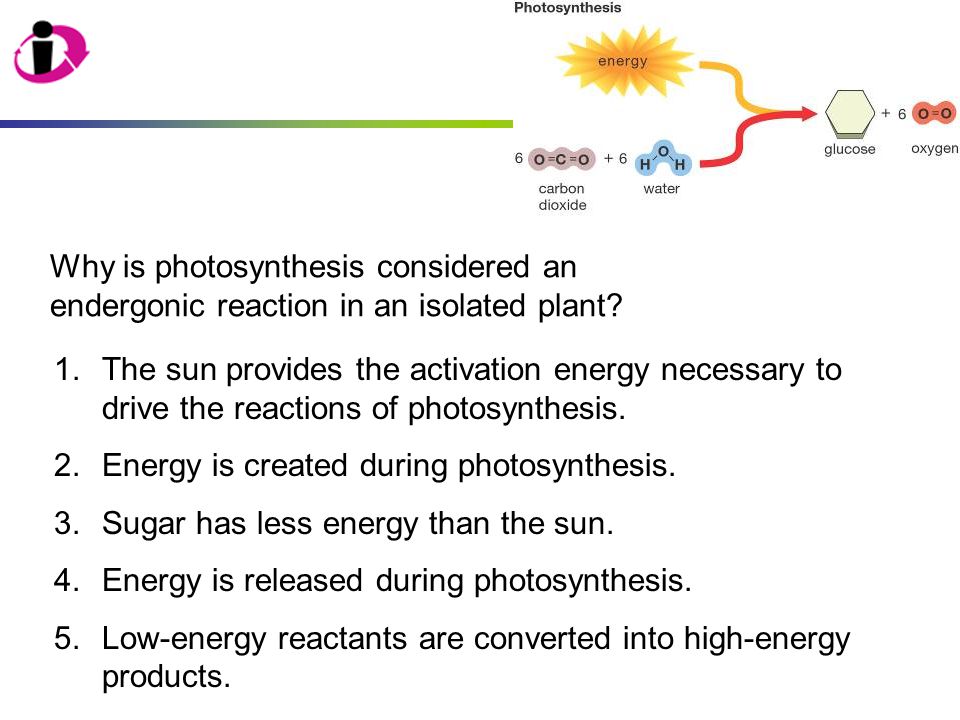
Why Is Photosynthesis Considered An Endergonic Reaction
Okay, so let's talk about plants. We all love plants, right? They give us oxygen, they make our homes look pretty, and some of them even make snacks for us. But have you ever stopped to think about how they actually do all this amazing stuff? It’s not like they have tiny little chefs in their leaves, whipping up meals. Nope. They have a secret weapon, a magical process called photosynthesis. And here's where things get a little… weird. Or at least, weird from a certain perspective. You see, photosynthesis is what scientists call an endergonic reaction.
Now, I know what you’re thinking. “Endergonic? Sounds like something a wizard would cast.” And honestly, it kind of does. But it’s not magic. It’s just… science. And this “endergonic” thing? It’s kind of an unpopular opinion in the plant world. I mean, if you ask a leaf, it’ll probably just rustle dismissively. But I’m here to tell you, with all the enthusiasm of a kid who just discovered ice cream, why photosynthesis is totally, undeniably, and hilariously endergonic.
