Worksheet Determination Of Protein Amino Acids

Hey there! So, you wanna chat about determining protein amino acids, huh? Sounds super fancy, right? Like something a mad scientist in a lab coat might do. But honestly, it’s not as intimidating as it sounds. Think of it as being a detective, but instead of fingerprints, we’re looking for… well, amino acids.
You know, those tiny building blocks that make up all the proteins in our bodies? Yeah, those guys. They’re like the LEGOs of life, seriously. And figuring out which LEGOs are in a particular protein, and how many of each, is what this whole “worksheet determination” thing is all about. Fun, right?
So, imagine you’ve got this mystery protein. Maybe it’s in your salad, maybe it’s doing something super cool in a cell. We wanna know its recipe. What’s it made of? That’s where the detective work begins! We're not just guessing; we have tools and techniques, and of course, the trusty worksheet to keep us organized. Because let's be real, who remembers all those amino acid names off the top of their head? Not me, that's for sure. I’d be lost without my cheat sheet, aka the worksheet!
First off, why bother? Why spend our precious time figuring out these little molecules? Well, knowing the amino acid composition can tell us a ton about a protein. Is it going to be flexible or rigid? Does it like water, or does it prefer to hide away? It's like getting a sneak peek at its personality before you even really meet it. Super useful for understanding how proteins work, what they do, and even how they might go wrong in diseases. Talk about power!
So, how do we do it? It’s a multi-step process, and each step has its own little quirks and challenges. But the core idea is pretty straightforward: you break down the protein, you identify the individual amino acids, and then you count them up. Easy peasy, right? (Spoiler alert: it’s never as easy as it sounds, but we can pretend).
The very first hurdle, the big boss level, is getting your hands on a pure protein sample. Imagine trying to identify the ingredients in a smoothie when someone’s already dumped a whole fruit bowl into it. Not ideal! So, scientists spend a lot of time making sure their protein is super clean. No unwanted extras allowed. Think of it as setting the stage for a very important scientific performance.
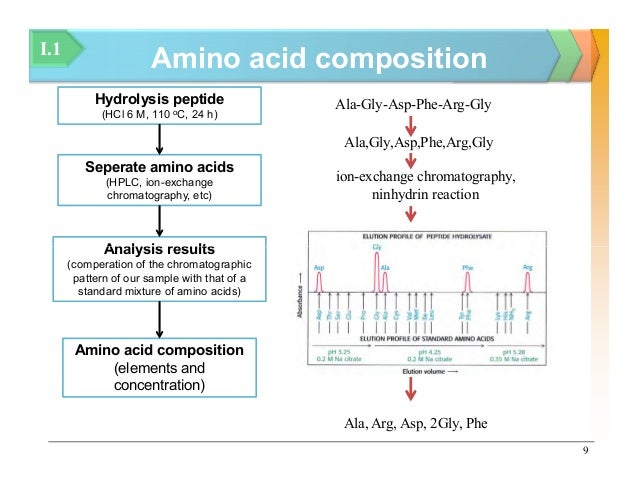
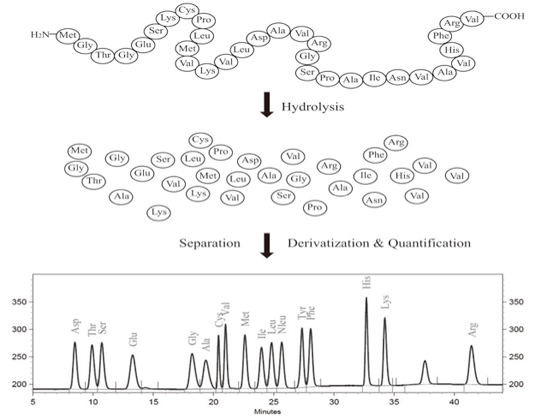
Once we have our pristine protein, we gotta break it down. Proteins are long chains, right? Like a super long string of beads. To count the individual beads, we need to snip the string. And how do we do that? With hydrolysis. Fancy word, I know. It basically means using water to break those peptide bonds, those strong links holding the amino acids together. Think of it like a chemical scissors, powered by water. Pretty neat, huh?

Now, there are a few ways to do this hydrolysis. Heat is usually involved. Lots of heat. Like, really hot. You’re talking boiling acids for hours. It’s not a gentle process, this protein breakdown. It's more of a full-on chemical assault. But it works! It liberates those individual amino acids, setting them free to be identified. Freedom! For the amino acids, at least.
