Write The Electron Configuration For A Neutral Atom Of Tellurium.

Imagine the universe as a giant, bustling city, and within this city, every atom is like a tiny apartment building. These buildings have floors, and on each floor, there are rooms. Now, the electrons are the super energetic residents of these apartment buildings. They just love to zip around, filling up the rooms and floors in a very specific order. It’s not quite as chaotic as it might seem; there’s a method to their madness, a quirky architectural plan that every element follows.
Today, we're going to peek into the apartment building of a rather interesting character: Tellurium. Now, Tellurium might not be a household name like Oxygen or Gold, but it's got its own unique charm. Think of it as a slightly eccentric but ultimately very helpful neighbor. This neutral atom of Tellurium is like a well-organized individual, making sure all its electron residents have a cozy spot to call home, without any extra baggage (because it’s neutral, you see – no protons or electrons are feeling left out or over-represented).
So, how do we figure out where all these little electron residents will settle in Tellurium’s building? Well, it’s like following a set of very old, very specific house rules. These rules dictate which floors get filled first, and which rooms on those floors are the most popular. Think of it as a game of musical chairs, but instead of chairs, we have orbitals, and instead of music, we have energy levels. The electrons, being good little followers, go to the lowest energy spots first – the basement, if you will, of their atomic apartment.
Let's start with the absolute lowest floor. This is the first energy level, and it’s a rather small one, only big enough for two residents. We call this the '1s' orbital. So, our first two electron residents happily move into the 1s. They’re like the pioneers, claiming the very first available space.
Next up, we move to the second floor. This floor has a bit more space. It has an 's' room again, and this one can also hold two residents. So, two more electrons move into the 2s. But wait, there’s more! On this second floor, there are also three 'p' rooms. These 'p' rooms are a bit more spacious and can accommodate a total of six residents. So, the next six electrons fill up the 2p orbitals. It's getting a bit crowded on the second floor, but everyone’s still getting along famously.
Now, we ascend to the third floor. This floor is getting quite grand! It has another 's' room, the 3s, which takes in two more electrons. Then comes the 3p section, which, just like on the second floor, can hold up to six electrons. So, another six electron residents are settling in here. But the third floor has a secret, a whole new wing of rooms! These are the 'd' orbitals, and they’re quite capacious, able to house a whopping ten electrons. So, ten more electrons move into the 3d orbitals. By this point, Tellurium’s apartment building is looking pretty full on the lower levels!
We're not done yet! We need to climb to the fourth floor. This is where things get really interesting. We have the familiar 4s orbital, which takes in two more electrons. Now, here's where the rulebook gets a little playful. After the 4s is filled, the electrons don't immediately jump into the 4p rooms. Instead, they’re drawn to the 3d orbitals, which we’ve already filled. But in Tellurium's case, we still have more electrons to place! So, after the 4s, the next electrons actually go into the 3d orbitals that we already thought were full. This might seem odd, like someone showing up to a party after the main event, but it’s all about energy! The 3d orbitals, even though they’re on a lower floor, are energetically favorable for these next electrons to join.

Let's backtrack a tiny bit to clarify the order. After the 3p orbitals are filled, the electrons actually go to the 4s first because it’s a lower energy level, even though it’s on a higher floor. So, two electrons go into 4s. Then, the next electrons fill up the 3d orbitals, and all ten of those spots get taken. It's like a surprise guest list where some people are seated at a slightly less prominent table, but it’s still a great spot!
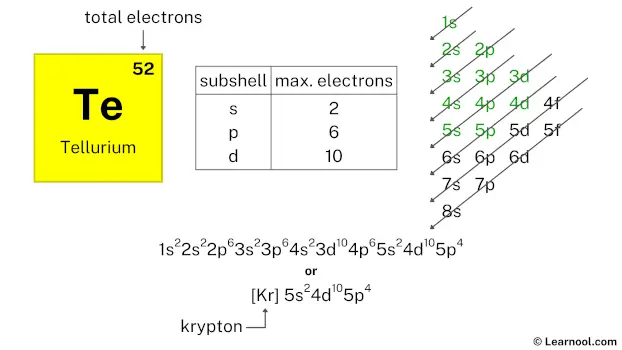
Now, back to the fourth floor. After the 3d orbitals are all snugly filled, it's time for the 4p orbitals. These 'p' orbitals, similar to their lower-floor cousins, can hold six electrons. So, the last set of electron residents for a neutral Tellurium atom will fill up these 4p orbitals. We need to count how many electrons we've placed so far to know exactly how many go into the 4p. Tellurium has 52 protons, and since it's neutral, it has 52 electrons. Let's tally: 2 (in 1s) + 2 (in 2s) + 6 (in 2p) + 2 (in 3s) + 6 (in 3p) + 10 (in 3d) + 2 (in 4s) = 30 electrons. We still have 52 - 30 = 22 electrons to place. Ah, but wait! The 3d can only hold 10 electrons. Let me re-tally. 2 (1s) + 2 (2s) + 6 (2p) + 2 (3s) + 6 (3p) + 10 (3d) + 2 (4s) = 30 electrons. We need 52 electrons. So, after 4s, we go to 3d which takes 10. That's 30. Then we go to 4p. So, we have 52 - 30 = 22 electrons to place. Hmm, this isn’t adding up. Let me consult my trusty elemental encyclopedia! Ah, yes! The order is a bit like a spiral staircase. It’s 1s, then 2s, 2p, 3s, 3p, 4s, 3d, 4p, 5s, 4d, 5p, and so on. So, for Tellurium, which has 52 electrons:
The 1s orbital gets 2 electrons.
The 2s orbital gets 2 electrons.

The 2p orbitals get 6 electrons.
The 3s orbital gets 2 electrons.
The 3p orbitals get 6 electrons.
The 4s orbital gets 2 electrons.
The 3d orbitals get 10 electrons.
So far, that's 2 + 2 + 6 + 2 + 6 + 2 + 10 = 30 electrons. We have 52 - 30 = 22 electrons left.
Now, the next energy level to fill after 3d is the 4p. And the 4p orbitals can hold a maximum of 6 electrons. So, 6 electrons go into the 4p orbitals. This brings our total to 30 + 6 = 36 electrons. We have 52 - 36 = 16 electrons remaining.
After the 4p, the next orbitals to fill are the 5s. The 5s orbital gets 2 electrons. Now we have 36 + 2 = 38 electrons placed. We have 52 - 38 = 14 electrons left.

The next orbitals to fill are the 4d orbitals. These can hold up to 10 electrons. So, the 4d orbitals get 10 electrons. This brings our total to 38 + 10 = 48 electrons. We have 52 - 48 = 4 electrons left.
Finally, the last electrons go into the 5p orbitals. The 5p orbitals can hold up to 6 electrons, but we only have 4 left. So, the 5p orbitals get the remaining 4 electrons.
So, the complete electron configuration for a neutral atom of Tellurium, with all its electrons happily housed, is:
1s2 2s2 2p6 3s2 3p6 4s2 3d10 4p6 5s2 4d10 5p4
It's like a beautifully orchestrated symphony of electrons, each playing its part in the grand design of the Tellurium atom. And the best part? This intricate arrangement is what gives Tellurium its unique personality, allowing it to interact with other elements in fascinating ways, perhaps even contributing to the making of things we use every day, all thanks to its tidy little electron residents!
