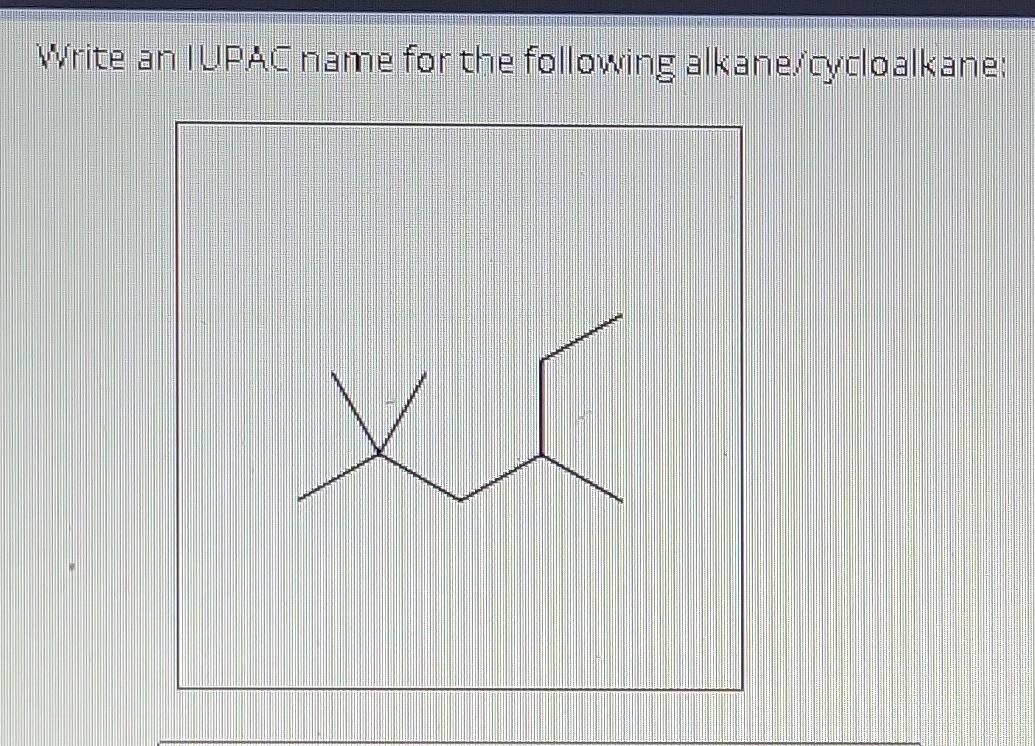
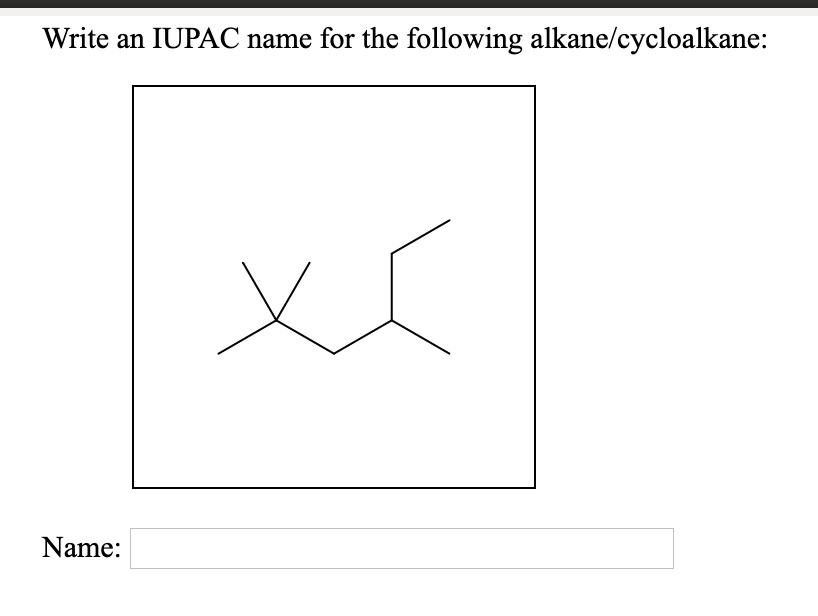
Write An Iupac Name For The Following Alkane/cycloalkane
Ever looked at a complex chemical formula and felt your brain do a little shimmy, like it’s trying to do the cha-cha with a textbook? Yeah, me too. Especially when we’re talking about alkanes and cycloalkanes. They sound all fancy and scientific, like something you’d find on a lab coat in a movie, right? But honestly, naming these things using the fancy-pants IUPAC (International Union of Pure and Applied Chemistry) rules is just like giving your pet a proper nickname. You could just call your dog “Dog,” but wouldn’t “Sir Reginald Fluffernutter the Third, Esquire” be way more fun and descriptive? That’s essentially what IUPAC naming is doing for these molecules – giving them their full, official, and sometimes hilariously long, birth certificates.
