Why Does Hf Have A Higher Boiling Point Than Hcl

Hey there, science curious folks! Ever found yourself wondering about the tiny, invisible world of molecules? It’s kind of like a miniature universe, full of its own rules and quirks. Today, we’re going to dive into something that might sound a bit technical at first, but stick with me, because it’s actually super fascinating: why does Hydrogen Fluoride (HF) have a way higher boiling point than Hydrogen Chloride (HCl)?
Seriously, it's a pretty big difference. Imagine trying to boil water (which is H2O, by the way) and then trying to boil something like, say, methane (CH4). The amount of heat you’d need for methane would be way less, right? That’s kind of the ballpark we’re talking about here, but the reason is more specific and, frankly, pretty cool.
So, let's break it down. We're comparing two molecules that look pretty similar on the surface. Both are made of one hydrogen atom bonded to a halogen atom. Halogens are a group of elements on the periodic table known for their… well, let's just say they like to grab electrons. Think of them as the electron magnets of the periodic table.
In HF, we have hydrogen and fluorine. In HCl, we have hydrogen and chlorine. Both of these are gases at room temperature, and to turn them into liquids, you need to cool them down. But here's the kicker: to get HF to become a liquid, you need to cool it down a lot more than you need to for HCl. And then, to turn that liquid into a gas (boil it), HF needs a whole lot more energy – or heat – to make it happen.
Why the fuss? It all comes down to how these molecules behave when they're hanging out together. You see, molecules, even though they’re tiny, have forces that pull them towards each other. These are called intermolecular forces. Think of them as tiny molecular hugs or static cling between atoms. The stronger these hugs are, the more energy it takes to pull the molecules apart, and that means a higher boiling point.
Now, the key difference between HF and HCl lies in their halogen atoms: fluorine and chlorine. Fluorine is the undisputed champion of electron-grabbing. It’s the most electronegative element on the entire periodic table. Imagine it as the ultimate magnet, pulling electrons towards itself with all its might. Chlorine is also pretty good at this, but it's not quite the same level of… intensity.

The Power of the Polar Bond
When hydrogen bonds with fluorine in HF, that electron-grabbing power of fluorine is in full effect. This creates a very, very polar bond. What does polar mean? Think of a tiny battery. One end is a little bit negative, and the other end is a little bit positive. In HF, the fluorine end becomes significantly negative because it’s hogging all the electrons, and the hydrogen end becomes significantly positive.
Now, with HCl, chlorine is also electronegative, so the bond is polar too. But fluorine is just so much more electronegative. So, the polarity in HF is much stronger than in HCl. This means the little "battery" of HF is much more charged up!
The Super-Duper Hug: Hydrogen Bonding
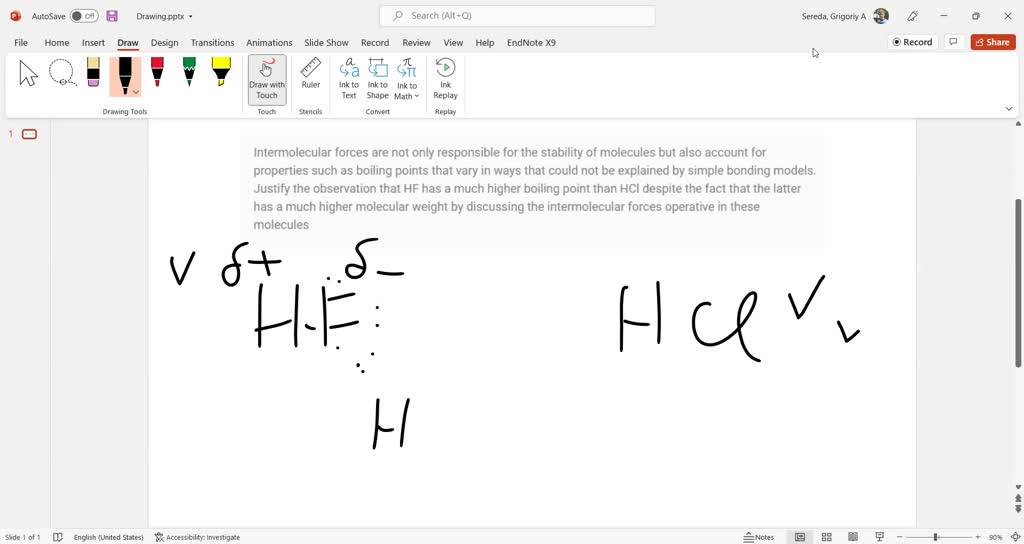
This is where things get really interesting and where the magic happens for HF. Because the hydrogen in HF is so positively charged (thanks to that super-electronegative fluorine), it’s like a tiny beacon, looking for something negative to attract. And guess what’s negative? The fluorine atoms in other nearby HF molecules!
This attraction between the positive hydrogen of one HF molecule and the negative fluorine of another HF molecule is called hydrogen bonding. It’s a special, super-strong type of intermolecular force. Imagine it as a really, really strong handshake between molecules, much stronger than a regular handshake.
HCl molecules also have attractions between them, but they’re not as strong. They have what we call dipole-dipole interactions. It's like a weaker handshake, or maybe just a friendly wave from afar. The hydrogen in HCl isn't as positive because chlorine isn't as negative as fluorine. So, the attraction between HCl molecules is less intense.
And because hydrogen bonding is so much stronger than the dipole-dipole interactions in HCl, it takes a lot more energy to break those HF "super-hugs" and separate the molecules. That’s why HF has that impressively higher boiling point. It’s literally resisting being turned into a gas because it’s so strongly bonded to its neighbors.
Think of it like this…
Imagine you have a bunch of kids playing.
With HCl, it's like they're all holding hands loosely. If you want to get them to scatter and run around (like becoming a gas), it doesn't take much effort. A gentle nudge is enough.
With HF, however, it's like they're all in a giant, super-tight group hug. To get them to break apart and run around, you need to put in a lot more energy. You have to really pull and push to get them to let go of each other.
Another fun way to think about it is like sticky notes.

HCl molecules are like regular sticky notes. They might stick together a bit, but you can easily peel them apart.
HF molecules are like those super-strength, industrial-grade sticky notes that you need a pry bar to remove. They’re just that much stickier.
So, the next time you hear about boiling points or intermolecular forces, remember the story of HF and HCl. It’s a fantastic example of how the subtle differences in atomic properties, like electronegativity, can lead to dramatic differences in a substance's behavior. It’s all thanks to that powerful, unique bond that fluorine can form with hydrogen – a bond strong enough to make HF quite a bit more stubborn when it comes to boiling!
Isn't it neat how much chemistry is going on, even in seemingly simple molecules? The universe is full of these little puzzles, and figuring them out is pretty rewarding. Keep wondering, keep exploring!
