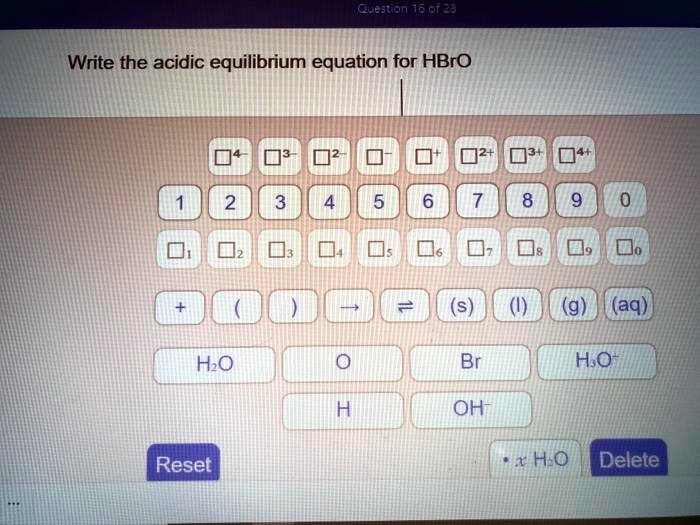
Write The Acidic Equilibrium Equation For Hbro

Hey there, science adventurers! Ever feel like some chemical reactions are just hiding in plain sight, playing a little game of give-and-take? Well, today we're going to peek behind the curtain of one such playful performer, and its name, drumroll please... is Hypobromous Acid!
Now, I know what you might be thinking. "Hypobromous Acid? That sounds super fancy and, dare I say it, a little intimidating!" But fear not, my curious comrades! Think of it like a shy friend who's great at parties but needs a little encouragement to join the dance.
Our star, HBrO, is a bit of a drama queen when it comes to its watery abode. It loves to be in water, but it's not always the most comfortable being all by itself. It's got this little habit of making friends with the water molecules, and sometimes, it even lets go of a tiny bit of itself!
Imagine you're at a playground, and you've got a really fun toy, say a super bouncy ball. You're happily bouncing it around, but sometimes, whoosh, the ball bounces just a little too high and lands somewhere else, leaving you with just a part of your original fun. That's kind of what HBrO does in water!
So, how do we capture this delightful little interaction in a scientific sentence? We use something called an acidic equilibrium equation. Don't let the big words scare you! It's just a fancy way of drawing a picture of what's happening when HBrO decides to share its space and its bits with water.
First off, we need our main character, Hypobromous Acid. We represent it with its chemical shorthand: HBrO. This is like its official name tag.
Now, where does the magic happen? In water, of course! So, we add our HBrO to some H₂O. Think of the water as the stage where all the action unfolds.
Here's where the fun begins! HBrO, in its moment of playful generosity, decides to dissociate. This just means it breaks apart a little. It's like when you're sharing cookies with friends, and you break off a piece for them.
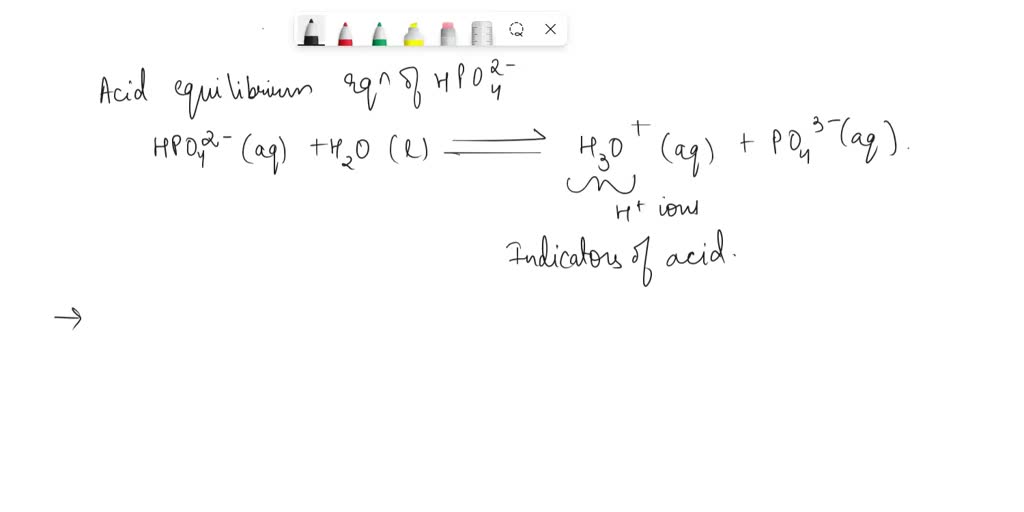
When HBrO breaks apart, it gives away a hydrogen ion. We scientists have a special symbol for this hydrogen ion: H⁺. This is like the tiny, energetic spark that flies off.
And what's left behind? When HBrO loses its H⁺, it becomes the hypobromite ion. Its chemical shorthand is BrO⁻. This is the remaining part of our bouncy ball, still pretty cool and ready for more adventures.
So, on one side of our equation, we have our starting materials: HBrO and H₂O. They're like the audience before the show begins.
