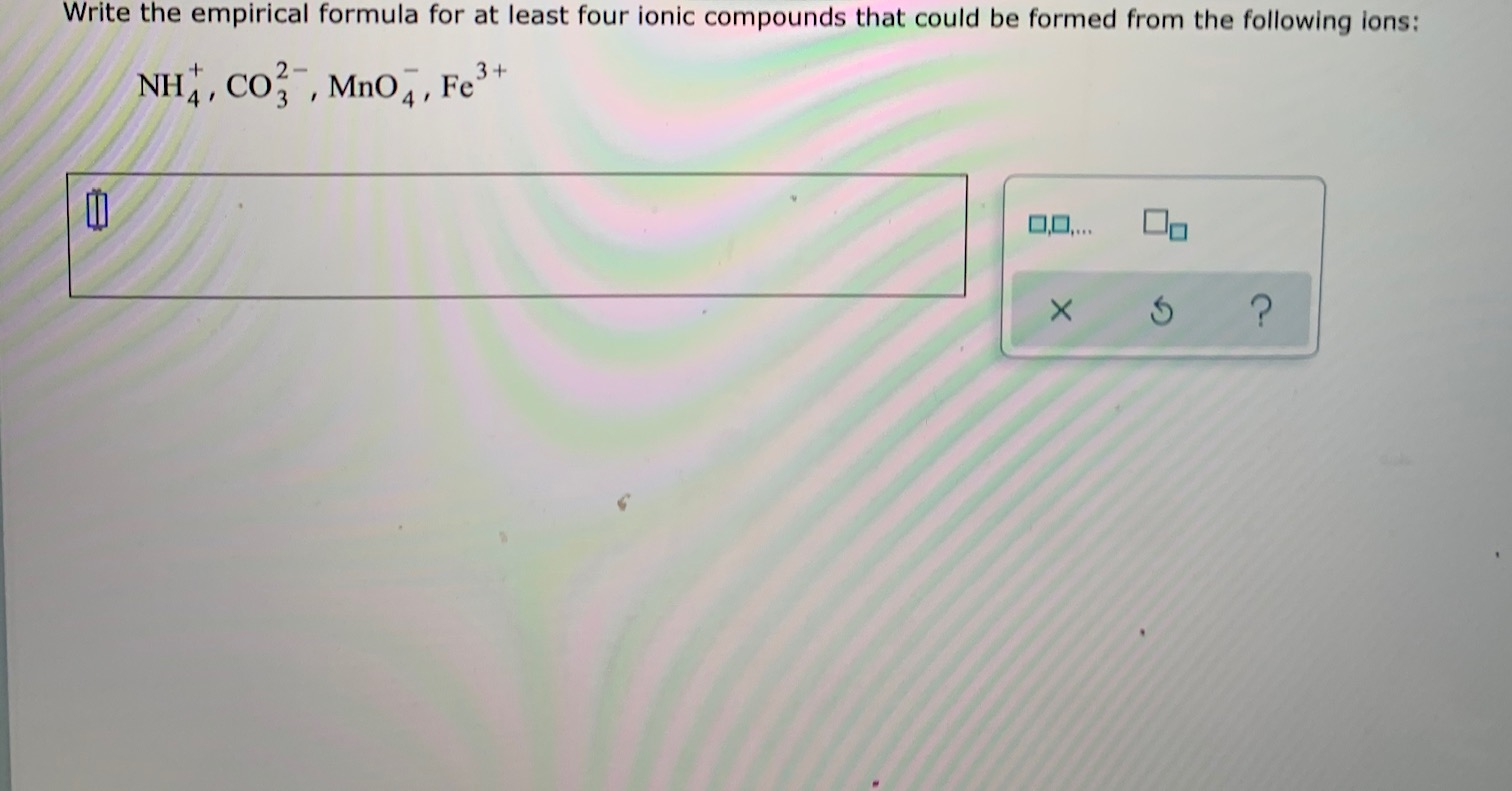
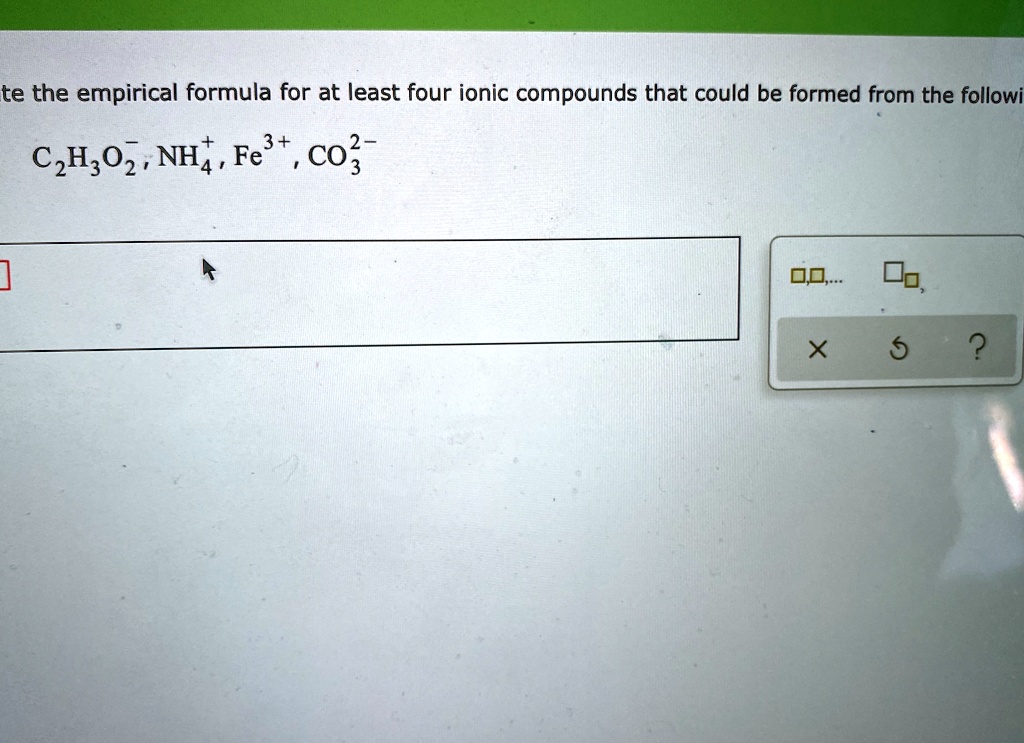
Write The Empirical Formula For At Least Four Ionic Compounds
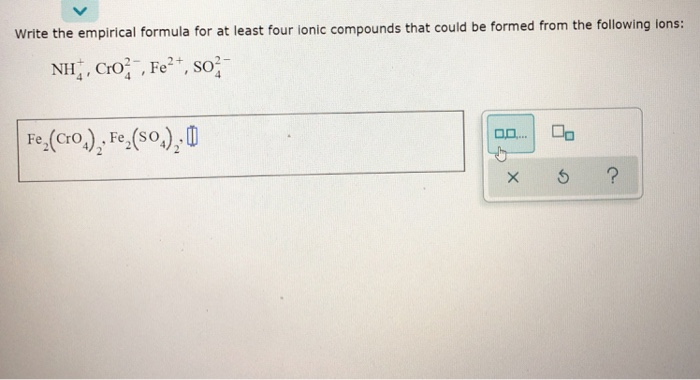
Ever looked at a salt shaker and wondered about the tiny, invisible building blocks that make up everything around us? It’s like a secret code! And guess what? Figuring out that code for certain chemical buddies is super fun. We’re talking about ionic compounds here, and their empirical formulas. Sounds fancy, right? But it’s actually like solving a little puzzle, and it’s way more entertaining than you might think.
Imagine you’ve got two different kinds of LEGO bricks, each with its own special way of sticking to the other. Ionic compounds are a bit like that. They’re made of two types of atoms: one that likes to give away electrons (think of them as friendly sharers!) and one that loves to take electrons (they’re the eager receivers!). When they get together, they form these amazing, strong bonds, creating all sorts of cool stuff. And the empirical formula? That's just the simplest, most basic ratio of these LEGO bricks in the whole structure. It’s like saying for every two red bricks, you always have one blue brick. Simple, yet powerful!
Let’s dive into some examples because, honestly, seeing it in action is the best part. Ready for some chemical celebrity sightings? First up, the classic sodium chloride. Yep, that’s your everyday table salt!
Sodium chloride. The OG of ionic compounds. NaCl.
How did it get that simple formula? Well, sodium (Na) loves to give away one electron, and chlorine (Cl) is thrilled to take one. So, they pair up in a perfect 1:1 ratio. Easy peasy! It’s like a perfectly matched dance pair, always two by two. And the result? The salt we sprinkle on our fries. How cool is that? From invisible atoms to a flavor enhancer. That’s the magic!
