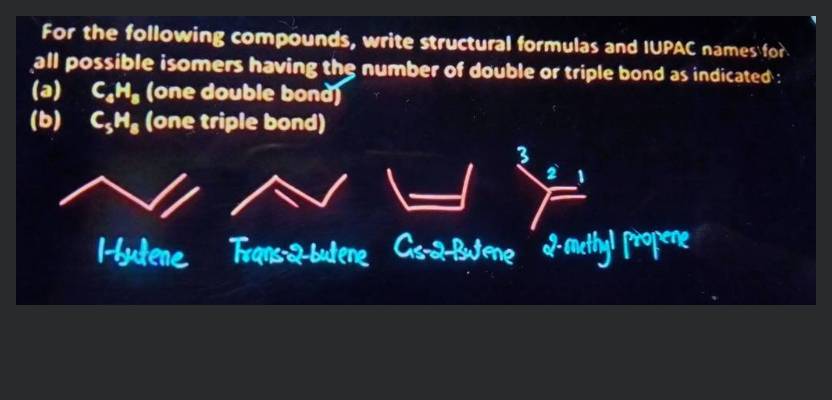
Write Structural Formulas For The Following Compounds

Imagine you’re at a huge family reunion, and everyone’s showing up with their own unique style. You’ve got Uncle Bob in his loud Hawaiian shirt, Aunt Carol with her elegant pearls, and little Timmy, who insists on wearing his superhero cape everywhere. Well, molecules are a bit like that! They’re all made of the same basic building blocks – atoms – but they can arrange themselves in so many different ways, creating a dazzling array of personalities and functions.
Today, we’re going to peek behind the curtain and get a glimpse of how these tiny personalities come together. It’s like learning the secret handshake of the molecular world! We’re going to be drawing out what are called structural formulas. Think of it as a super-simple map that shows us exactly how the atoms in a molecule are holding hands. No need to worry about complicated math or scary science jargon. We’re just going to have some fun discovering the shapes and connections that make our favorite things tick.
Let’s start with something you’ve probably encountered more times than you can count, perhaps even this morning with your coffee or tea. We're talking about water! We all know it’s essential for life, but have you ever stopped to think about its structure? It’s surprisingly simple and, in its own way, quite elegant. Picture this: one oxygen atom, which is like the parent in our little molecular family, and two smaller hydrogen atoms, like two energetic kids, all linked together. The oxygen atom is a bit of a diva; it likes to be in the middle, and it holds onto the two hydrogen atoms with a strong grip. So, if you were to draw it, it would look like a little ‘V’ shape, with the oxygen at the pointy end. This bent shape is actually super important for how water behaves – it’s why water molecules are so good at dissolving other things and why they stick together so well, giving us those lovely raindrops and the vast oceans.
Now, let’s move on to something a bit sweeter, something that might bring a smile to your face. Think about sugar. Not the processed stuff, but the natural sweetness you find in fruits. The most common type is called glucose, and its structure is a bit more of a party! Instead of just three atoms, glucose is a much bigger molecule. It has six carbon atoms, which are like the sturdy frame of a house, and then lots of hydrogen and oxygen atoms hanging off them. It’s a hexagonal ring structure, like a tiny, perfectly formed gemstone, with a few extra bits and bobs attached. Seeing its structural formula is like looking at a miniature, intricate blueprint. It’s this specific arrangement of atoms that gives glucose its sweet taste and makes it the go-to energy source for our bodies. It's truly a marvel of nature's design, all packed into something so tiny.
What about something that fuels our laughter and our late-night study sessions? Let’s consider caffeine, the magical ingredient in coffee and tea! Caffeine is a bit of a more complex character. It has a few rings of atoms linked together, creating a more intricate shape. Imagine a small, slightly tangled nest. It has carbon, hydrogen, oxygen, and even some nitrogen atoms, which add a bit of extra punch. This complex structure is what allows caffeine to interact with our brains in that unique way, making us feel more alert and focused. It’s fascinating to think that this tiny molecular arrangement is responsible for so many of our daily rituals and the boost we get from our favorite beverages. It’s like a tiny key that unlocks a specific door in our brains!
Let's take a detour into the world of smells, the kind that make you instantly feel at home. Think of the scent of a freshly cut lemon. That zesty aroma comes from a molecule called citric acid. Now, this one is a bit more of a branching structure, like a small tree with several branches. It’s made up of carbon, hydrogen, and oxygen atoms, and its specific shape is what allows it to interact with the scent receptors in our noses. The arrangement of those atoms is like a perfect little puzzle piece that fits just right to create that wonderfully refreshing smell. It’s a reminder that even the simplest sensory experiences are rooted in these intricate molecular designs.
Finally, let’s think about something that’s all around us, invisible but incredibly important: oxygen gas. This is the stuff we breathe! It’s surprisingly simple, consisting of just two oxygen atoms holding hands. But they hold on tightly! It’s a double bond, meaning they are sharing two pairs of electrons, making them a very stable and energetic pair. This simple, diatomic molecule is the very essence of life for most creatures on Earth. It's a stark contrast to the more complex structures we've seen, showing that sometimes, simplicity is the most powerful.
So, there you have it! A little peek into the secret lives of molecules. These structural formulas might look like scribbles to the untrained eye, but they are the blueprints for everything we see, smell, taste, and interact with. They tell a story of how atoms, like little building blocks, come together in countless combinations to create the vast and wonderful world around us. It’s a world of tiny engineers, constantly building and shaping our reality, one connection at a time.
