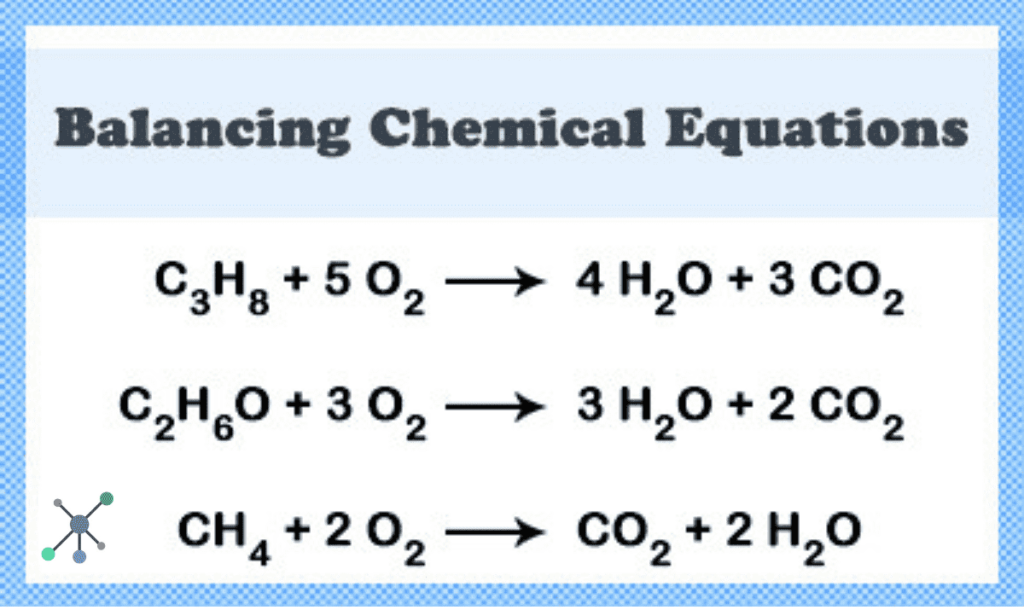
Write Balanced Equations For The Following Reactions

Hey there, lovely people! Ever feel like your life is a chaotic jumble of to-dos, social media scrolls, and that never-ending pile of laundry? Yeah, me too. But what if I told you that a little bit of balance, a sprinkle of order, could actually make things… well, smoother? We’re not talking about Marie Kondo-level minimalism here (though props to her!). We’re talking about finding that sweet spot, that harmonious equilibrium in the everyday. And guess what? The secret ingredient might just be hiding in plain sight, in the fascinating world of chemistry.
No, seriously! Think about it. Life is all about reactions, right? You react to stress, your friends react to your amazing jokes, and sometimes, your sourdough starter just reacts to being neglected. It’s all about inputs and outputs, causes and effects. And in chemistry, we have a super cool way of making sense of these transformations: balanced chemical equations.
Now, before your eyes glaze over and you start picturing Bunsen burners and dusty textbooks, let's take a deep breath. This isn't a pop quiz. This is about unlocking a little secret to understanding how things change, and by extension, how we can bring more balance to our own lives. Think of it like a beautifully choreographed dance, where every move has a purpose and everything is accounted for.
So, what exactly is a balanced chemical equation? Imagine you’re baking a cake. You need flour, sugar, eggs, and butter. When you mix them all up and bake them, you get a delicious cake. A balanced chemical equation is just like that, but for atoms. It’s a way of showing that in any chemical reaction, the number of atoms of each element on one side of the reaction (the ingredients, or reactants) must be exactly equal to the number of atoms of that same element on the other side (the product, or products). It's the universe's way of saying, "Hey, nothing is created or destroyed, just rearranged!" Pretty neat, huh?
This principle, known as the Law of Conservation of Mass, is a fundamental concept in chemistry. It’s like the ultimate tidy-up rule for the molecular world. Every atom that goes in has to come out. No sneaky atom disappearing acts allowed!
Let's dive into some of these reactions and see how we can make them sing. We'll keep it light, breezy, and with a side of fun facts, because who says learning can't be enjoyable?
The Combustion of Methane: A Fiery Tale
First up, let’s talk about something you might encounter every day, whether you realize it or not: burning natural gas. That’s often methane (CH4) doing its thing. When methane burns, it reacts with oxygen (O2) and produces carbon dioxide (CO2) and water (H2O).