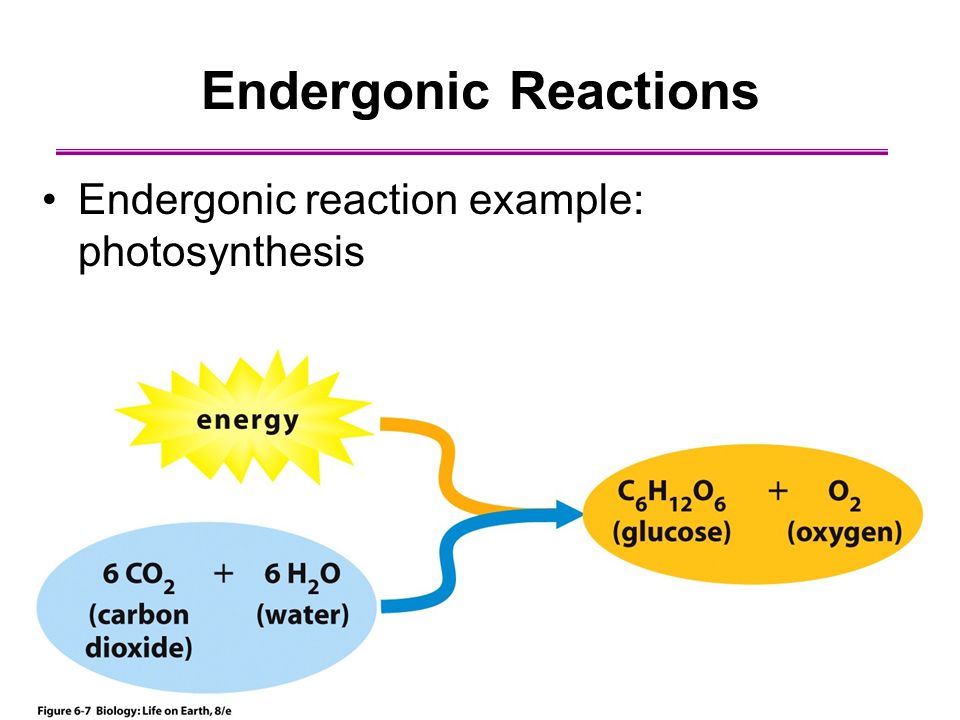
Think about it. What does an endergonic reaction mean? It means it needs energy. It’s like your phone battery. If you want it to do cool stuff, like show you cat videos or help you navigate to the nearest pizza place, you have to plug it in. It needs that jolt of electricity. Photosynthesis is kind of the same way. Plants aren't just sitting there, passively chilling, converting air and water into food. Oh no. They are working HARD. They are actively taking energy and shoving it into this whole food-making process.
Imagine a little plant, soaking up the sun. It’s not just getting a tan. It’s collecting energy. It’s like a solar-powered snack machine. But instead of accepting coins, it accepts light. And it’s not a small amount of energy either. It’s a significant amount of energy that goes into building those sugar molecules. They take simple things – carbon dioxide from the air (which is basically plant breath-out) and water from the soil (which they slurp up with their roots, like us with a giant straw) – and they smash them together.
But smashing things together without a good push? That’s not going to do much. They need that extra oomph. That’s where the sunlight comes in. Sunlight is the powerhouse. It’s the energy drink for plants. Without it, their little molecular party would just fizzle out. They capture those photons, those little packets of light energy, and they use them to break apart the water molecules and then rearrange everything to make glucose – that’s the sugar, the plant’s food. And as a bonus, they spit out oxygen. You’re welcome, us!

Now, if photosynthesis were exergonic, it would be like a spontaneous, energetic burst. Things would just happen, and energy would be released. Think of a log on a campfire. You light it, and poof, heat and light fly everywhere. It’s a one-way ticket to energy liberation. But plants? They're not burning anything. They're building. And building things, especially complex molecules like sugar, requires a lot of effort. It's like trying to build a Lego castle. You can't just wish it into existence. You have to put the bricks together, one by one, and that takes energy.
So, next time you see a leafy green friend, give it a little nod of appreciation. It's not just lounging around. It's a tiny, sun-powered, energy-guzzling food factory. And that, my friends, is the beauty of an endergonic reaction. It’s all about the hustle.
Chapter 6 – Energy flow in the life of a cell - ppt download
And that’s why I think the whole “endergonic” label is just so fitting. It’s a little bit of an underdog term, isn’t it? Most people think of energy as something being given off. Like a loud noise or a bright flash. But photosynthesis is the opposite. It’s about taking in energy, storing it, and using it to create something amazing. It’s the quiet achiever of the chemical world. It’s the student who studies diligently, takes copious notes, and then aces the exam, rather than the one who just shouts out the answers.
Think of it like this: When you’re really hungry, you don’t just magically become full. You have to eat food. That food has stored energy. You consume it, your body breaks it down, and then you get the energy. Plants are doing the reverse, in a way. They are taking raw ingredients and creating the energy-storing molecules. They are the ultimate chefs, and their kitchen runs on sunshine.
So, yeah, photosynthesis. Endergonic. It means it needs energy to happen. It’s like a perpetual motion machine… that you have to keep plugging in. But the outcome? Pure awesomeness. It feeds itself, it feeds other things (hello, salads!), and it keeps our air breathable. All thanks to a little bit of light, a lot of water, some carbon dioxide, and a whole lot of endergonic effort. It’s a beautiful, energy-demanding, life-sustaining masterpiece. And I, for one, am here for it. It’s a reminder that sometimes, the most impressive feats require us to actively take energy, not just give it away. It’s the quiet, persistent work of building something wonderful. And that, my friends, is a kind of magic all its own.