With What Compound Will Nh3 Experience Only Ion-dipole Intermolecular Forces

Imagine you're at a party, and everyone's trying to make friends. Some people are super outgoing and hug everyone they meet – those are like ionic compounds, all charged up and ready for a big embrace. Others are a bit more reserved, maybe offering a polite handshake – these are like polar molecules, with a slight positive and negative side. Then there are the really chill folks who just kind of float around, not really connecting with anyone in particular – these are our nonpolar molecules.
Now, let's talk about our special guest at this party: Ammonia. You probably know it as NH3, that stuff that can have a pretty strong smell, like in cleaning products or even some fertilizers. Ammonia is a bit of a drama queen. It’s got this one nitrogen atom that’s hogging a bit of the electron love, making it slightly negative, and the three hydrogen atoms are a little bit positive. This makes NH3 a polar molecule. Think of it as having a little built-in magnetic attraction. It loves to get close to things that have the opposite charge, or at least a little bit of a positive or negative lean.
Ammonia, bless its pungent little heart, is always looking for a connection!
So, we're at this chemical party, and Ammonia (NH3) is looking for a dance partner. It's a polar molecule, so it's going to be drawn to things that can offer it a bit of positive or negative energy. What kind of interactions does it have? Well, if it meets another polar molecule, they might get really cozy, like a hug! That’s called a dipole-dipole interaction. If it meets an ion – like a little positively or negatively charged atom that's broken off from a bigger molecule – it’ll get super excited and wrap itself around it. That’s an ion-dipole interaction. It’s like Ammonia seeing a lonely, charged-up atom and saying, "Hey, you! You look like you need a friend!"
But here’s the interesting part, and where things get a little bit tricky and, dare we say, romantically selective. Ammonia, our dear NH3, is looking for a very specific kind of interaction to be its only form of intermolecular fun. It wants to avoid any overly enthusiastic embraces or overly clingy relationships. It wants a gentle, friendly connection, a sort of polite nod across the room, but nothing too intense. It’s like Ammonia doesn’t want to get too serious with just anyone.

What kind of compound would make Ammonia feel just right? We need something that isn’t too much of a show-off, something that doesn’t have a strong positive or negative charge all the time, but also isn’t completely aloof. Imagine our shy friend, NH3, at the party. It sees the super popular kids (ionic compounds) and thinks, "Whoa, too much energy, too much drama!" It sees the really intense couples (other strong polar molecules engaging in dipole-dipole forces) and thinks, "Hmm, a bit too intense for me right now." And it definitely doesn't want to be near the folks who are so bland they barely register, the nonpolar molecules. Those are like talking to a wall – no connection at all!
Ammonia needs a partner that offers a bit of something, but not too much. It needs someone with a subtle, gentle presence. This is where the magic of ion-dipole forces comes in, but with a twist. Ammonia itself is a polar molecule. So, it can participate in ion-dipole forces. But the question asks, with what compound will NH3 experience only ion-dipole intermolecular forces? This means we need a compound that, when interacting with NH3, generates an ion-dipole force as the primary or sole mode of attraction.
Think of it like this: Ammonia has its little positive and negative ends. It’s looking for a compound that can provide a charge, but not in a full-blown, take-over-the-world kind of way. What if we had a compound that could, under certain circumstances, offer up a positively or negatively charged part of itself, or perhaps something that readily forms ions itself, but in a way that Ammonia can respond to with its own polarity?

The key here is the only part. Ammonia is naturally polar. So, it can interact with other polar molecules through dipole-dipole forces. It can also interact with ions through ion-dipole forces. And if it finds another molecule that can form hydrogen bonds, it's all over that! But we want a situation where the only significant force is ion-dipole.
This means we need to introduce a compound that, when it meets Ammonia, creates an ion. And then Ammonia, with its inherent polarity, will be attracted to that newly formed ion. What kind of compound would do that? Something that's readily broken down into charged particles. Not just another polar molecule, because that would lead to dipole-dipole forces too. Not a nonpolar molecule, because that wouldn’t create ions.
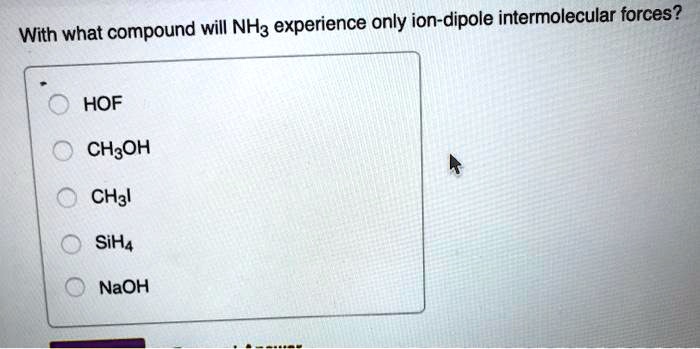
Let's consider a compound that is an ionic compound. When you dissolve an ionic compound, like table salt (NaCl), in water, it breaks apart into positively charged Na+ ions and negatively charged Cl- ions. Now, if we were to introduce these ions to Ammonia, the positive ions would attract the negative end of Ammonia, and the negative ions would attract the positive end of Ammonia. This is precisely the definition of an ion-dipole force!
So, if we had a situation where Ammonia encountered a dissolving ionic compound, the dominant intermolecular force that Ammonia would experience from the ions of that compound would be ion-dipole. The question hinges on the only aspect. In a solution of an ionic compound, where the ions are the primary interacting species, and considering Ammonia’s polarity, the ion-dipole force would be the star of the show. It's the most direct and strong attraction between Ammonia's charged poles and the free-floating ions.
It's like Ammonia walking into a room where there are lots of little charged marbles rolling around. It doesn't care about the other people at the party; it's just drawn to these little charged marbles, and it will arrange itself around them, with its positive ends facing the negative marbles and its negative ends facing the positive marbles. It’s a very specific, very focused kind of interaction, and in this scenario, the ion-dipole force is the undisputed champion of intermolecular attractions for our dear NH3!
