Why Does Fluorine Have A Higher Ionization Energy Than Iodine

Imagine a big, happy family of elements, all living together in their own little houses on the periodic table. Some are big and cuddly, others are small and feisty. Today, we're going to peek into the homes of two very interesting characters: Fluorine and Iodine.
Now, these two are related, sort of. They're both members of the same family, the halogens. Think of them as cousins who share a similar grandparent, but they've grown up in very different environments.
Fluorine is like the tiny, energetic toddler of the halogen family. He's practically bursting with excitement, always wanting to be the center of attention. He's so eager, in fact, that he practically clings to his electrons with all his might.
On the other hand, Iodine is more like the wise, gentle elder. He's much larger, with more space and fewer close neighbors. He's seen a lot in his elemental life and is a bit more relaxed about his electrons.
So, what's this "ionization energy" business we're talking about? Think of it like trying to convince someone to share their favorite toy. Ionization energy is basically the 'persuasion power' it takes to snatch an electron away from an atom. The higher the ionization energy, the harder it is to convince that atom to let go of its electron.
Now, here's where things get a little surprising. You might think that the bigger, more spread-out Iodine would be easier to convince to give up an electron, right? After all, his electrons are a bit further from his core. But nope, that's not quite how it works!
Fluorine, our little dynamo, has an incredibly high ionization energy. It's like trying to pry a favorite teddy bear from a toddler's grip – it takes a LOT of effort!
Why is this feisty little Fluorine so darn attached to his electron? It all comes down to his size and the way his electrons are packed. He's so small, and his outer electron is so close to his nucleus (the "heart" of the atom), that the pull is super strong.

Imagine a tiny magnetic ball. If you bring a metal marble very close, it sticks like superglue. That's kind of like Fluorine and his electron. The attraction is intense!
Iodine, on the other hand, is a much bigger atom. Think of him as a much larger bouncy castle. His outer electrons are further away from his nucleus, and there are more "shields" in between.
These shields are actually the inner electrons. They act like a crowd of excited kids around a popular celebrity. They can somewhat block the "fan mail" (the attraction from the nucleus) from reaching the outer electrons as strongly.
So, while Iodine's outer electrons are further away, making the distance factor seem like it should be easier, the shielding effect and the sheer size of the atom means the nucleus doesn't have quite the same "grip" as it does on Fluorine's electron.
It's a bit like trying to get a cookie from a small child standing right next to you versus a child on the other side of a crowded playground. Even though the child on the other side is further, all the people in between might make it harder to reach them.
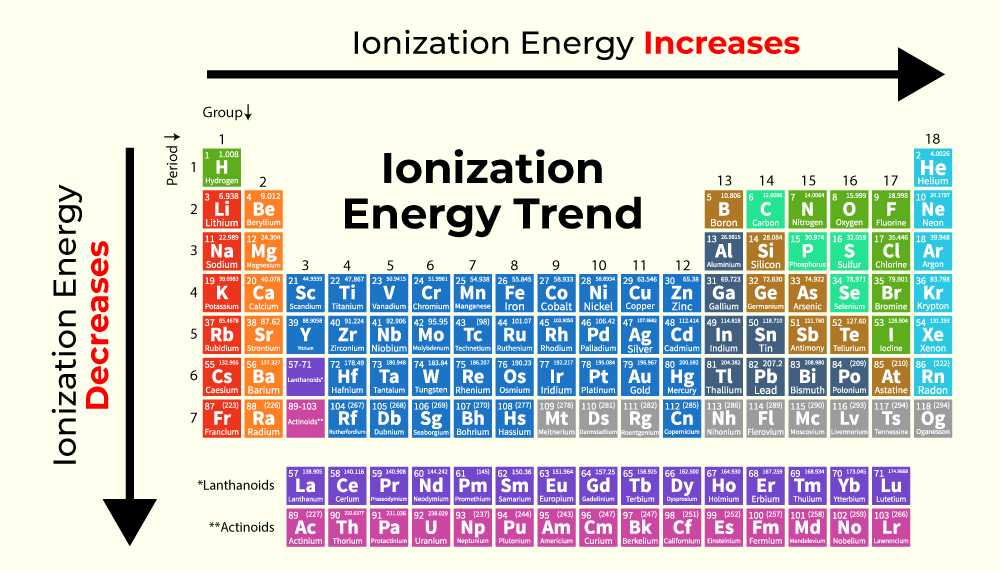
Fluorine's small size means his nucleus has a very direct and powerful influence on his single outer electron. There's no buffer, no significant shielding. It's a one-on-one, super-tight connection.
This intense attraction is why Fluorine is known for being the most reactive element on the periodic table. He's so eager to attract other atoms to "borrow" or "share" electrons that he's always ready for action. He's the life of the elemental party!
Think of it this way: Fluorine is like a tiny, powerful magnet that's desperate to connect. He'll grab onto anything he can to make that connection. This is why when you encounter Fluorine, it's often in compounds where it's already bonded with other elements, like in toothpaste to keep your teeth strong.
Iodine, while still reactive, is a bit more mellow. He's like a strong magnet, but he's a bit further out. He still has a good pull, but it's not the same all-consuming grip that Fluorine has.
This difference in ionization energy has huge implications for how these elements behave and the kinds of substances they form. It's the silent driver behind a lot of chemistry.
So, the next time you see Fluorine in your toothpaste or Iodine in your salt, remember the story of these two elemental cousins. One, the tiny, fiercely protective toddler, and the other, the more spacious, seasoned elder.
It's a beautiful example of how size and arrangement within the atomic family can lead to such dramatic differences in personality and behavior. The universe is full of these fascinating quirks, and even something as simple as ionization energy has a delightful story to tell!
It's like the difference between a puppy that never wants to let go of its favorite squeaky toy and an older dog who might nudge a ball your way occasionally. Both love their toys, but their intensity and how they hold onto them are different.
And isn't it wonderful that even these minuscule building blocks of our world have such unique and surprising characteristics? It makes you appreciate the complexity and elegance of everything around us, from the smallest atom to the largest star.
So, while Fluorine might seem like a troublemaker with his high ionization energy, it's actually a sign of his incredible eagerness to engage with the world. And Iodine's slightly less intense pull is just a reflection of his larger, more diffused presence.

It's a cosmic ballet of attraction and repulsion, all dictated by these fundamental properties. And it all starts with understanding that sometimes, the smallest packages hold the most powerful energy!
Think of it as a fundamental rule of the elemental universe: sometimes, the closer you are to the source of power, the more fiercely you hold on. And Fluorine is the ultimate champion of that rule!
It’s a fun reminder that even in the seemingly abstract world of chemistry, there are relatable stories and understandable reasons behind why things are the way they are. And that’s what makes learning about these elements so engaging and, dare we say, heartwarming!
So next time you think about Fluorine, imagine that tiny, energetic atom with a grip so strong, it's practically a superpower. And for Iodine, picture a more expansive presence, still with a significant influence, but in a gentler, more distributed way.
The periodic table is truly a treasure trove of such delightful comparisons and intriguing facts. It’s a testament to the intricate beauty of nature, where even the smallest differences can lead to profound consequences.
And the tale of Fluorine and Iodine's ionization energies is just one of many amazing stories waiting to be discovered within its rows and columns.