On the other side, we have our performers after the dissociation: the tiny but mighty H⁺ and the graceful BrO⁻. They're the stars of the show, doing their energetic dance.
Now, here's the really cool part: this isn't a one-way street! It's not like HBrO just breaks apart and then that's it. Oh no, these molecules are social butterflies! The H⁺ and BrO⁻ can also decide to get back together and reform HBrO.
This back-and-forth action is what we call an equilibrium. It's like a game of tag where everyone is both "it" and "not it" at the same time. The reaction is happening in both directions, like a lively conversation.
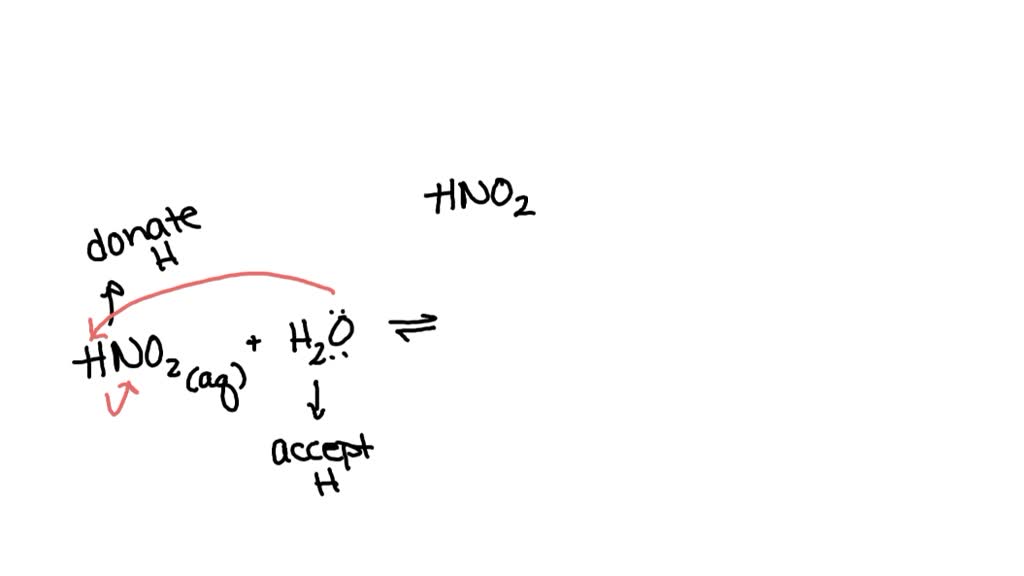
To show this two-way street, we use special arrows in our equation. Instead of a simple arrow pointing in one direction, we use double arrows. They look like little arrows pointing at each other, saying "and back again!"
So, let's put it all together! On the left side, we have our reactants: HBrO (the shy star) and H₂O (the stage). We write it as:
HBrO (aq) + H₂O (l)
The (aq) means it's dissolved in water, like a solid gem sparkling in a liquid crystal. The (l) means water itself is in its liquid form, just chilling.
Then, we have our magnificent double arrows, signifying that glorious dance of forward and backward action. It looks like this:
⇌
And finally, on the right side, we have our products, the wonderfully separated H⁺ (the energetic spark) and BrO⁻ (the remaining part of the fun). We write it as:
H₃O⁺ (aq) + BrO⁻ (aq)
Wait a minute! You might be asking, "Where did that extra 'H' come from in H₃O⁺?" Excellent question, super sleuth! Remember that hydrogen ion, H⁺, that flew off from HBrO? It's super attracted to the water molecules!
Water (H₂O) has two hydrogen atoms and one oxygen. When the H⁺ comes around, it’s like a little kid spotting a shiny new toy and immediately grabbing onto it. The H⁺ latches onto a water molecule, forming what we call the hydronium ion, H₃O⁺.
So, the complete equation, our grand scientific masterpiece, looks like this:
HBrO (aq) + H₂O (l) ⇌ H₃O⁺ (aq) + BrO⁻ (aq)
Isn't that just the coolest? It's a snapshot of Hypobromous Acid in action, showing us how it interacts with water, creating new buddies along the way, and then sometimes decides to be friends again. It's a constant, lively exchange!
This equation tells us about the acid's personality. Is it a strong personality that completely breaks apart, like a firework exploding with all its might? Or is it a more reserved personality, only giving away a little bit at a time, like a gentle whisper?
Hypobromous Acid, our featured performer today, is a bit on the reserved side. It doesn't give up all its hydrogen ions. Most of the HBrO molecules will happily stay as HBrO in the water, just like a few brave souls staying on the sidelines at a party while others are dancing.
This means that at any given moment, you'll find a mix of HBrO, H₂O, H₃O⁺, and BrO⁻ all hanging out together. They've reached a beautiful balance, an equilibrium, where the rate of forming products is the same as the rate of forming reactants. It's a perfect, ongoing truce!
So, next time you hear about Hypobromous Acid, don't just think of a complex chemical. Think of our shy but friendly molecule, playing its game of give-and-take in water, all beautifully captured in its acidic equilibrium equation. It’s a little piece of scientific poetry, a dance of atoms and molecules that keeps our world interesting!
Keep exploring, keep questioning, and remember, science is all around us, even in the shyest of acids! Hooray for chemistry!
