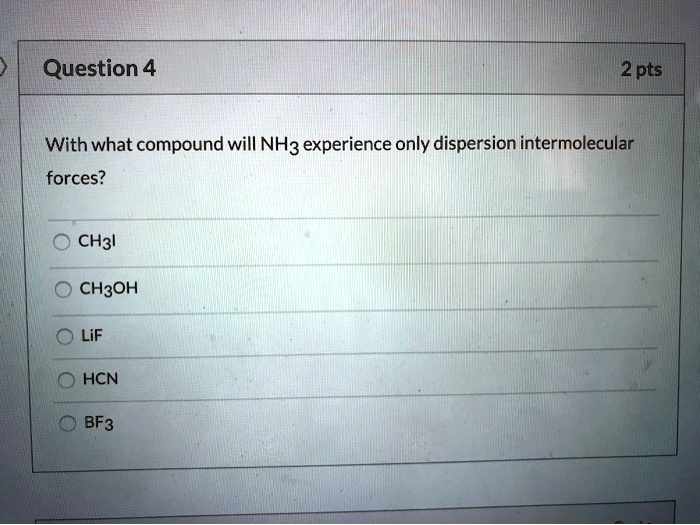
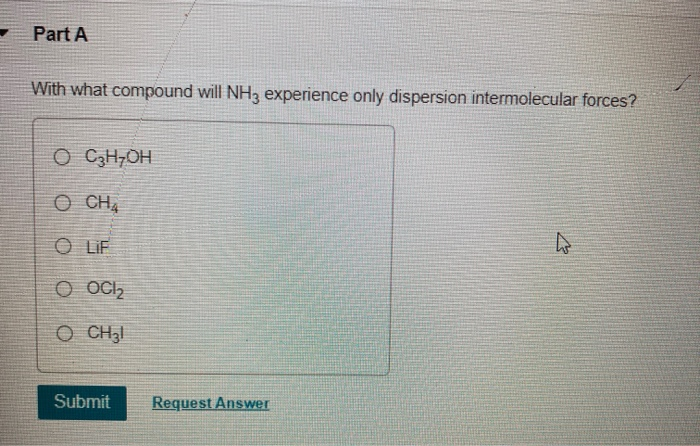
With What Compound Will Nh3 Experience Only Dispersion Intermolecular Forces
Imagine tiny invisible dancers swirling around in a big ballroom. These dancers are like the building blocks of everything we see and touch, from the air we breathe to the coffee in our mugs. They’re called molecules, and they have a special way of interacting with each other, almost like a polite little dance.
Sometimes, these molecule-dancers are a bit clingy, holding onto each other with invisible strings. These strings are called intermolecular forces. Think of it like static cling, but for molecules! They're the reason why some things are liquids, some are solids, and some just float away into the air.
Today, we’re going to meet a particularly bouncy and energetic molecule named Ammonia. We often write it down as NH3, which is like its secret code. Ammonia is super popular, it’s found in cleaning products that make your kitchen sparkle and it’s even a building block for things like fertilizers that help our yummy veggies grow.
Ammonia molecules, when they meet other molecules, can have different kinds of dances. Some dances are like a big group hug, with strong, tight connections. Others are a bit more casual, like a friendly wave. But we’re on a quest to find a special partner for our Ammonia dancer, a partner that will only engage in the most basic, most charming of dances: the dispersion force.
Now, what exactly is this "dispersion force" dance? Imagine two little dancers, each with a slightly lopsided distribution of their energetic sparkles. Sometimes, by pure chance, those sparkles might momentarily bunch up on one side of a molecule. This creates a tiny, fleeting "oops!" moment, a temporary little wobble.
When this happens, it can nudge its neighbor, making its own sparkles bunch up on the opposite side. It’s like a shy glance that makes the other dancer blush! This little chain reaction of temporary wobbles and blushes is the dispersion force. It’s the most universal of all the intermolecular forces, meaning it’s present between all molecules, but it's often the weakest one.

