Write The Electron Configuration For A Neutral Atom Of Tin

Ah, Tin. That shiny, silvery metal. You know, the stuff from which we used to make those adorable little toy soldiers. Or maybe you’ve got a few old tin cans lying around. Seems pretty straightforward, right? Just a metal. Easy peasy.
But then, someone, somewhere, decides it’s a good idea to talk about its electron configuration. And suddenly, what seemed like a simple metal transforms into something that looks like a secret code. It’s like your friendly neighborhood superhero suddenly starts speaking fluent Klingon.
Let’s just get this out of the way, because I have an unpopular opinion. I think writing out the full electron configuration for an element like Tin should come with a warning label. Like, “Proceed with caution. May induce mild existential dread or a sudden urge to rewatch cartoons.” Seriously, it’s a marathon, not a sprint.
But hey, we’re here to talk about it. So, let’s dive in. For a neutral atom of Tin, we’re essentially trying to figure out where all its tiny, zippy electrons like to hang out. Think of it like a cosmic apartment building. Each floor and each room has a specific capacity and energy level. Electrons are the tenants.
We start from the lowest energy levels, the basement apartments, if you will. These are the 1s orbitals. They can hold up to two electrons. So, we fill those up first. Then we move to the next floor, the 2s orbitals. Again, two little electrons squeeze in there. It’s getting crowded, but they’re polite.

Next up are the 2p orbitals. These guys are a bit more spacious, offering three rooms. Each room can hold two electrons, so the 2p orbitals can house a total of six electrons. It’s like a mini-suite on the second floor. Then we’re onto the 3s, and yep, you guessed it, two more electrons. They’re really just following the rules, aren’t they?
After that, we hit the 3p orbitals. More room here, six electrons. These electrons are starting to explore the building quite a bit now. It’s like they’ve got a whole wing of the second and third floors to themselves. This is where things start to get a little more interesting, but still pretty predictable.
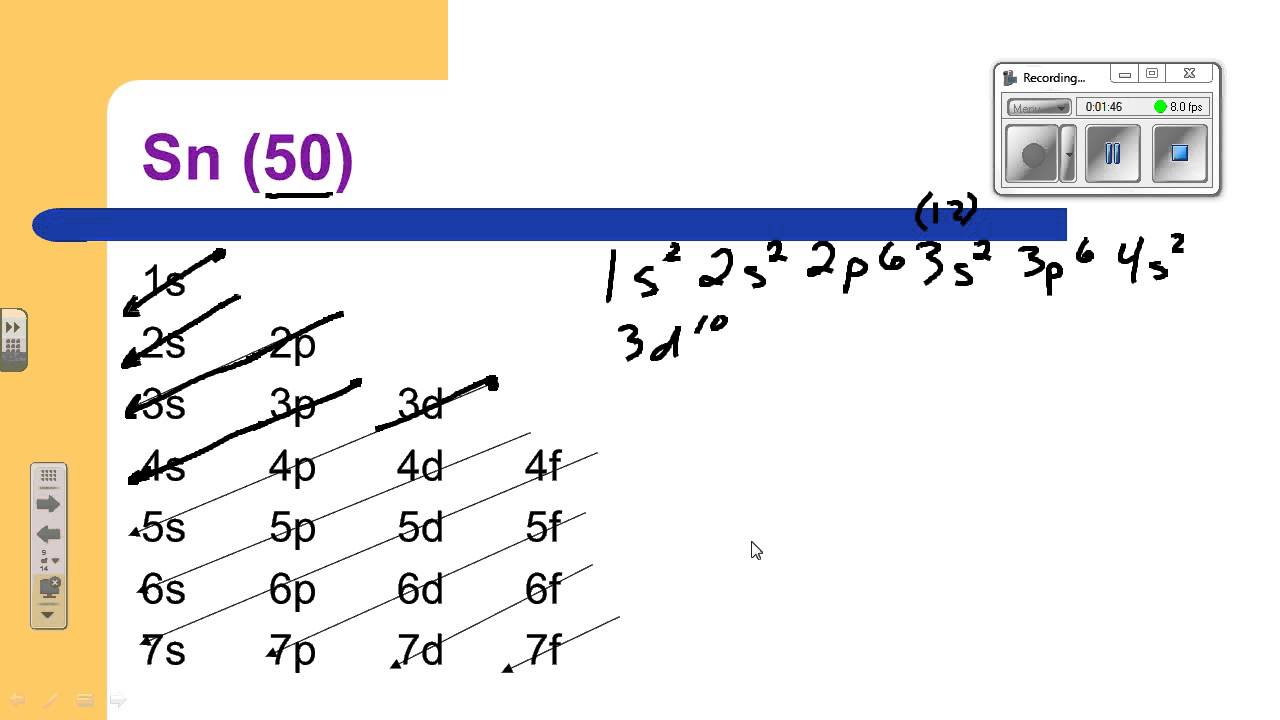
Then comes the 4s. Two electrons. Simple enough. But then, we have the 3d orbitals. These are like the premium, slightly more secluded apartments. They have five rooms and can hold a whopping ten electrons. So, we fill all of those up. Imagine a whole floor dedicated to these guys, tucked away a bit from the main hustle and bustle.
Now, we’re moving into the fourth floor. We have the 4p orbitals. Six more electrons. They’re settling in nicely. But the real adventure, or perhaps the real test of patience, begins with the 5s orbitals. Two electrons in. They’re on the fifth floor now, feeling quite elevated.
And then… the big one. The 4d orbitals. These are like the penthouse suites of the fourth floor. They have five rooms, and they can hold ten electrons. So, we meticulously fill all ten of those. This is where things start to feel… extensive. It’s like the building has expanded, and there are new wings being built for the electrons to inhabit.