There are also different kinds of hydrolysis. Acid hydrolysis is the classic. You chuck in some hydrochloric acid, crank up the heat, and voilà! But some amino acids are a bit sensitive to this extreme treatment. Methionine, cysteine, tryptophan – they can get a little messed up, like they’ve been through a rough laundry cycle. So, sometimes, you gotta be gentler. Enzymatic hydrolysis uses special protein-chopping enzymes. It’s more precise, but can be slower. It’s all about choosing your weapon wisely for your specific protein mission.
After all that intense breaking down, we're left with a soup of individual amino acids. Now, the real detective work starts: figuring out what is in that soup and how much of each. This is where chromatography comes in. Oh boy, chromatography. It sounds like a disease, but it's actually your best friend in this situation.
Think of chromatography as a super-sophisticated sorting machine. We use a technique called ion-exchange chromatography or sometimes reverse-phase chromatography. The basic idea is that different amino acids have different electrical charges and different levels of “stickiness” to certain surfaces. We pass our amino acid soup through a special column, and as it travels, the amino acids that are more charged or stickier will hang around longer, while the less charged or less sticky ones will zip through faster.
It’s like a race! The column is the track, and the amino acids are the runners. Some runners are slow and steady, others are sprinters. By carefully controlling the conditions – like the pH of the liquid flowing through the column – we can make them separate out beautifully. It’s pure scientific magic, if you ask me. And the result? Separate little pools of each amino acid. Ta-da!
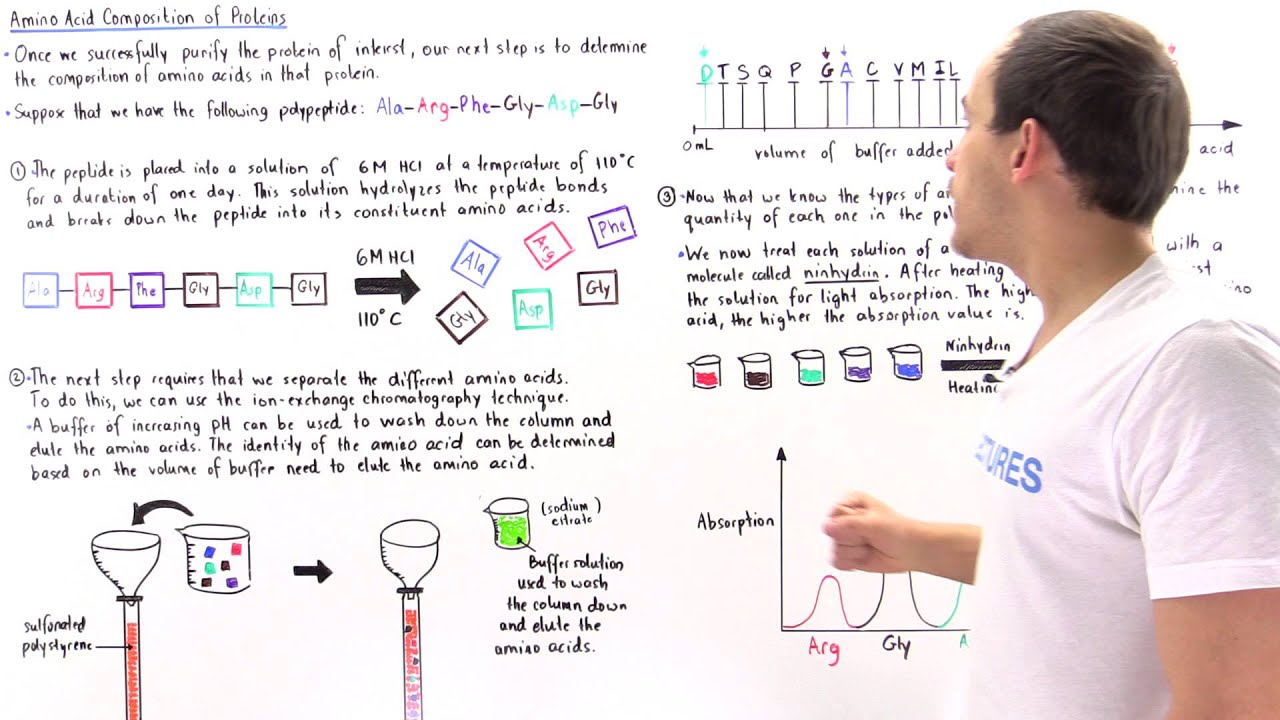
But wait, there’s more! Just separating them isn’t enough. We need to quantify them. We need to know how many of each amino acid we have. This is where detection comes in. Most amino acids are invisible to the naked eye, which is a bit inconvenient for a detective. So, we have to make them show themselves! We add a special chemical called a derivatizing agent. It’s like giving each amino acid a bright, glow-in-the-dark hat.
