Write Balanced Half Reactions For The Following Redox Reaction

Hey there, fellow humans! Ever feel like some things in life are just a little… unbalanced? Like when you eat a whole pizza and your significant other only had a salad? Yeah, that’s an unbalanced situation. Well, guess what? The universe, even at its tiniest atomic level, likes things to be balanced too. And today, we're going to peek behind the curtain at how scientists make sense of this cosmic balancing act, specifically when it comes to something called redox reactions.
Now, "redox" sounds like something from a sci-fi movie, right? Like a robot that’s been rebooted. But it’s actually just a fancy word for a fundamental process that happens all around us, all the time. Think of it like a cosmic give-and-take, a transfer of energy. Imagine your morning cup of coffee. That wonderful caffeine jolt? That's thanks to a whole bunch of tiny chemical reactions, some of which are redox reactions, getting your brain firing on all cylinders!
So, what's the big deal? Why should you, a person who probably has more important things to worry about like what’s for dinner or whether you remembered to pay that bill, care about these miniature chemical transfers? Well, knowing about them is like understanding how your body works. You don’t need to be a doctor to know that drinking water is good for you, right? Similarly, understanding redox reactions helps us understand everything from how batteries power our phones to how plants make their own food (photosynthesis!) and even how our own bodies digest food and breathe. It’s the secret sauce of so much that makes our modern world possible, and frankly, makes us alive!
Let’s break down this "redox" idea a bit. It's a combination of two words: reduction and oxidation. They always happen together, like a dynamic duo. You can’t have one without the other. It’s like a dance – one partner leads, the other follows, and the whole thing looks pretty cool when it’s done right.
Think about it this way: imagine a group of kids trading stickers. One kid might have a really rare holographic sticker, and another kid might have a bunch of common ones. The kid with the rare sticker might decide to trade it for a few common ones because they’re tired of it or want variety. In this scenario, the kid who loses the sticker is like the one being oxidized. They’re losing something valuable (in this case, electrons). The kid who gains the stickers is like the one being reduced. They’re gaining something.
Oxidation: The Giver
So, oxidation is all about losing electrons. Imagine a grumpy old man who’s always complaining about losing his keys. He’s constantly losing things, and in the chemical world, that “thing” is often electrons. When a substance gets oxidized, it gives away electrons. It’s like that friend who’s always lending you money – they’re giving something away.
Reduction: The Receiver
On the flip side, reduction is about gaining electrons. This is like the grateful person who always remembers to pay you back and then some. They’re taking something in. So, when a substance gets reduced, it accepts electrons from somewhere else. It’s like getting a surprise gift – you’re gaining something valuable!
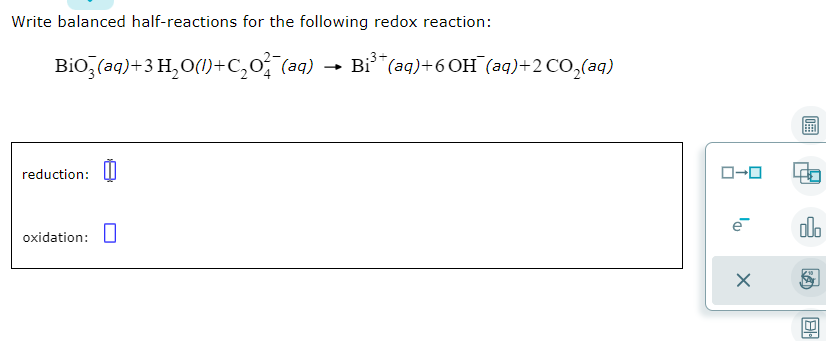
Now, here’s the crucial part: they always happen together. The electrons that one substance loses have to go somewhere, right? They don’t just disappear into the ether. They are transferred to another substance, which then gets reduced. So, the giver (oxidation) and the receiver (reduction) are locked in a chemical embrace. It’s like a seesaw – when one side goes down, the other must go up. It’s a beautifully balanced exchange.
Okay, so how do scientists actually *write this down and keep track of it? This is where we get to the nitty-gritty of writing balanced half-reactions. Think of it like splitting a complex recipe into two simpler steps. You wouldn’t try to explain how to bake a cake by just listing all the ingredients and saying "bake it," would you? You’d break it down into mixing the dry ingredients, then mixing the wet, then combining them, and finally baking. Half-reactions are similar; they break down the overall redox reaction into its two fundamental parts: the oxidation half and the reduction half.
Let's imagine we have a simple, everyday chemical reaction in mind. Think about rust. That reddish-brown stuff that forms on your bicycle if you leave it out in the rain? That’s iron (Fe) reacting with oxygen (O₂) in the air to form iron oxide. This is a classic redox reaction! The iron is losing electrons (getting oxidized), and the oxygen is gaining electrons (getting reduced).
The Irony of Rusting: A Daily Redox Example
Let's say we want to write the balanced half-reactions for this rusting process. It might seem a bit daunting, like trying to decipher ancient hieroglyphs. But we’ll take it step by step, like assembling a piece of IKEA furniture – with a bit of patience, you’ll get there!
First, we need to identify what’s being oxidized and what’s being reduced. In rusting, iron (Fe) turns into iron ions (like Fe²⁺ or Fe³⁺, depending on the exact conditions). This means the iron is losing electrons. So, the iron half-reaction will be an oxidation.