But we’re not done! Oh no. We still have the 5p orbitals. These get six electrons. And finally, we reach the outermost shell, the highest energy level, the very top floor with the best view: the 5s. Wait, I think I might have looped back. No, no, the actual outermost shell for Tin is where things get really specific. We are talking about the electrons that are most easily involved in chemical reactions.
For Tin, which has 50 electrons in total (that’s its atomic number, the number of protons it has, and for a neutral atom, the number of electrons matches), after all the filling up we’ve done, we end up with the 5p orbitals getting two electrons. So, the last bits of electrons settle into the 5p orbitals.
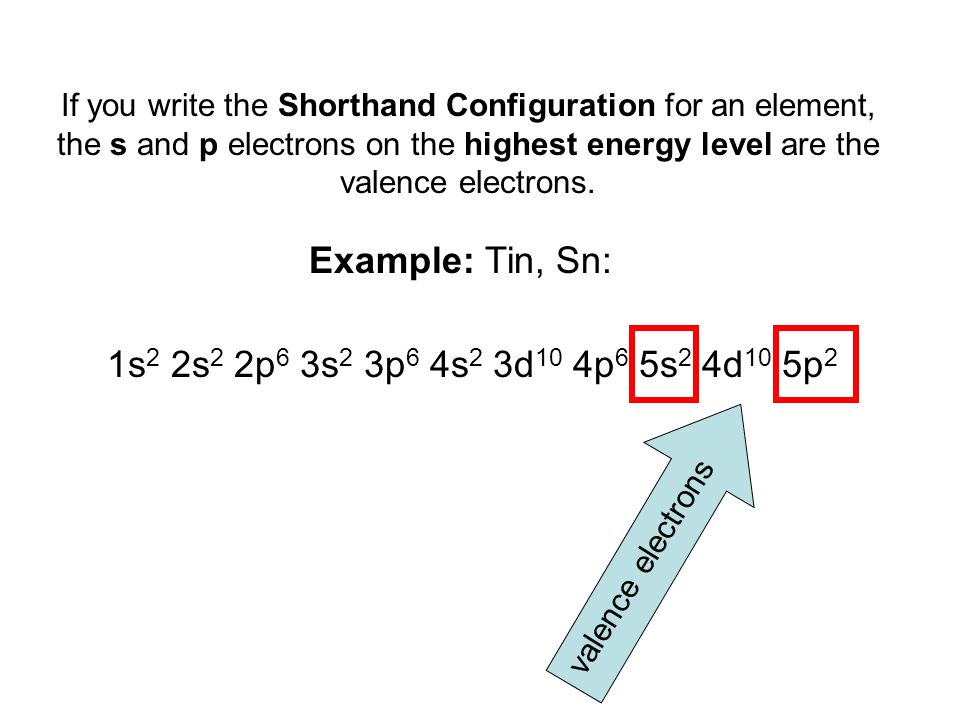
So, putting it all together, for a neutral atom of Tin, the electron configuration is: 1s² 2s² 2p⁶ 3s² 3p⁶ 4s² 3d¹⁰ 4p⁶ 5s² 4d¹⁰ 5p².
Take a moment. Let that sink in. It’s a lot of numbers and letters, isn't it? It’s like a recipe for a very, very complex cake. And the worst part? Sometimes you don’t even need the entire recipe! You can use this fancy thing called noble gas notation to shorten it. It’s like saying, “Yeah, I made that whole building, but let’s just focus on the top three floors for now.” It saves a lot of ink, and frankly, a lot of mental energy.
But for today, we’ve tackled the full, unadulterated, slightly intimidating, but ultimately fascinating electron configuration for Tin. And if you’re still with me, congratulations. You’ve earned an imaginary cookie. Or perhaps a small piece of tin foil to admire its simple, non-confusing outer beauty.
