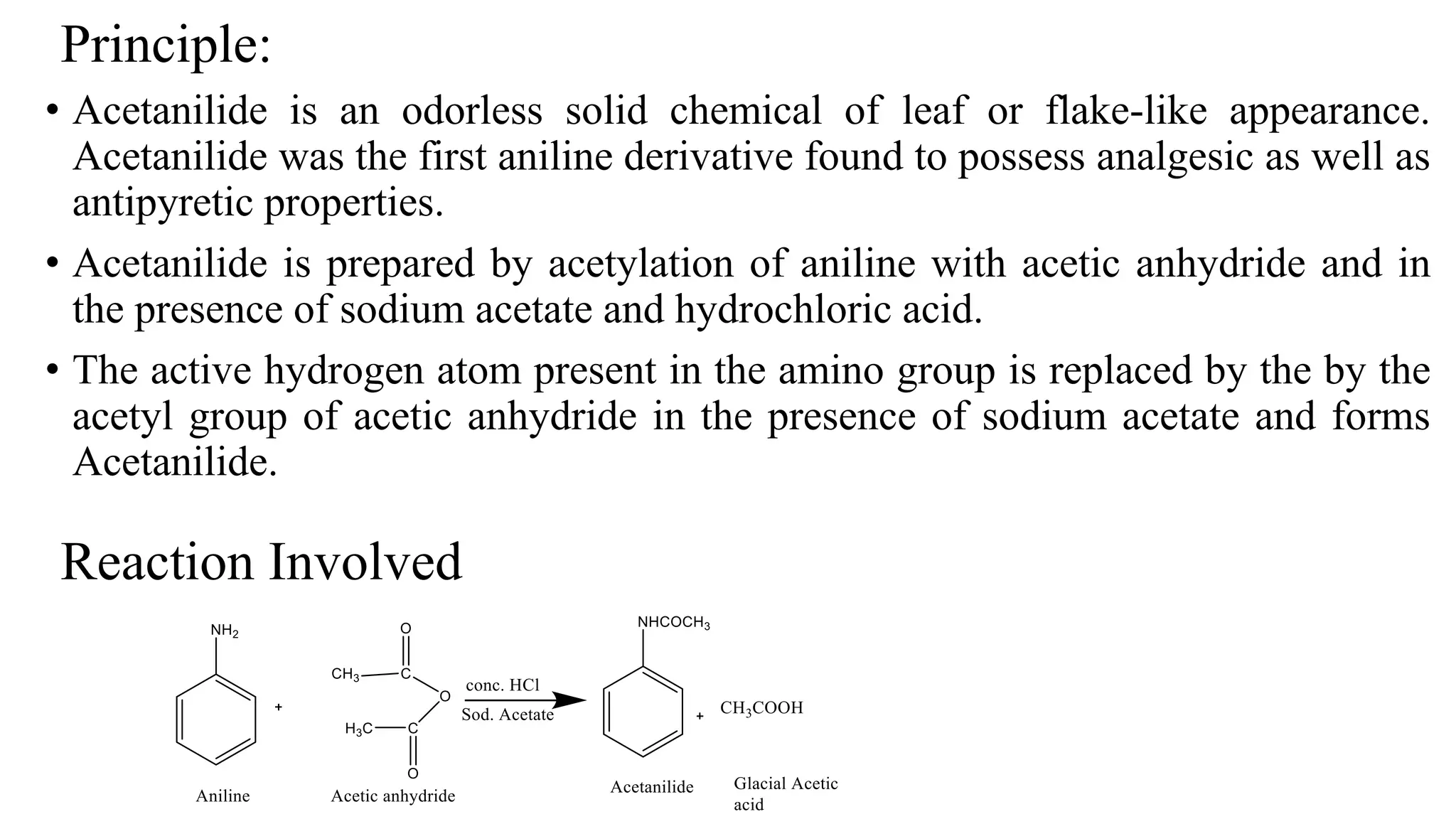
Why Is Sodium Acetate Used In Acetylation Of Aniline
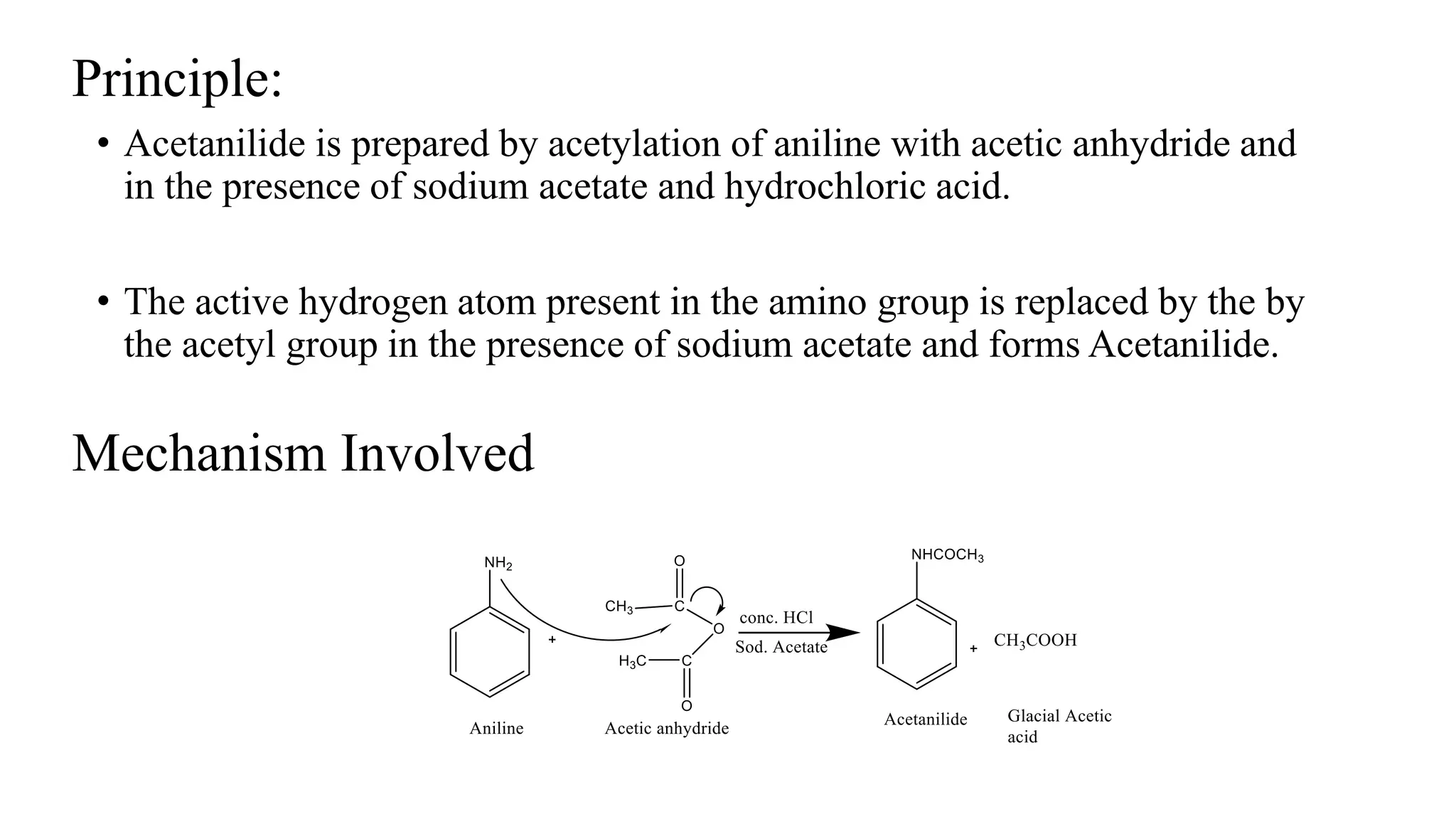
Hey there, science curious friends! Ever wondered about the cool chemistry happening behind the scenes of everyday life? Today, we're diving into something that sounds a bit fancy but is actually super neat: why sodium acetate is the go-to buddy for acetylating aniline.
Sounds like a tongue twister, right? But stick with me. This little chemical tango is actually pretty important. And guess what? It's got some fun quirks!
The "Why" Behind the Acetate Buddy
So, what's the big deal with acetylation? Think of it like giving a molecule a little makeover. We're basically attaching a little acetyl group to it. And in our case, the molecule getting the makeover is aniline. Aniline is a pretty important building block in making things like dyes, medicines, and even some plastics. Pretty cool, huh?
Now, why sodium acetate specifically? It's all about being a good wingman. When you want to acetylate aniline, you need a helper. You can't just shove that acetyl group on there willy-nilly. It needs a gentle nudge. And that's where our star, sodium acetate, shines.
Sodium Acetate: The Gentle Nudger
Sodium acetate is like that friend who always brings the snacks to the party. It’s a salt. Specifically, it's the salt of acetic acid (the stuff that makes vinegar smell… well, vinegary!). This salt form makes it super handy.
When you react aniline with something like acetic anhydride (the more common acetylating agent), things can get a bit… explosive. Okay, maybe not explosive like fireworks, but definitely vigorous. This can lead to unwanted side reactions and a messy product. Nobody wants a messy product, right?

This is where sodium acetate comes in. It acts as a buffer. Buffers are like the calm mediators of chemical reactions. They help keep the pH stable. Think of it as keeping a rowdy crowd from getting too out of control.