The unbalanced equation looks like this: CH4 + O2 → CO2 + H2O
Let's break it down. On the left side (reactants), we have one carbon atom, four hydrogen atoms, and two oxygen atoms. On the right side (products), we have one carbon atom, two hydrogen atoms, and three oxygen atoms (two from CO2 and one from H2O). Uh oh, our hydrogens and oxygens are out of whack!
To balance it, we need to adjust the numbers in front of the chemical formulas (these are called coefficients). We want the same number of each type of atom on both sides.
Let's start with hydrogen. We have 4 H on the left and 2 H on the right. To balance them, we put a ‘2’ in front of H2O: CH4 + O2 → CO2 + 2H2O. Now we have 4 hydrogens on both sides. Sweet!
But wait, now let’s look at oxygen. On the left, we still have 2 O. On the right, we have 2 O in CO2 and 2 O in 2H2O (since there are two water molecules, and each has one oxygen, 2 x 1 = 2. Oops, that’s wrong. Each water molecule has two hydrogens and one oxygen. So in 2H2O, we have 2 x 2 = 4 hydrogens and 2 x 1 = 2 oxygens. My apologies, fellow learners! Let's correct that.)
Let's re-evaluate the oxygen count after balancing hydrogen: Left: 1 C, 4 H, 2 O Right: 1 C, 4 H (from 2H2O), 2 O (from CO2) + 2 O (from 2H2O) = 4 O total.
So, we have 2 O on the left and 4 O on the right. We need more oxygen on the left! If we put a ‘2’ in front of O2, we get 2 x 2 = 4 oxygen atoms.
The balanced equation is: CH4 + 2O2 → CO2 + 2H2O
Let's check: Left: 1 C, 4 H, 4 O Right: 1 C, 4 H, 4 O. Perfect! It's balanced!
Fun Fact: Methane is the primary component of natural gas, and it’s also produced naturally by animals, including us! So, that little bit of… gas… is actually a chemical reaction happening in your own body. How’s that for a conversation starter at your next dinner party?
Practical Tip: Just like balancing this equation requires careful counting, so does managing your energy. Don’t overcommit yourself (too much O2 on one side!). Make sure you have enough "ingredients" of rest and relaxation to fuel your "combustion" of activities.
The Synthesis of Ammonia: A Nitrogen Fix
Next up, we have the Haber-Bosch process, which is a HUGE deal for agriculture. It’s how we make ammonia (NH3), a key ingredient in fertilizers that helps feed the world. It involves nitrogen gas (N2) and hydrogen gas (H2) reacting to form ammonia.
The unbalanced equation is: N2 + H2 → NH3
Looking at the left side, we have 2 nitrogen atoms and 2 hydrogen atoms. On the right, we have 1 nitrogen atom and 3 hydrogen atoms. Our numbers are all over the place!
Let’s balance the nitrogen first. We have 2 N on the left and 1 N on the right. So, we put a ‘2’ in front of NH3: N2 + H2 → 2NH3. Now we have 2 N on both sides. Excellent!
Now, let’s check the hydrogen. On the left, we have 2 H. On the right, in 2NH3, we have 2 x 3 = 6 hydrogen atoms. We need more hydrogen on the left!
To get 6 hydrogen atoms on the left, we need to put a ‘3’ in front of H2 (since 3 x 2 = 6).
The balanced equation is: N2 + 3H2 → 2NH3
Let’s check again: Left: 2 N, 6 H (from 3H2) Right: 2 N (from 2NH3), 6 H (from 2NH3) It’s balanced!
Cultural Reference: The Haber-Bosch process is considered one of the most important inventions of the 20th century, without which, it's estimated, the world's population would be significantly smaller. It's a testament to human ingenuity and our ability to harness chemical reactions for the greater good. Think of it as the ultimate life hack, created by science!
Practical Tip: Just like this process combines simple elements to create something vital, we can combine simple daily habits to create a more fulfilling life. A little bit of exercise, a few moments of mindfulness, a healthy meal – these "reactants" can lead to a more balanced and energized "product." Don't underestimate the power of small, consistent actions.