Meanwhile, oxygen (O₂) in the air is what the iron’s electrons are going to. Oxygen atoms tend to grab electrons to become more stable. So, the oxygen half-reaction will be a reduction.
Now, let’s get to the balancing act. We’re going to write two separate equations, each representing just one half of the overall process.
The Oxidation Half-Reaction (Iron's Loss)
We start with our iron. Let’s imagine our iron atom is neutral to begin with. When it gets oxidized, it forms an ion. For simplicity, let’s say it forms a Fe²⁺ ion, which means it has lost two electrons. So, our initial thought might be:
Fe → Fe²⁺
But we need to account for those lost electrons. Remember, oxidation is about losing electrons. So, we add the electrons to the side that lost them:

Fe → Fe²⁺ + 2e⁻
There we have it! The iron atom (Fe) starts off neutral, and it gives away two electrons (2e⁻) to become a positively charged iron ion (Fe²⁺). This is the oxidation half-reaction. See? Iron is being oxidized; it’s losing its precious electrons. It’s like the grumpy old man finally throwing away some of his old clutter.
The Reduction Half-Reaction (Oxygen's Gain)
Now for the oxygen. Oxygen usually exists as O₂ molecules. These molecules will gain electrons. Oxygen atoms typically want to gain two electrons to become oxide ions (O²⁻). But we have an O₂ molecule, which means two oxygen atoms. So, each oxygen atom in the O₂ molecule will likely gain two electrons, for a total of four electrons gained by the O₂ molecule to form two oxide ions.
Let's think about it: O₂ needs to become O²⁻ ions. Since there are two oxygen atoms, we'll end up with two O²⁻ ions. Each oxygen atom needs 2 electrons, so the O₂ molecule needs 4 electrons.
So, our reduction half-reaction involves O₂ gaining electrons to form oxide ions:

O₂ + 4e⁻ → 2O²⁻
Here, the oxygen molecule (O₂) grabs four electrons (4e⁻) and transforms into two oxide ions (2O²⁻). This is the reduction half-reaction. Oxygen is being reduced; it’s gaining those electrons that the iron so readily gave up. It’s like that generous friend who’s always happy to receive a thoughtful gift.
And there you have it! We’ve broken down the overall rusting process into its two essential halves: the iron giving away electrons (oxidation) and the oxygen grabbing those electrons (reduction). By writing these balanced half-reactions, we can see exactly what’s happening at the atomic level. It’s like looking at a crime scene and meticulously cataloging each piece of evidence to understand how the whole event unfolded.
Why is this so important, beyond just satisfying our scientific curiosity? Understanding these half-reactions is the key to designing and improving things like batteries. When you charge your phone, you're essentially reversing a redox reaction. When you use your phone, the battery's redox reaction is providing the power. Scientists balance these half-reactions to make sure batteries are efficient, long-lasting, and safe. It's also crucial in understanding how our bodies generate energy from food or how pollutants are broken down in the environment.
So, the next time you see rust, or charge your phone, or even just take a deep breath, remember the incredible dance of electrons happening all around you. And know that by breaking down complex processes into balanced half-reactions, scientists are not just doing homework; they're unlocking the secrets of the universe, one electron transfer at a time. Pretty neat, huh?
