Write Both The Complete Electron-configuration Notation
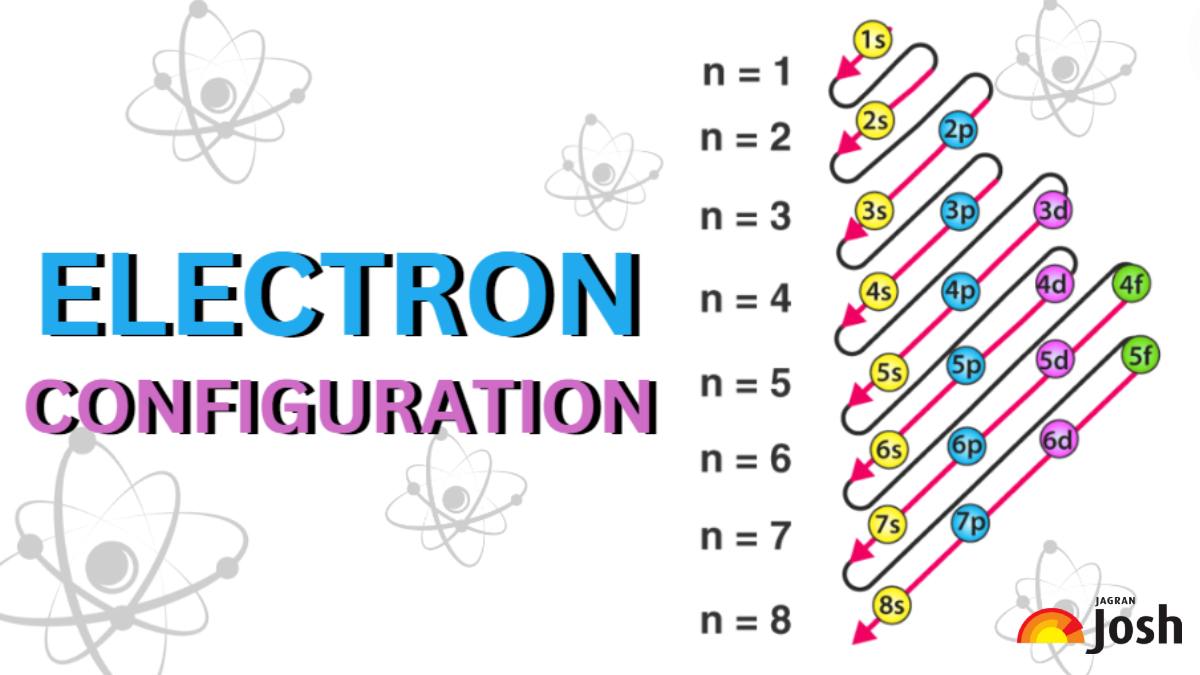
Ah, the humble electron configuration! For some, the very words might conjure up images of dusty textbooks and intimidating chemistry classes. But for a growing number of enthusiasts, delving into the intricate dance of electrons around an atom's nucleus is a surprisingly satisfying and even entertaining pursuit! Think of it like solving a cosmic puzzle, where each element has its own unique and elegant arrangement. It’s a way to understand the fundamental building blocks of everything we see and touch, and there's a certain thrill in deciphering these atomic blueprints.
So, why bother with electron configurations? Beyond the sheer intellectual curiosity, understanding how electrons are organized within an atom is absolutely crucial for predicting and explaining chemical behavior. This knowledge allows us to understand why different elements react the way they do, why water is a liquid at room temperature, and why metals conduct electricity. In everyday life, this translates to everything from the development of new medicines and materials to the creation of more efficient batteries and the design of vibrant dyes. Even understanding how your smartphone screen works or why your cooking food involves chemical reactions, is rooted in the principles of electron configuration. It’s the hidden language of the universe, and learning it unlocks a deeper appreciation for the world around us.
You might be surprised at how often we encounter applications of electron configurations. Every time you use a pharmaceutical, from aspirin to antibiotics, chemists have used electron configuration principles to design how those molecules will interact with your body. The creation of innovative plastics that make our cars lighter and our packaging more sustainable relies heavily on understanding how atoms bond, which is dictated by their electron arrangements. Even the humble act of baking involves chemical reactions driven by the electron configurations of the ingredients! The development of cutting-edge electronics, the vibrant colors in your clothes, and the fuels that power our transportation – all have their origins in the fundamental rules governing electron placement.
To truly enjoy the journey of learning electron configurations, a few practical tips can make all the difference. Firstly, visual aids are your best friend. Many online resources and textbooks offer diagrams of atomic orbitals and electron shells. Seeing these representations can make the abstract concept much more concrete. Secondly, start with the basics. Don't try to tackle the entire periodic table at once. Begin with the first few rows and focus on understanding the filling order of the orbitals (the famous 1s, 2s, 2p, etc.). Practice writing configurations for common elements like hydrogen, helium, lithium, and carbon. The more you practice, the more intuitive it becomes. Finally, connect it to the periodic table. Notice how the structure of the periodic table itself is a direct reflection of electron configurations. Elements in the same group often have similar outermost electron arrangements, explaining their similar chemical properties. It’s like a giant map that guides you through the atomic world, and understanding this connection is incredibly rewarding. So, embrace the puzzle, enjoy the discovery, and you might just find yourself captivated by the elegant world of electron configurations.