The Decomposition of Hydrogen Peroxide: A Bubbling Wonder
Ever seen hydrogen peroxide (H2O2) bubble up when you pour it on a cut? That's it breaking down into water (H2O) and oxygen gas (O2). It’s a pretty common reaction, and sometimes it’s sped up by a catalyst, like the manganese dioxide you might see in science demonstrations.
The unbalanced equation: H2O2 → H2O + O2
Let’s count. Left side: 2 H, 2 O. Right side: 2 H, 3 O (2 from H2O, 1 from O2). Our oxygen is not happy!
To balance the oxygen, we need an even number on both sides. Let's try putting a ‘2’ in front of H2O2: 2H2O2 → H2O + O2. Now we have 2 x 2 = 4 hydrogen atoms and 2 x 2 = 4 oxygen atoms on the left.
On the right, we still have 2 H and 3 O. We need to adjust the products. Let's put a ‘2’ in front of H2O: 2H2O2 → 2H2O + O2. Now we have 2 x 2 = 4 H and 2 x 2 = 4 O on the left. On the right, we have 2 x 2 = 4 H and 2 O (from 2H2O) + 2 O (from O2) = 4 O.
The balanced equation is: 2H2O2 → 2H2O + O2
Checking: Left: 4 H, 4 O Right: 4 H, 4 O. Balanced!
Fun Fact: Hydrogen peroxide is naturally present in small amounts in rain and is thought to play a role in its antibacterial properties. So, next time it rains, you can think of it as a gentle, natural disinfectant!
Practical Tip: Sometimes, in life, we feel like we're "decomposing" under pressure. But just like hydrogen peroxide, this breakdown can lead to the release of something essential – like new insights or a stronger sense of self. Embrace those moments of shedding the old to make way for the new. It’s a natural process of growth.
The Formation of Water: A Classic Combination
We’ve seen water as a product, now let’s see how it’s formed from its basic components: hydrogen gas (H2) and oxygen gas (O2). This is a fundamental reaction, and it’s important for understanding so many other chemical processes.
The unbalanced equation: H2 + O2 → H2O

On the left, we have 2 H and 2 O. On the right, we have 2 H and 1 O. Our oxygen is out of balance.
To balance the oxygen, we need more on the right. Let’s put a ‘2’ in front of H2O: H2 + O2 → 2H2O. Now we have 2 H and 2 O on the left, and 2 x 2 = 4 H and 2 x 1 = 2 O on the right. Our hydrogen is now out of balance!
To balance the hydrogen, we need more on the left. We have 2 H on the left and 4 H on the right. So, let’s put a ‘2’ in front of H2: 2H2 + O2 → 2H2O.
The balanced equation is: 2H2 + O2 → 2H2O
Let’s do our final check: Left: 4 H (from 2H2), 2 O Right: 4 H (from 2H2O), 2 O (from 2H2O) It’s balanced!
Cultural Reference: Water is so fundamental to life that it features in countless myths, legends, and religious practices across the globe. From the mythical rivers of the afterlife to the cleansing rituals of baptism, its importance is undeniable. It’s the ultimate symbol of purity, renewal, and, of course, balance.
Practical Tip: Just as the right ratio of hydrogen and oxygen creates life-giving water, the right balance of different elements in our lives creates a rich existence. It’s not about having everything, but about having the right things in the right proportions. A healthy diet, meaningful relationships, time for hobbies, moments of quiet reflection – these are the "atoms" that form the "water" of a fulfilling life.
So, there you have it! A little peek into the world of balanced chemical equations. It might seem like just numbers and symbols, but at its core, it’s about order, about ensuring that everything that goes in comes out, just in a different form. It’s a beautiful reminder that even in the most complex transformations, there’s an underlying principle of balance at play.
And isn't that what we're all striving for? Balance. In our schedules, in our relationships, in our well-being. The universe, in its infinite wisdom, has this beautiful way of showing us these principles, even in the seemingly mundane. So, the next time you see a chemical reaction, whether it's your morning coffee brewing or that bubbling cut on your knee, take a moment to appreciate the quiet, elegant dance of atoms, and remember the power of balance. It’s a concept that’s as essential to chemistry as it is to living a happy, harmonious life. Until next time, stay balanced, stay curious, and keep those equations in your life… balanced!