By controlling the pH, sodium acetate prevents the reaction from getting too acidic. And a less acidic environment means a smoother, more controlled reaction. This is key for getting a pure, clean product. We want our aniline to get its acetyl makeover, not go on a wild chemical rampage!
The "How" It Works (The Fun Bit!)
Let's get a little more specific, but still keep it fun. When acetic anhydride is used to acetylate aniline, it produces acetic acid as a byproduct. If this acetic acid builds up, it can cause problems. It can protonate the aniline (basically, add a hydrogen ion), making it less reactive and potentially leading to unwanted reactions.
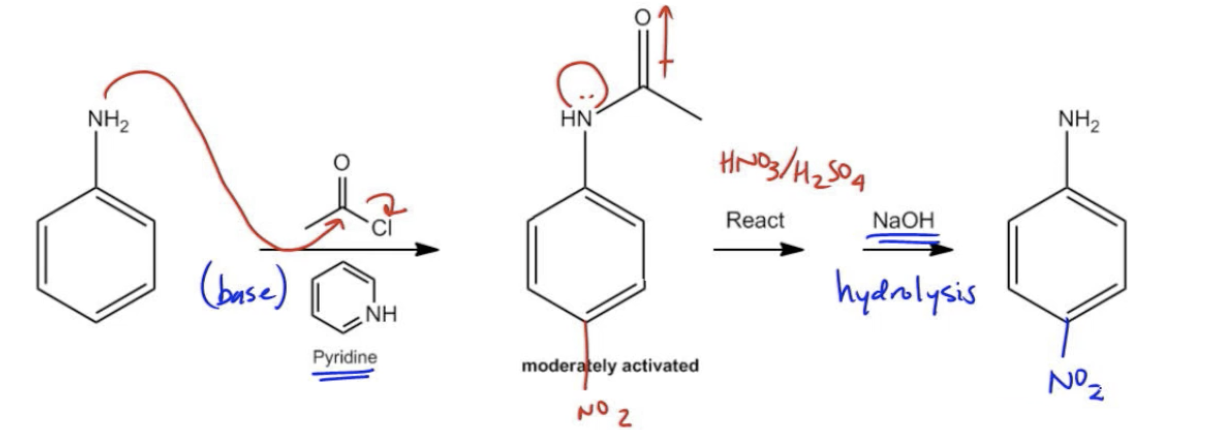
Enter sodium acetate! It's there to mop up that excess acetic acid. How? It reacts with the acetic acid, essentially neutralizing it. It forms more sodium ions and acetate ions, and the acetic acid becomes more acetic acid salt. This keeps the concentration of free acetic acid low.
Think of it like this: the acetic anhydride is like the enthusiastic party planner, and aniline is the guest of honor. The party planner is a bit too eager and keeps spilling drinks. Sodium acetate is the responsible one, quietly cleaning up the spills so the party (the reaction) can proceed without a hitch.
More Than Just a Buffer: A Catalyst Buddy?
While its primary role is buffering, some people argue that sodium acetate might have a subtle catalytic effect too. It can help to activate the acetic anhydride slightly, making it more eager to do its job. It's like giving the party planner a gentle pat on the back, saying "You're doing great, just a little bit more controlled, please!"
This dual role – buffering and potentially assisting – makes sodium acetate a really efficient and popular choice. It’s not just a one-trick pony; it’s a versatile chemical friend.
Quirky Facts and Funny Details
Did you know that sodium acetate has some other super cool uses? It’s famously used in reusable hand warmers! When you bend the little metal disc inside, it triggers a crystallization process that releases heat. It's like a tiny chemical magic trick. Pretty neat for a chemical that’s helping with aniline makeovers!
And speaking of vinegar, the smell of acetic acid can be quite pungent. So, having sodium acetate around to tame that acidity is a win for everyone involved, including your nose!
The reaction itself can be quite satisfying to watch. You add the reagents, and with the help of sodium acetate, you get a nice, clean product. It’s like watching a messy room magically tidy itself up.
Why This Topic is Just Fun to Talk About
Honestly, it's fun because it’s a little bit of mystery solved. You see a chemical name, and it sounds intimidating. But when you break it down, it’s just molecules doing their thing, and often, they have amazing helpers!
Sodium acetate isn't just some random chemical; it's a smart choice for a specific job. It’s the unsung hero of many acetylation reactions. It’s the reason why we can get high-quality products without a lot of fuss.
It reminds us that even in complex chemistry, there’s often an elegant solution, a clever way to manage the chaos. And that, my friends, is pretty darn fun to think about. It sparks curiosity and makes you wonder what other chemical friendships are out there, quietly making our world work!
So next time you hear about acetylation or see a chemical reaction happening, remember our friend, sodium acetate. It’s the silent, smiling mediator, keeping things smooth and sparkly. And that, in the world of chemistry, is a pretty big deal!
