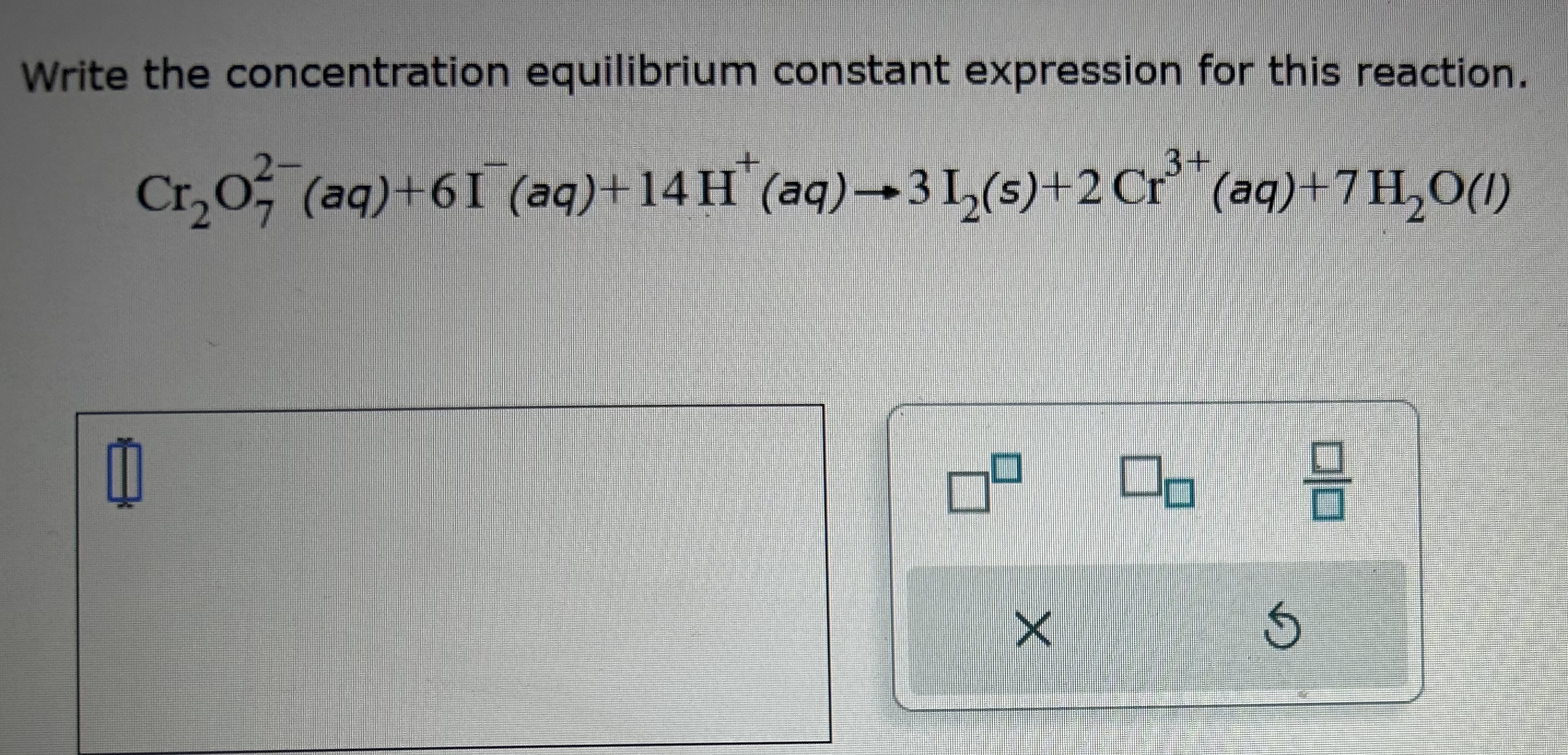
Write The Concentration Equilibrium Constant Expression For This Reaction
Ever found yourself staring at a recipe, a paint swatch, or even the perfect shade of lipstick and thinking, "How do they get it that way?" Well, you're not alone! That little spark of curiosity, that desire to understand the "why" and "how" behind the things we interact with every day, is what makes exploring topics like chemical equilibrium so satisfying. It's like unlocking a secret language that governs so much of our world, from the browning of an apple to the way our bodies function.
But what exactly is this equilibrium thing, and why should you care? Think of it as a state of balance, a point where two opposing processes are happening at the same rate, so it looks like nothing is changing. In chemistry, this often involves reactions that can go both forward and backward. For instance, when you dissolve sugar in your tea, it dissolves, but some sugar molecules also re-crystallize out. Equilibrium is reached when the rate of dissolving equals the rate of re-crystallization. This concept is incredibly useful because it helps us predict and control chemical reactions. Understanding equilibrium allows scientists and engineers to design more efficient industrial processes, develop better medicines, and even create materials with specific properties. It’s the silent architect behind much of modern technology and comfort.
The beauty of equilibrium is that it’s everywhere! Consider the water cycle. Evaporation and condensation are opposing processes that reach equilibrium within a closed system, influencing weather patterns. In our kitchens, the way food cooks is a dance of chemical reactions, many of which are influenced by equilibrium. Even the air we breathe relies on the equilibrium between oxygen and carbon dioxide in our atmosphere. More practically, think about the production of ammonia, a key ingredient in fertilizers. The Haber-Bosch process, which synthesizes ammonia from nitrogen and hydrogen, is a prime example of applied chemical equilibrium. By understanding and manipulating the equilibrium, we can produce enough ammonia to feed the world!
So, how can you get more enjoyment out of this fascinating concept? Firstly, start simple. Don't dive into complex equations right away. Look for everyday examples. Observe how a glass of iced tea reaches a point where it stops sweating, or how a saturated salt solution has undissolved salt at the bottom. Secondly, visualize it. Imagine tiny particles moving back and forth. Think of a tug-of-war where both sides are pulling with equal force. Thirdly, if you’re feeling adventurous, explore the concentration equilibrium constant expression. This is a mathematical way to describe the ratio of products to reactants at equilibrium. For a general reversible reaction like aA + bB ⇌ cC + dD, the expression is Kc = ([C]c[D]d) / ([A]a[B]b). Don't let the symbols intimidate you! It simply tells us whether the reaction favors products or reactants at equilibrium. The higher the Kc value, the more products are formed. Finally, connect it to your interests. Are you into baking? Equilibrium plays a role in how dough rises and how cakes set. Are you into gardening? Fertilizer production is all about equilibrium. The more you see it in action, the more you’ll appreciate its fundamental importance.
