Write A Balanced Equation For The Combustion Of Methanol

Alright, gather 'round, my friends, and pull up a comfy chair! We're about to embark on a culinary (or rather, chemical!) adventure that's far more exciting than your average microwave meal. Today, we're talking about combustion. Now, I know what you're thinking. "Combustion? Isn't that just fancy talk for 'setting things on fire'?" And you'd be hilariously, wonderfully right! But there's a method to the madness, a scientific ballet happening in every flickering flame. We're going to demystify the balanced equation for the combustion of methanol, and trust me, it’s going to be way more fun than you expect.
Imagine this: you're at a cozy little cafe, the aroma of freshly brewed coffee is filling the air, and you've just ordered a ridiculously decadent slice of chocolate cake. Suddenly, your eccentric chemistry professor, Professor Fizzwick, slides into the seat opposite you. He's got that twinkle in his eye that usually means he's about to unleash a torrent of fascinating, slightly alarming facts. He slams a glass of what looks suspiciously like rubbing alcohol on the table. "My dear chap," he booms, "this, my friend, is methanol! And it's about to go on a fiery adventure!"
Now, methanol, bless its little molecular heart, is a simple alcohol. Think of it as the budget version of the ethanol you find in your celebratory beverages. It’s got one carbon atom, four hydrogen atoms, and one oxygen atom. We write it as CH₃OH. It's like a tiny, one-bedroom apartment for atoms. And when this little apartment gets a bit too excited, it decides to have a grand old party. This party, in chemistry terms, is called combustion. It's essentially methanol meeting its fiery soulmate: oxygen.
Where does this oxygen come from? Well, unless you're conducting your experiments in a vacuum (which, let's be honest, sounds incredibly boring), it's usually floating around in the air, just waiting for its cue. We represent oxygen as O₂. Notice the '2' there? That means oxygen atoms are a bit like a clingy couple; they rarely go anywhere without each other. They're always holding hands, a diatomic duo!
So, we have our methanol (CH₃OH) and our oxygen (O₂). When these two get together, sparks fly! Literally! The methanol grabs the oxygen, and a rather vigorous dance ensues. This dance, my friends, breaks all the old chemical bonds and creates entirely new ones. It's like a messy breakup followed by a whirlwind romance, all happening at the molecular level. And what's the result of this fiery tango? Drumroll, please… carbon dioxide and water!
Carbon dioxide, that stuff your fizzy drinks are made of and what you exhale with every breath, is written as CO₂. Think of it as the energetic, slightly gassy teenager of the chemical world. And water? Well, you know water (H₂O). It’s the calm, refreshing friend who's always there for you. So, our methanol, in its fiery transformation, produces these two. Pretty neat, right?
But here's where the "balanced equation" part comes in, and this is where Professor Fizzwick would adjust his spectacles with a knowing smile. A balanced equation is like making sure everyone gets an equal slice of the cake. In chemistry, it means that the number of atoms of each element we start with on one side of the equation must be the same as the number of atoms of that element we end up with on the other side. No atoms are created or destroyed in this chemical fiesta; they just rearrange themselves like playful Lego bricks.
Let's start writing it down, shall we? On the "reactant" side (that's the stuff we start with, the ingredients for our fiery feast), we have methanol and oxygen:
CH₃OH + O₂