This derivatizing agent attaches to the amino acids, making them fluorescent. Now, when the separated amino acids come out of the chromatography column, they pass through a detector that can measure their fluorescence. The brighter the fluorescence, the more amino acid there is! It’s like a treasure hunt, but instead of gold, we’re looking for bright little sparks of light.
The detector spits out a graph, and this graph is our chromatogram. It’s got little peaks, and each peak represents a different amino acid. The area under each peak is directly proportional to the amount of that amino acid present. So, bigger peak, more amino acid. Simple enough, right? (Again, let's just roll with the simplicity).
Now, here’s where the worksheet really shines. It’s our organized way of capturing all this information. You’ll typically have columns for each amino acid you’re looking for. And then, next to it, you’ll have space to record the peak area from the chromatogram. You might also have a column for a standard, which is essentially a known amount of a pure amino acid used to calibrate your detector. It’s like having a ruler to measure your unknown quantities.
You’ll compare the peak area of your unknown amino acid to the peak area of your standard. And voilà! With a little bit of math (don’t run away!), you can calculate the exact amount of each amino acid in your original protein sample. For example, if your standard of glycine gave you a peak area of 100 units, and your unknown protein sample gave you a glycine peak area of 500 units, then you have about 5 times more glycine in your sample than in your standard. See? Math!
The worksheet helps you keep track of all these calculations. You’ll convert peak areas into molar amounts, and then you can express your results as percentages of the total amino acid content. So, you might find your protein is, say, 10% glycine, 8% alanine, 5% valine, and so on. It’s like getting the complete ingredient list and their exact proportions.
But here's a little secret: it's not always perfectly straightforward. As I mentioned, some amino acids are tricky. Tryptophan can be destroyed during acid hydrolysis, so you might get a really low number, or even zero, if you’re not careful. And cysteine can form disulfide bonds with itself or other cysteine molecules, making it harder to quantify. So, sometimes, you have to do extra steps, like perform reduction and alkylation, to break those disulfide bonds and free up the cysteines. It’s like putting on special armor for your cysteine detectors.
And then there's proline. Proline is a bit of a rebel. It's an imino acid, not a true amino acid, and its peptide bonds can be a bit more resistant to hydrolysis. So, sometimes, you need longer hydrolysis times or more vigorous conditions to break them down completely. It’s like proline is saying, "Nah, I'm not breaking that easily!"
Don't forget about ammonia! During hydrolysis, the amide groups on asparagine and glutamine get converted to ammonia. So, when you measure ammonia, you’re actually measuring the sum of the original asparagine and glutamine. You have to figure out how to distinguish between them, which can involve other analytical techniques or making assumptions based on typical protein compositions. It’s a little puzzle within a puzzle, isn’t it?
Also, the detection method itself isn't perfect. Some amino acids absorb light or fluoresce differently. You might have to use extinction coefficients or relative fluorescence values to accurately compare them. It's all about fine-tuning your measurements. It’s like trying to get the perfect color balance in a photograph; you need to adjust the settings.
And the purity of your protein sample? Still a big deal. If there are other proteins or contaminants in your sample, their amino acid profiles will also show up on your chromatogram, messing with your results. So, that initial purification step is crucial. You can’t build a reliable recipe with a bunch of mystery ingredients already in the bowl.
So, your worksheet is your command center. You'll have a place to note down the hydrolysis conditions (temperature, time, acid concentration – all important details!). You'll record the peak areas for each amino acid, the standard amounts used, and any calculations you perform. You might even have a section for notes, like "Tryptophan peak looked a bit weak, might have been degraded" or "Cysteine calculation needs adjustment due to disulfide bonds." These notes are gold, trust me. They tell the story behind the numbers.
Finally, you’ll end up with a table of percentages. This is your protein’s amino acid fingerprint. You can then compare this fingerprint to known proteins. Does it look like a common enzyme? Does it resemble a structural protein? This fingerprint can give you valuable clues about the protein’s function and its role in the biological system. It’s like matching a suspect’s DNA to a database.
So, in a nutshell, determining protein amino acid composition using a worksheet is about: 1. Getting a pure protein. 2. Breaking it down (hydrolysis). 3. Separating the individual amino acids (chromatography). 4. Detecting and quantifying them. 5. Recording everything meticulously on your trusty worksheet. 6. Calculating the final percentages. 7. Interpreting the results.
It’s a bit of a journey, for sure. There are steps that require patience, a good dose of precision, and maybe a little bit of caffeine to get through. But the payoff? Understanding the fundamental building blocks of life? Pretty darn cool if you ask me. It’s not just numbers on a page; it’s the secret code of proteins, and your worksheet is the decoder ring. Now, who wants another coffee while we dive into the exciting world of amino acid side chains?
