Why Is Elimination Favored At High Temperatures
Hey there, science adventurers! Ever wondered why some reactions just love to get a little toasty? You know, like when you're cooking and things start to bubble and change, or when you see something rusting away a bit faster on a hot summer day? Well, there's a cool reason behind it, and it all boils down to something called "elimination". Sounds a bit dramatic, right? Like we're getting rid of something. And in a way, we are!
Think of it like this: imagine you have a bunch of friends hanging out, and some of them are holding onto each other really tightly. Now, if you start playing some really energetic music and crank up the temperature, those friends are going to start bouncing around a lot more, right? They've got way more energy. And sometimes, with all that extra jiggle and wiggle, they might just let go of each other and go do their own thing. That's kind of what happens in chemistry.
At its heart, elimination is about a molecule shedding a piece of itself, or even two bits, to become something new. It's like a caterpillar deciding it's had enough crawling and needs to become a butterfly. Pretty neat transformation, isn't it? But why does this transformation suddenly get a VIP pass when the temperature goes up?
The Speedy Shuffle
So, what's the deal with heat? Well, heat is basically just a measure of how much things are moving and vibrating. The hotter it gets, the more energetic those tiny atoms and molecules become. They're like toddlers who've had too much sugar – they just can't stay still!
In chemistry, this increased energy means that molecules are bumping into each other more often and with more force. It's like a crowded party where everyone's really excited. With all this commotion, chemical bonds that might be pretty stable at room temperature can start to break. It’s as if the "rules" of staying connected get a little looser when the energy levels are through the roof.
Letting Go to Grow
Now, when we talk about elimination reactions, we're often thinking about specific types of molecules. Imagine a molecule that has a few "attachment points" or groups hanging off its main structure. In an elimination reaction, these groups are kicked off. This often happens when two nearby atoms decide to ditch their own little buddies.

Think of a molecule like a team playing a game. Sometimes, two players on opposite ends of the field might decide to leave their positions and run towards the center, leaving gaps behind. In elimination, these "gaps" are what allow the molecule to form a new kind of bond, often a double or triple bond. It's like the molecule is saying, "Okay, you take that guy, and I'll take that other one, and we can link up in a new way!"
Why is this favored at high temperatures? Because breaking those existing bonds and forming new ones requires a certain amount of "oomph," or activation energy. It's like needing a good push to get over a small hill. Heat provides that push! The more energy you give the system, the more likely these molecules are to have enough energy to overcome the barrier and undergo the elimination.
The Competition: Substitution vs. Elimination
But here's where it gets really interesting. Elimination isn't the only show in town when molecules interact. There's another popular act called "substitution". In substitution, one group on a molecule is swapped out for another. Think of it like replacing one player on a team with another. It's a neat trick, but it’s a different kind of dance.

At lower temperatures, substitution reactions often have the upper hand. Why? Because substitution reactions can sometimes be a bit more "gentle." They don't always require the same amount of energetic jostling to get going. It’s like a polite handshake where one person offers their hand and the other takes it.
However, as you crank up the heat, elimination starts to steal the spotlight. It’s like the energetic music kicks in, and the polite handshake turns into a full-blown dance-off. The increased energy makes it much more probable for those bonds to break and for the molecule to rearrange itself through elimination. The molecules are just too buzzed and energetic to stick to the slower, more controlled substitution.
It's a bit like when you’re trying to get through a crowded doorway. If people are moving slowly and politely, you can substitute (squeeze through) more easily. But if everyone is rushing and excited, the most likely thing to happen is people getting shoved aside and creating a bigger opening – that’s elimination!
The Thermodynamics Tango
Beyond just raw energy, there's another layer to this hot preference: thermodynamics. Don't let the fancy word scare you! It's just about how stable things are and how they like to settle down.
At higher temperatures, systems tend to favor reactions that produce more "disorder" or entropy. Think of your bedroom after a week of not cleaning – it's much more disordered! In chemistry, elimination reactions often lead to the formation of smaller molecules or molecules with more freedom to move. This increases the overall disorder of the system, which is a good thing for the system at higher temperatures.