Think of it this way: we’ve all got our own unique names, and sometimes we have a nickname that’s shorter, cuter, or just plain funnier. My cousin Brenda? Her official name is Brenda, but to us, she’s “Bren-Buns” because she makes amazing cinnamon buns. See? A nickname. IUPAC naming is like giving our little carbon-chain friends their very own “Bren-Buns” but with more structure and a lot less flour.
So, why do we even bother with this whole IUPAC rigmarole? Well, imagine you’re trying to order a pizza and you just say, “Give me a round, cheesy thing.” The pizza guy would be like, “Uh, what kind?” You need specifics! Is it pepperoni? Mushroom? Extra anchovies (don’t judge)? IUPAC gives chemists that same kind of specificity. It’s a universal language so that no matter where you are in the world, or what lab you’re in, if you say “2,4,6-trichloroheptane,” everyone knows exactly what you’re talking about. No more confused head-tilting. It’s all about preventing misunderstandings that could lead to, you know, accidentally creating a super-villain ray instead of a mild disinfectant. Been there, done that. (Okay, maybe not that dramatic, but you get the drift).
Let’s start with the simplest ones, the alkanes. These are basically the plain white t-shirts of the hydrocarbon world. They’re made of only carbon and hydrogen atoms, all linked up like a string of tiny, happy pearls. No double bonds, no triple bonds, just good old single bonds. They’re saturated with hydrogen, like a sponge that’s had a good soak. Hence, “alkane.”
Now, when it comes to naming them, the IUPAC folks are pretty systematic. It’s like following a recipe. First, you gotta find the longest continuous chain of carbon atoms. This is your main character, the star of the show. Everything else is a supporting actor. So, if you have a chain of, say, five carbons, that’s your base. The prefix for five is “pent-”. Easy peasy, lemon squeezy.
Here’s a little cheat sheet for the first few prefixes, because memorizing these is almost as fun as watching paint dry, but way more useful: * 1 carbon: Meth- (like methamphetamine, but way less exciting and definitely not illegal!) * 2 carbons: Eth- (think ethanol, the stuff in your hand sanitizer… and some drinks!) * 3 carbons: Prop- (propeller? maybe… or propane for your grill) * 4 carbons: But- (butane, like in your lighter) * 5 carbons: Pent- (pentagon, has five sides!) * 6 carbons: Hex- (hexagon, six sides!) * 7 carbons: Hept- (hepta- means seven) * 8 carbons: Oct- (octopus has eight arms!) * 9 carbons: Non- (nonagon, nine sides) * 10 carbons: Dec- (decade, ten years!)

And guess what? If it’s an alkane, you just tack on “-ane” to the end of the prefix. So, a single carbon chain with only single bonds is methane. Two carbons? Ethane. Three? Propane. Four? Butane. See? It’s like you’re building with LEGOs, and each brick has a name. Methane is like a single LEGO brick, ethane is two clicked together, and so on. Pretty neat, huh?