Our Ammonia, NH3, is a bit of a social butterfly. It loves to form those stronger dances, like the hydrogen bond, which is like a super-strong handshake. This happens because the hydrogen atoms in Ammonia are a bit “positive” and the nitrogen atom is a bit “negative,” creating a pull between them and other molecules that have similar quirks.
Think of it like having a magnet with a positive and negative end. Ammonia has these little magnetic poles within itself, making it quite attractive to other molecules that can grab onto those poles. This is what makes water so special, for example, because water molecules also have these positive and negative bits.
But we're looking for a molecule that won't do the strong dances with Ammonia. We want a partner that will only offer that gentle, fleeting dispersion force. This means we need to find a molecule that’s incredibly nonpolar. Nonpolar means it's like a perfectly balanced spinning top, with no permanent "oops!" moments of sparky bunching.

So, we need a molecule that doesn't have those slightly positive or negative ends. It needs to be perfectly even-steven, all the way through. This is where things get a little bit like a treasure hunt!
Let’s consider some possibilities. What about another Ammonia molecule? Well, they’d do the strong hydrogen bond dance, so that’s a no-go. What about something like methane (CH4)? Methane is pretty balanced, but it’s still a bit too close to Ammonia in its behavior to only have dispersion forces. There might be just enough tiny, almost imperceptible wobbles to create a very weak, but still present, interaction beyond just dispersion.
We need something truly aloof, something that just doesn't have that "personality" to engage in the more intimate dances. The perfect candidate for this exclusive dispersion-force-only relationship with Ammonia is a molecule that is both very symmetrical and composed of atoms that don't create significant charge differences.
Enter our superstar: Carbon Tetrachloride! We write it down as CCl4. Now, this might sound like something from a science fiction movie, but it’s a real thing. It looks like a little carbon atom at the center, with four chlorine atoms hanging off in a perfect, symmetrical arrangement, like a perfectly balanced star.

Because of this perfect symmetry, even though the chlorine atoms are a bit more "sparkly" than the carbon, the molecule as a whole is perfectly balanced. Imagine four friends holding hands in a circle, and they are all equally strong. No one person is pulling more than the others, so the whole circle stays perfectly still and balanced.
This makes CCl4 a nonpolar molecule. It doesn't have those handy positive and negative ends that Ammonia loves to grab onto. So, when Ammonia meets Carbon Tetrachloride, the only dance they can do is the most basic one: the dispersion force dance. It's like two strangers politely nodding to each other as they pass on the street, with no deeper connection forming.
It's a bit of a funny thought, isn't it? Our energetic Ammonia, usually so ready for a big group hug with its neighbors, has to restrain itself and only offer a fleeting, accidental nudge to its CCl4 friend. The CCl4 molecule, in turn, is just as happy to only be nudged back, its perfectly even sparkle distribution unbothered by Ammonia's energetic charm.
This is what makes chemistry so endlessly fascinating! It’s not just about complicated formulas; it’s about the personalities and the dance moves of these invisible building blocks. NH3 and CCl4, in their own unique way, show us how even the simplest of interactions can be governed by specific rules.
So, next time you’re cleaning with ammonia-based products or thinking about fertilizers, remember our bouncy friend NH3. And imagine its shy, polite encounter with its perfectly balanced friend, CCl4, engaged only in the gentlest of intermolecular dances – the dispersion force. It's a tiny, invisible ballet, happening all around us, all the time, powered by the charming simplicity of molecules.
It’s a reminder that even the most complex substances are made of these fundamental interactions, and understanding them can give us a whole new appreciation for the world. The story of NH3 and its preference for only dispersion forces with CCl4 is a sweet little anecdote in the grand book of molecular friendships.
It’s like finding out your most outgoing friend only really wants to exchange a polite smile with one particular person. It's not a rejection, but a specific kind of connection, or rather, a lack of a deeper one. And in the world of molecules, this distinction is everything.
