Why Does Fluorine Have A Smaller Atomic Radius Than Chlorine

Hey there, curious minds! Ever stop to think about the tiny building blocks of everything around us? I'm talking about atoms, those minuscule marvels that make up, well, everything. Today, we're going to dive into a little atomic drama, a tale of two halogens: fluorine and chlorine. And trust me, it’s more exciting than you might think!
Now, you might be wondering, “Why on earth would I care about the atomic radius of fluorine versus chlorine?” And that’s a fair question! But stick with me, because understanding these little differences can actually unlock a whole new way of seeing the world. Think of it like understanding why a hummingbird flits so much faster than a pigeon – it’s all in the details, right?
So, let’s get down to brass tacks. We're looking at two elements from the same family, the halogens. They're like cousins, sharing a lot of similar traits. They both love to grab an electron, and they both have that distinctive, sometimes feisty, personality. But here’s the kicker: fluorine is smaller than chlorine. Yep, you heard me. The tiny one is actually tinier than its slightly bigger sibling!
The Nucleus: The Heart of the Atom
To get to the bottom of this, we need to zoom way, way in. Picture an atom. At its center, you’ve got the nucleus, a bustling hub of positively charged protons and neutral neutrons. Whizzing around this nucleus are negatively charged electrons, like tiny planets orbiting a sun. The atomic radius is basically a measure of how big that "solar system" is – from the center of the nucleus to the outermost electron.
Now, fluorine and chlorine are both in the same column of the periodic table. This means they have the same number of "shells" or energy levels for their electrons. So, why the size difference? It all boils down to the number of protons in the nucleus.
Proton Power!
Fluorine, the little powerhouse, has 9 protons in its nucleus. Chlorine, its beefier counterpart, boasts 17 protons. More protons mean a stronger positive charge at the center of the atom. Think of it like a super-magnet!
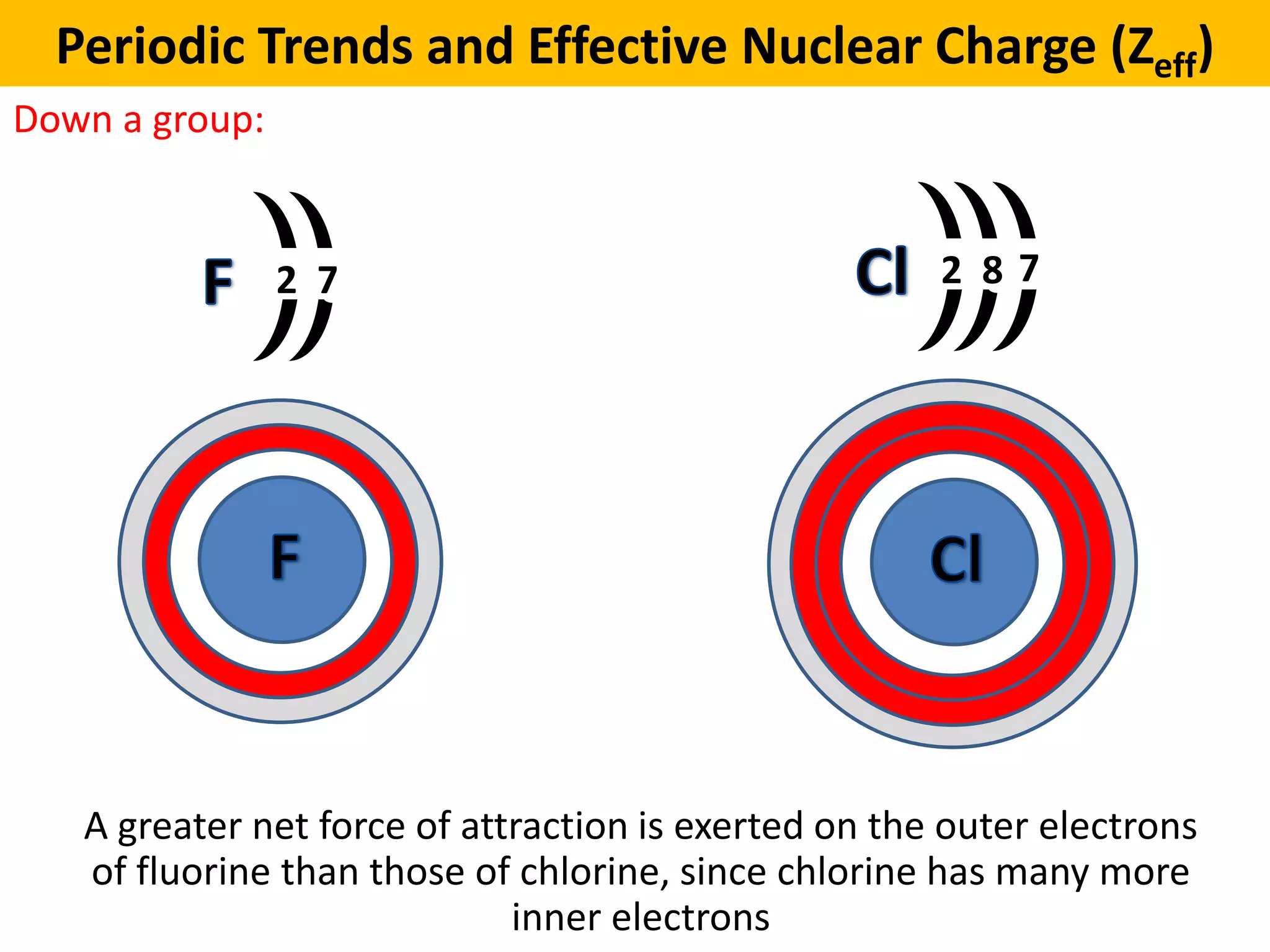
This extra "pull" from the larger number of protons in chlorine's nucleus is what we chemists (and now, you!) call effective nuclear charge. It’s like a stronger grip on those outer electrons.
Imagine you have two magnets. One is a small, strong fridge magnet, and the other is a much larger, more powerful industrial magnet. If you tried to pull a paperclip away from both, the paperclip would be much harder to dislodge from the industrial magnet, right? It's the same principle with atoms!
Electrons Get Squeezed!
Because fluorine has fewer protons (a weaker "magnet"), its outer electrons are not held as tightly. They can spread out a bit more, giving fluorine a larger atomic radius. But wait, didn't I just say fluorine is smaller? Ah, the wonderful trickery of chemistry! Let's clarify.
Here's where the confusion sometimes happens, and it's totally okay. When we compare fluorine to elements in the same row of the periodic table, it gets smaller as you move from left to right because of the increasing nuclear charge. But when we compare fluorine to chlorine, which is below it in the same column, the number of electron shells is actually the same. The crucial difference is the number of protons and the resulting pull on the electrons.