And on the "product" side (what we get after the combustion party), we have carbon dioxide and water:
CO₂ + H₂O
So, our initial, unbalanced equation looks something like this:
CH₃OH + O₂ → CO₂ + H₂O
Now, let's count our atoms. On the left side (the reactant side), we have: * Carbon (C): 1 atom (in CH₃OH) * Hydrogen (H): 4 atoms (3 in CH₃OH + 1 in CH₃OH) * Oxygen (O): 3 atoms (1 in CH₃OH + 2 in O₂) On the right side (the product side), we have: * Carbon (C): 1 atom (in CO₂) * Hydrogen (H): 2 atoms (in H₂O) * Oxygen (O): 3 atoms (2 in CO₂ + 1 in H₂O)
Uh oh. See the problem? Our hydrogens are out of whack. We start with 4 hydrogens, but we only end up with 2. This is like inviting 10 people to a party and only having 5 chairs. Not cool. We need to add some coefficients (those are like little numbers in front of the chemical formulas) to even things out. Think of coefficients as telling us "how many" of each molecule we need.
Let's tackle the hydrogen first. We need 4 hydrogens on the product side. Our water molecule (H₂O) only has 2. So, if we put a '2' in front of the water, we'd have 2 * 2 = 4 hydrogens. Perfect! Our equation now looks like this:
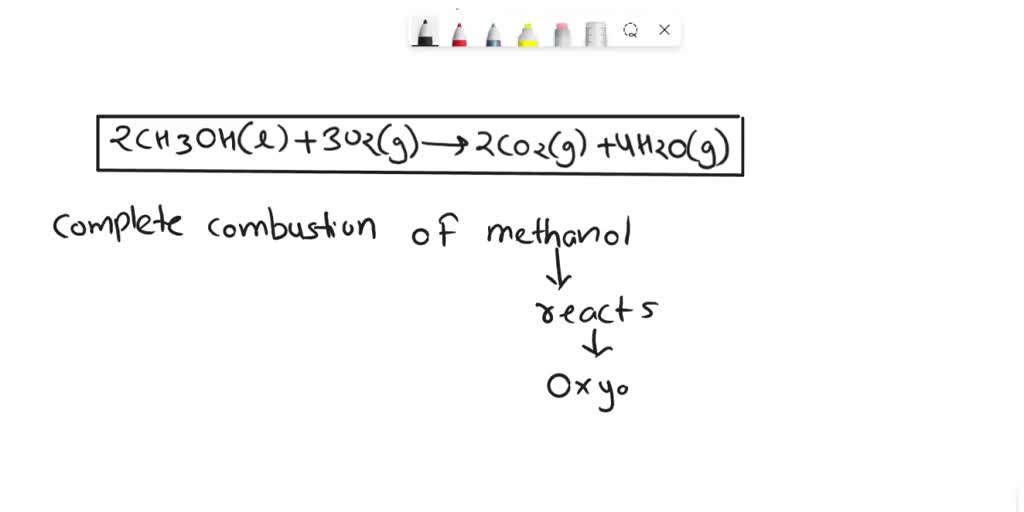
CH₃OH + O₂ → CO₂ + 2H₂O
Let's recount our atoms:
Left side:
* C: 1 * H: 4 * O: 3 (1 + 2) Right side:* C: 1 * H: 4 (2 * 2) * O: 4 (2 in CO₂ + 2 * 1 in H₂O)Now our hydrogens are balanced! High five! But look at the oxygen. We started with 3 on the left and now we have 4 on the right. Still not quite right. We need to adjust our oxygen on the reactant side. We have one oxygen in the methanol (CH₃OH), which we can't touch because it’s part of the methanol itself (it's like the host at the party). So, we need to adjust the oxygen molecules (O₂).
We need 4 oxygens on the product side, and we already have 1 in the methanol. That means we need 3 more oxygens from the oxygen molecules. Since oxygen molecules come in pairs (O₂), and we need 3 atoms, that's like asking for 1.5 pairs. In chemistry, we don't do halves of molecules (that would be weird!), so we use fractions if necessary, but the final balanced equation will have whole numbers. So, we need 1.5 molecules of O₂. A '1.5' is written as '3/2'.
Our equation now becomes:
CH₃OH + 3/2 O₂ → CO₂ + 2H₂O
Let's check again:
Left side:
* C: 1 * H: 4 * O: 1 (in CH₃OH) + (3/2 * 2) = 1 + 3 = 4 Right side:* C: 1 * H: 4 * O: 2 (in CO₂) + (2 * 1 in H₂O) = 2 + 2 = 4Voila! We have 1 carbon on both sides, 4 hydrogens on both sides, and 4 oxygens on both sides. The equation is balanced! This is our friend, Professor Fizzwick's, masterpiece!

However, we chemists generally prefer to deal with whole numbers. Fractions like '3/2' can feel a bit… incomplete. So, to get rid of that pesky fraction, we multiply the entire equation by 2. Think of it as doubling the recipe to make sure everyone gets a full serving!
Multiplying everything by 2:
(2 * CH₃OH) + (2 * 3/2 O₂) → (2 * CO₂) + (2 * 2H₂O)
This gives us our final, elegant, and perfectly balanced equation:
2CH₃OH + 3O₂ → 2CO₂ + 4H₂O
So, what does this mean in plain English? It means that for every two molecules of methanol that decide to have a fiery party, you need three molecules of oxygen. And when the dust (or rather, the smoke and steam) settles, you get two molecules of carbon dioxide and four molecules of water. It’s a perfect chemical stoichiometry, a dance of atoms where everyone knows their part.
And there you have it! The balanced equation for the combustion of methanol. It’s not just about fire; it’s about order, precision, and making sure no atom feels left out. So next time you see a flame, remember the incredible molecular choreography happening within. Now, about that chocolate cake…