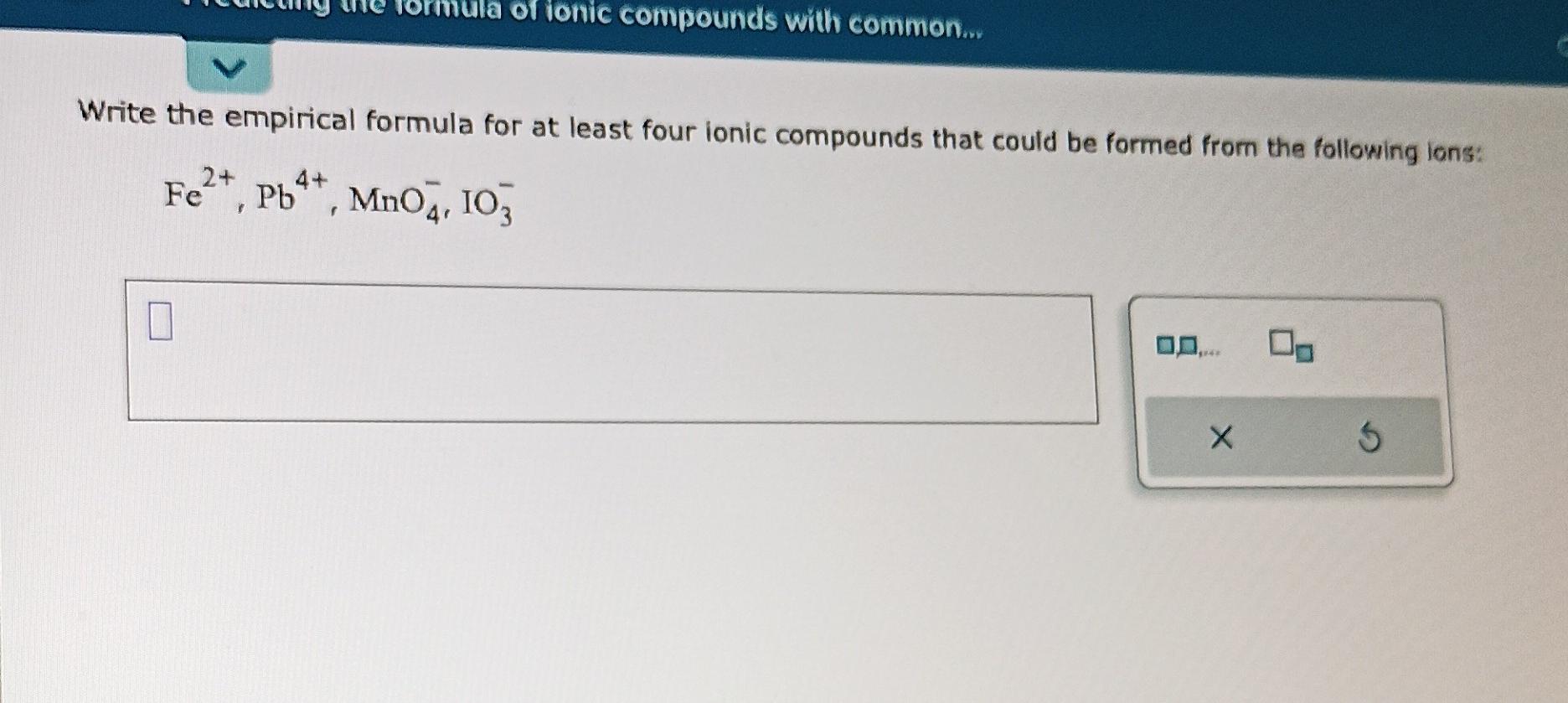
Next on our chemical adventure, let’s meet magnesium oxide. This one is a bit tougher, literally. It’s used in things like antacids and even in furnace linings because it can handle a lot of heat.
Magnesium oxide. Tough stuff. MgO.
Solved Write the empirical formula for at least four ionic | Chegg.com
Here, magnesium (Mg) is a bit of a generous soul, giving away two electrons. And oxygen (O) is super happy to accept those two. So, again, we get a neat 1:1 ratio. One magnesium atom is just the perfect partner for one oxygen atom. It's another straightforward recipe for a strong and useful compound. It shows us that even with different numbers of electrons being shared, the simplest ratio can still be one to one.
Now, let’s spice things up a bit with aluminum oxide. This compound is also incredibly strong and heat-resistant, used in abrasives and ceramics.
Aluminum oxide. Strong and dependable. Al₂O₃.
SOLVED: 'Help find this write instructions te the empirical formula for
This one is where the puzzle gets a tiny bit more intricate, but in a good way! Aluminum (Al) is eager to share three electrons. Oxygen (O), on the other hand, only wants to take two. So, how do they make it work? They have to find a common ground. For every two aluminum atoms giving away their electrons (that’s 2 x 3 = 6 electrons in total), three oxygen atoms can happily snatch them up (that’s 3 x 2 = 6 electrons in total). So, the simplest ratio you’ll find in a big chunk of aluminum oxide is two aluminums for every three oxygens. See? It's like a carefully balanced team, making sure everyone gets what they need. It’s a beautiful example of chemical harmony.
Let’s look at one more for good measure: calcium fluoride. You might know this one better as a component of toothpaste that helps strengthen your teeth!
Calcium fluoride. Tooth protector. CaF₂.
Here’s another fun one. Calcium (Ca) is ready to give away two electrons. Fluorine (F), however, is only looking to take one. So, what happens? One calcium atom is so generous that it needs two fluorine atoms to happily accept its extra electrons. Each fluorine atom takes one, and the calcium atom is perfectly satisfied having given away its two. This results in a ratio of one calcium atom for every two fluorine atoms. It’s a clear illustration of how atoms combine to achieve stability, and the formula CaF₂ tells us this story in the most concise way possible. Isn't that neat? A simple formula telling a tale of electron sharing and atomic teamwork.
The reason this is so engaging is that it’s not just about memorizing letters and numbers. It’s about understanding the fundamental rules of how the universe puts itself together. Every time you see these compounds, you’re looking at a tiny, perfect ratio that explains their existence. It's like cracking a secret code that nature uses everywhere. From the salt on your table to the materials that build our world, these simple empirical formulas are the keys to understanding it all. It's a journey into the microscopic world, and it’s surprisingly delightful!