But what happens when those carbon chains get a bit of personality? When they start branching out like a nervous sapling trying to find more sunlight? That’s where things get a little more interesting. These branches are called alkyl groups, and they’re basically shorter alkane chains that have broken off from the main chain. Think of it like a family tree – you have your main lineage, and then you have cousins who branched off a generation or two ago.
When naming these branched alkanes, the IUPAC rules get a bit more detailed, like explaining the rules of a complex board game. First, you find the longest continuous carbon chain, just like before. This is your main road. Then, you have to number the carbons on this main chain. And this is where it gets crucial: you start numbering from the end that gives the substituent (the branch) the lowest possible number. This is like giving directions – you want to tell someone the shortest route, not send them on a wild goose chase.
Let’s say you have a chain of five carbons (pentane) and a methyl group (a one-carbon branch, hence “meth-” and since it’s a branch, we use “-yl”, so it’s a methyl group) attached to it. If you number from one end, the methyl group might be on carbon number 2. If you number from the other end, it might be on carbon number 4. We want the lowest number, so we’d choose the numbering that puts it on carbon 2. So, it would be called 2-methylpentane. See? 2 for the position, methyl for the branch, and pentane for the main chain. It’s like saying, “On the second floor, there’s a cute little studio apartment (methyl) in the five-story building (pentane).”
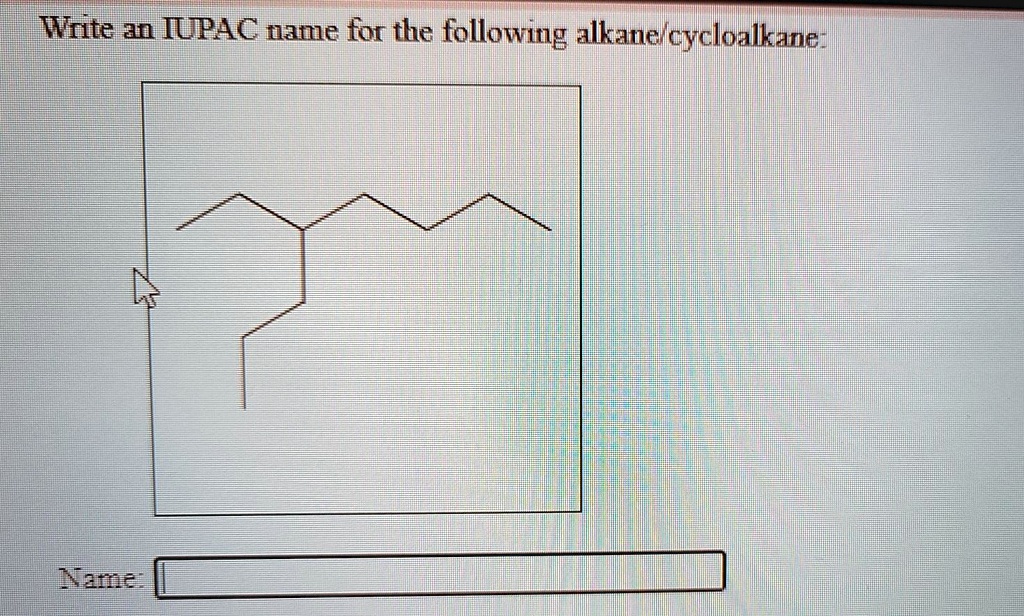
![[Solved]: Write an IUPAC name for the following alkane/](https://media.cheggcdn.com/study/45f/45fff308-3635-41d4-aa27-fc34f168500b/image.jpg)
What if there are multiple branches? This is where it gets like assembling a very specific IKEA furniture piece. You have to identify all the branches, determine their position on the main chain, and then list them in alphabetical order. And here’s a fun quirk: if you have multiple identical branches, you use prefixes like di- (for two), tri- (for three), tetra- (for four), and so on. So, if you have two methyl groups on carbon 2 and carbon 4 of a heptane chain, it would be 2,4-dimethylheptane. You’d list the positions separated by commas, then the prefix (di-), then the name of the branch (methyl), and finally the main chain (heptane).
