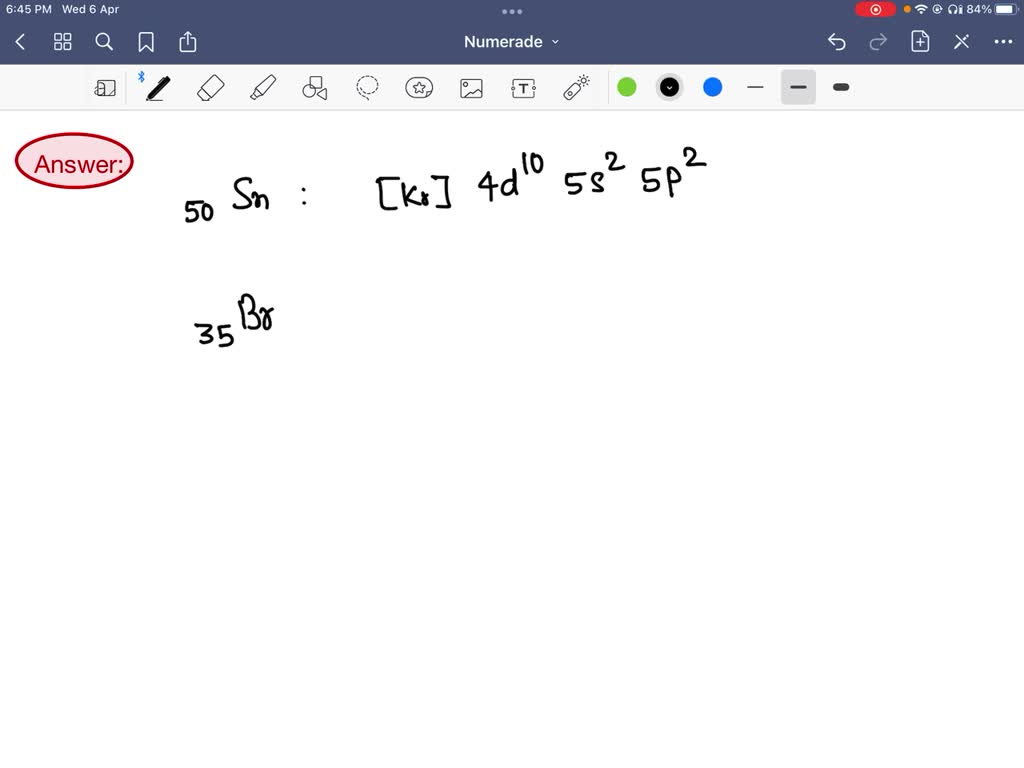
Write The Condensed Electron Configurations For The Sn Atom.
Hey there, science adventurers! Ever feel like you’re trying to describe a whole city by listing every single house, street, and mailbox? That’s kind of what it’s like describing where all the tiny little electrons hang out in an atom. They’re like the super-energetic kids at a giant playground, zipping around everywhere! But fear not, because we’ve got a secret weapon, a shortcut, a… well, a super-cool abbreviation that makes things way easier!
Today, we’re going to peek into the world of the element Tin, or Sn as the cool kids in chemistry call it. Now, tin is pretty neat. It’s that shiny stuff that used to coat our tin cans (remember those?), and it’s even used in things like solder to join wires together. It’s a hardworking element, and like any hardworking star, it’s got a whole entourage of electrons buzzing around its nucleus. If we were to write out the full address for every single one of those electrons, it would be like writing a novel! We’d be talking about shells, and sub-shells, and individual orbitals… honestly, it would make your eyes glaze over faster than a poorly glazed donut.
But here’s where the magic happens. Scientists, bless their brilliant brains, figured out a way to make it super snappy. They realized that most atoms have a core group of electrons that are basically just hanging out, chilling in a very predictable pattern. Think of it like the "regular guests" at a party. They always show up, they always occupy the same space, and for the most part, they’re not the ones causing all the excitement. These inner electrons are so consistent, in fact, that we can just say, "Oh, yeah, they've got the same electron setup as the element Krypton!"
Krypton (Kr)! Now, that’s a name that probably sparks some sci-fi vibes, right? Imagining Superman’s weakness, or maybe a super-strong gas. Well, in the atom world, Krypton is like the ultimate "base model" electron configuration. It’s a noble gas, meaning it’s super stable and doesn’t like to get into chemical drama. It’s the chillest atom on the block when it comes to its electron setup.
So, instead of listing out all the electrons that Krypton has (and trust me, that’s still a good chunk of them!), we just say, "Okay, Tin has all of Krypton’s electrons, plus a few extra ones running around and causing all the fun!" This is our condensed electron configuration, and it’s a total game-changer. It’s like having a VIP pass that lets you skip the long line and get straight to the important stuff.

For our friend Tin (Sn), this condensed electron configuration looks like this:
[Kr] 4d10 5s2 5p2
See that? We start with [Kr]. That’s our nod to Krypton, our shorthand for all those inner electrons. We’re saying, "Everything Krypton has, Tin has too!" And then we add the exciting bits, the electrons that make Tin uniquely… well, Tin! We've got some in the 4d sub-shell, some in the 5s, and a couple in the 5p. These are the electrons on the outermost "floors" of the atom's electron "hotel," and they're the ones that get to interact with other atoms and make chemical reactions happen. They’re the life of the party, the ones getting all the attention!
It’s like if you’re describing your amazing party. Instead of listing every single person who RSVPed and is just milling around the snack table, you can say, "Well, Sarah and her whole crew from accounting are here (that’s our [Kr] part – the reliable crowd), and then we’ve got the main performers: the rock band, the DJ, and the acrobatic dancers!” These are your special guests, the ones bringing the real entertainment, just like those outer electrons in Tin.
This condensed notation makes our lives so much simpler. It allows us to focus on what makes each element unique without getting bogged down in the details of the core. It's the difference between a grocery list with every single grain of rice and just saying "rice for the stir-fry." You get the point, and you can move on to the more interesting ingredients!
So, next time you hear about electron configurations, remember our friend Tin (Sn) and its super-efficient, super-cool condensed electron configuration. It’s a testament to how chemists have found clever ways to simplify complex ideas, making the universe of atoms just a little bit more approachable and a whole lot more fun!