It’s like letting a bunch of balloons out into the sky. They’ll all spread out and float around independently, creating a lot of disorder. Substitution, on the other hand, is more like carefully arranging those balloons in a pattern – much more ordered!
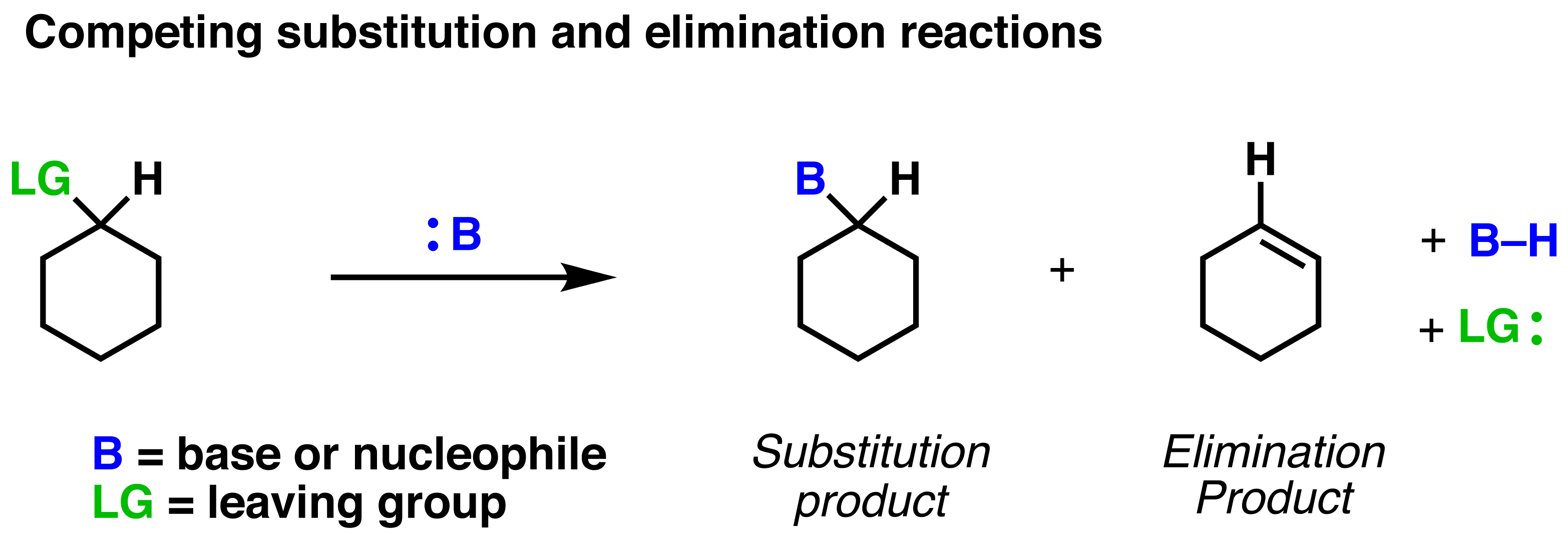
So, as the temperature rises, the universe is basically saying, "Let's embrace the chaos!" and elimination reactions are the perfect way to do it. They're the spontaneous spread of those balloons, creating more freedom and less order, which is what the system craves when it's got plenty of energy.
Practical Perks of High Heat
This isn't just some abstract chemical concept. It has real-world implications! Many industrial processes rely on controlling these reactions. For example, when we make plastics or certain fuels, we often need to encourage elimination reactions to happen.
Heating things up is a common way to speed up reactions and get the desired products. It’s like using a powerful hairdryer to dry your hair quickly versus just letting it air dry. You get the job done faster!
So, next time you see something reacting with gusto when it gets hot, remember the cool science behind it. It's the energetic molecules, the preference for disorder, and the sheer power of heat making elimination the star of the show. It’s a reminder that sometimes, a little heat and a lot of energy are exactly what you need to make something new and exciting happen!
