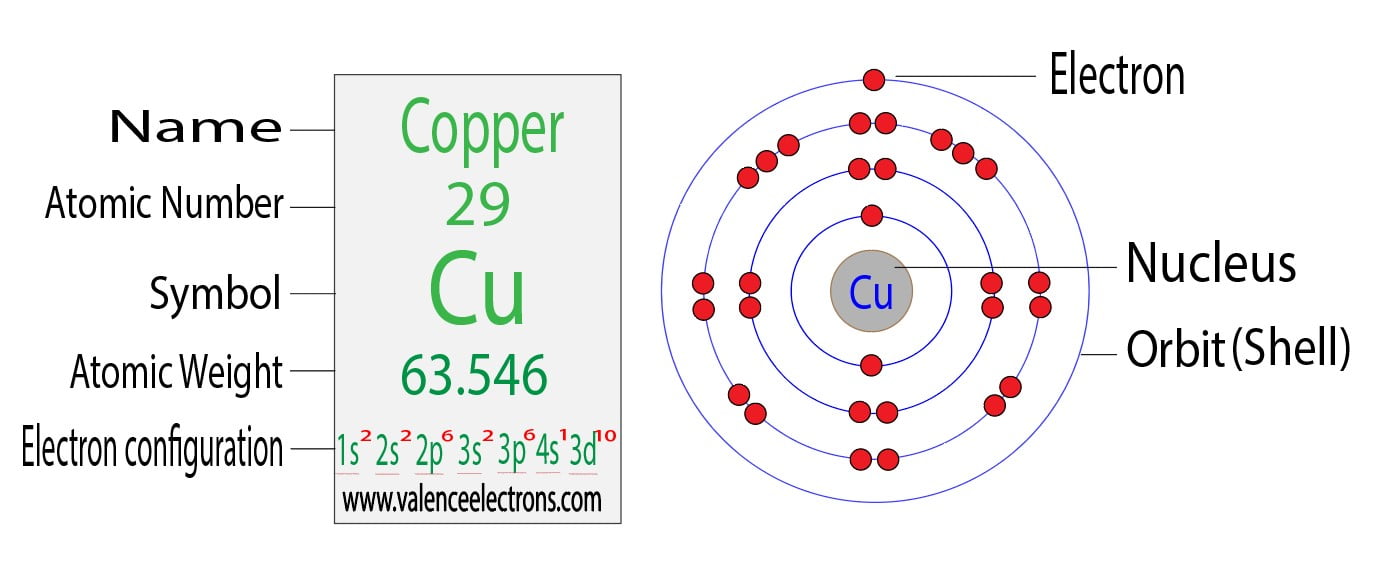
Write The Complete Electron Configuration For The Copper Atom
So, you’ve probably seen copper all around you, right? It’s that lovely reddish-brown metal that makes up the coins in your pocket, the pipes in your house, and the shiny bits on your favorite guitar. But have you ever wondered what makes copper, well, copper? It all comes down to its tiny, bustling inhabitants: electrons. Think of them like little energetic sprites zipping around the center of the atom, each with its own assigned ‘room’ to hang out in. And today, we’re going to peek into the super-organized, slightly quirky world of the copper atom’s electron neighborhood.
Imagine an atom as a miniature city. At its heart is the nucleus, kind of like the town hall. And orbiting this town hall are the electrons, each one a resident with a specific address. These addresses aren’t just random; they follow a set of rules, like a cosmic apartment complex manager has them neatly filed. Each electron has a specific energy level and a specific shape of ‘room’ it prefers. We can describe where all the electrons are chilling by writing out their complete electron configuration. It's like a detailed list of who lives where in the copper atom's apartment building.
Now, copper is a bit of a special case. It’s element number 29 on the periodic table, which means it has 29 protons in its nucleus and, when it’s neutral, 29 electrons buzzing around. If we were to just fill up the electron ‘rooms’ in a straightforward, by-the-book way, you might expect a certain arrangement. But copper, being the slightly rebellious but ultimately charming element it is, likes to do things a little differently. It’s like that friend who has a perfectly good desk but always chooses to do their homework on the floor because they find it more comfortable. Copper’s electrons have a similar kind of preference.
The basic building blocks for these electron ‘rooms’ are called orbitals. You can think of them as different types of apartments. There are s orbitals, which are like cozy, spherical studios. Then there are p orbitals, which are a bit more elongated, like a small one-bedroom. And then there are the even fancier, more complex d orbitals, which are like spacious, multi-room suites with all sorts of nooks and crannies. Each of these orbital types can hold a certain number of electrons, just like each apartment has a set capacity.
So, let’s start filling these rooms for our copper atom. We begin with the lowest energy levels, the ones closest to the nucleus, where it’s nice and quiet. The first ‘floor’ has a single s orbital, called 1s. It’s like the smallest, most basic studio apartment, and it can hold up to 2 electrons. So, we fill that up: 1s2. That’s our first two residents happily settled.

Next up is the second ‘floor,’ or the second energy level. This level has another s orbital (2s) and three p orbitals (2p). The 2s orbital is another studio, so it takes 2 electrons: 2s2. Then we have the three 2p orbitals. These are like three adjoining studios, and each can hold 2 electrons, making a total of 6 electrons for the p set. So, we fill those up too: 2p6. Now we’ve got 10 electrons housed.
Moving on to the third ‘floor,’ the third energy level. This one is quite expansive. It has a 3s orbital (taking 2 electrons: 3s2), three 3p orbitals (taking 6 electrons: 3p6), and then things get interesting with the 3d orbitals. These are the fancy, multi-room suites. There are five 3d orbitals, and each can hold 2 electrons, for a grand total of 10 electrons. So, we fill those up: 3d10. By this point, we've accounted for a whopping 28 electrons!

We’re almost there! We’ve got one electron left to place. According to the basic rules, it would likely go into the next available energy level and orbital, which is the fourth energy level. This level starts with a 4s orbital, which is another spherical studio. So, you might think, "Easy peasy, the last electron goes into 4s, making it 4s1." And for many elements, that would be the end of the story. But here’s where copper throws us a delightful curveball.
Copper is a bit of a minimalist when it comes to its outer shell, but it really likes its d orbitals to be completely full. Having a completely full set of d orbitals, like our 3d10, makes an atom feel particularly stable and content, like a perfectly organized closet. Similarly, having a half-filled set of orbitals also offers a nice, balanced feeling of stability. So, copper, in its wisdom, decides to rearrange things slightly. Instead of leaving the 4s orbital with just one electron and the 3d orbitals completely full, it nudges one of the electrons from the 4s orbital into the 3d orbital.

This means the final configuration looks like this: 1s2 2s2 2p6 3s2 3p6 3d10 4s1. It’s a subtle change, a little dance of electrons, but it’s what gives copper its unique properties. It’s like if you were packing for a trip, and instead of putting the last item in your suitcase, you decided to move something from your carry-on to your main bag to make both feel a little more evenly weighted and secure. Copper’s electrons are just looking for that sweet spot of stability.
So, the next time you see that shiny copper penny or feel the warmth of a copper pot, remember the intricate, almost personal arrangement of its electrons. It’s a tiny universe within, a testament to the elegant, and sometimes delightfully unexpected, rules that govern even the most ordinary of things. It’s the complete electron configuration for the copper atom: a story of electron neighborhoods, stability, and a little bit of elemental flair.