So, let's rephrase: Fluorine has 9 protons, and its electrons are in the first and second shells. Chlorine has 17 protons, and its electrons are also in the first and second shells. BUT! The 17 protons in chlorine's nucleus create a much stronger pull on its outermost electrons than the 9 protons in fluorine’s nucleus. This stronger pull effectively shrinks the space occupied by chlorine's electron cloud compared to what you might expect from simply adding another electron shell.
It's like having a very energetic toddler (fluorine) with a firm but gentle hand holding their balloon, versus a slightly older, more boisterous child (chlorine) who's got a much tighter, almost vice-like grip on their balloon. The older child’s balloon might appear a bit more compressed, even if the string is technically the same length!
So, it's not about adding more shells in this specific comparison. It’s about the intensity of the nuclear attraction. Chlorine, with its more powerful nucleus, is essentially squeezing its outermost electrons closer to itself. This results in a smaller atomic radius for chlorine relative to what its position in the periodic table might initially suggest if you only considered electron shells. And fluorine, with its less intense nuclear pull, has a slightly larger atomic radius in this direct comparison. This is a nuanced point, but it’s the beauty of understanding the underlying forces!

Why This Little Difference Matters (More Than You Think!)
Now, why should you care about this tiny difference in size? Because these atomic dimensions have a huge impact on how elements behave! Fluorine, being so small and having that intense nuclear pull, is the most electronegative element on the periodic table. Electronegativity is basically an atom's "greediness" for electrons. Fluorine is the ultimate electron-hoarder!
This makes fluorine incredibly reactive. It's like a tiny, super-charged athlete ready to jump into action. It readily forms strong bonds with other atoms, and this reactivity is what makes it so useful in so many applications!
Think about Teflon, the non-stick coating on your pans. That’s made using fluorine compounds! Or toothpaste that fights cavities – yep, there's fluoride in there, which is a form of fluorine, working its magic to protect your teeth. It’s all thanks to fluorine’s super-powered, small-but-mighty nature.
Chlorine, while also reactive, is a bit less intense than fluorine. It’s still a significant player, used in things like water purification (keeping our swimming pools sparkling and our drinking water safe!) and in the production of PVC plastics.

The subtle difference in their size and electron attraction dictates their exact roles and strengths in chemical reactions. It’s like the difference between a precision scalpel (fluorine) and a sturdy, reliable multi-tool (chlorine) – both incredibly useful, but for slightly different tasks.
The Joy of Discovery!
Isn't it amazing? All of this, from the tiny size of an atom to its ability to make our lives better, stems from fundamental principles of physics and chemistry. Learning about these things isn't just about memorizing facts; it's about understanding the hidden architecture of the universe.
Every time you brush your teeth, cook a meal, or even just breathe, you're interacting with the consequences of these atomic properties. It’s a constant, beautiful dance of attraction and repulsion, size and charge, all playing out on a scale we can't even see!
So, the next time you hear about atomic radii or the periodic table, don't shy away. Dive in! Explore! Because the more you learn about these incredible building blocks, the more you'll realize how much there is to be inspired by. The world is a fascinating place, and the journey of understanding it, one atom at a time, is an adventure that’s truly out of this world. Keep asking questions, keep exploring, and never stop being amazed by the wonders of science!