It’s like a very organized scavenger hunt for your molecules! You’re looking for the longest chain, then you’re hunting down all the little branches, noting their locations, and finally arranging them all neatly like little soldiers in a parade. Alphabetical order for the branches, lowest numbers for their positions. It’s all about being as clear and unambiguous as a stop sign.
Now, let’s talk about the cycloalkanes. These guys are the ones who decided to form a circle instead of just a straight line. They’re like that one friend who always wants to hold hands and form a circle during games, but in a chemically responsible way. The most common ones are cyclopropane (a triangle of carbons), cyclobutane (a square), cyclopentane (a pentagon), and cyclohexane (a hexagon). Think of them as the tidy, self-contained members of the hydrocarbon family.
Naming them is actually pretty straightforward. You just take the name of the corresponding alkane (based on the number of carbons in the ring) and add the prefix “cyclo-”. So, a ring of three carbons is cyclopropane. A ring of six carbons is cyclohexane. It’s like putting a little “cyclo” hat on the alkane name.
What if there are branches on these cyclic structures? Well, it’s a bit like a circular street party. If there’s only one substituent on the ring, the carbon it’s attached to is automatically considered carbon number 1. You don’t need to number it. It’s like the VIP section of the party – it gets the prime spot by default. So, if you have a methyl group attached to cyclohexane, it’s just called methylcyclohexane.
However, if you have more than one substituent, you have to start numbering the ring. And just like with the branched alkanes, the rule is to give the substituents the lowest possible numbers. You also need to list the substituents in alphabetical order. For example, if you have a methyl group and an ethyl group (a two-carbon branch) on a cyclopentane ring, you'd number it so the ethyl group gets a lower number (since ‘ethyl’ comes before ‘methyl’ alphabetically). So, if they are on adjacent carbons, it would be 1-ethyl-2-methylcyclopentane. The ‘1’ and ‘2’ are the lowest numbers you can get while keeping them in alphabetical order. It’s like assigning seat numbers at a very exclusive dinner party – everyone gets their best spot.
Let’s try a quick one. Imagine a molecule that looks like a hexagon with a little tail on one of the corners. The hexagon part is six carbons, so that’s a cyclohexane. The tail is a single carbon, a methyl group. Since there’s only one branch, the methyl group is automatically at position 1. So, the name is simply methylcyclohexane. Easy peasy!
Now, what if that same hexagon had two tails? Let’s say one tail is a methyl group and the other is an ethyl group (two carbons). They’re both branches on the cyclohexane ring. To figure out the numbering, we look at their alphabetical order: ethyl comes before methyl. So, we want to give the ethyl group the lowest possible number. If they are on adjacent carbons, the ethyl group will be at position 1 and the methyl group at position 2. So, the name would be 1-ethyl-2-methylcyclohexane. If they were on carbons that were two apart, it might be 1-ethyl-3-methylcyclohexane, and so on. We always go for the lowest set of numbers that keeps the alphabetical order.
It might seem a bit like a puzzle at first, but once you get the hang of the basic rules – finding the longest chain, numbering correctly, identifying branches, and alphabetizing – it becomes much less intimidating. Think of it as learning to read a map. At first, it’s a jumble of lines and symbols, but with a little practice, you can navigate anywhere!
The beauty of IUPAC naming is its consistency. It’s designed to be a universal language so that chemists worldwide can communicate effectively. It’s the molecular equivalent of a handshake that means the same thing everywhere. So next time you see a long, complex chemical name, don’t let it scare you. Just remember it’s a system, a way of giving each molecule its own unique and descriptive identifier, much like how we give our friends and family their own special names. It’s just science, but with a touch of organizational flair!
Ultimately, writing an IUPAC name for an alkane or cycloalkane is like being a detective. You’re looking for clues (the longest chain, the branches), identifying the suspects (the types of groups), and then putting them all in order according to the rulebook. It’s a logical process, and once you break it down, it’s actually quite satisfying to get it right. So, next time you’re faced with a skeletal structure, channel your inner molecular detective and have fun naming your way to discovery!
