Why Can't Kinetic Energy Ever Be Greater Than Potential Energy

Hey there, science enthusiast (or just someone who stumbled upon this page looking for answers to life's big questions, like "why can't my toast always land butter-side up?"). Today, we're diving into a topic that might sound a little intimidating: kinetic and potential energy. But don't worry, we're going to break it down in a way that's as easy as, well, watching a ball roll down a hill!
So, you've heard of these energy things, right? Potential energy is like the promise of energy, the stored-up goodness waiting to happen. Think of a kid perched at the top of a slide – they've got loads of potential energy! Kinetic energy, on the other hand, is the energy of motion. Once that kid starts sliding, whoosh, they've got kinetic energy. Pretty straightforward so far, huh?
Now, the million-dollar question (or, more accurately, the joule-dollar question): why can't kinetic energy ever be greater than potential energy? It’s like asking why you can’t have more fun at a party before it even starts. Seems a bit counterintuitive, right? Let's get our metaphorical lab coats on and figure this out.
The Grand Unified Theory of Energy (Well, Kind Of)
Our journey starts with a super important concept called the Law of Conservation of Energy. This is basically the universe's golden rule for energy, and it's a real buzzkill for anyone trying to cheat the system. It says that energy can't be created or destroyed; it can only be transformed from one form to another.
Think of it like this: energy is like LEGO bricks. You can build a castle, a spaceship, or a wobbly tower with them, but you always end up with the same number of bricks. You can't magically get more bricks out of thin air, and you can't make them disappear into another dimension (unless you're a magician, which is a whole other topic). Energy works the same way. It just changes its outfit, going from potential to kinetic and back again.
So, if the total amount of energy in a system stays the same, and we're just swapping between potential and kinetic, how can one ever get bigger than the other? This is where things get really interesting.
The Ball Dropping Bonanza!
Let's use our classic example: dropping a ball. Imagine holding a ball up in the air. At this moment, it has a ton of potential energy. Why? Because gravity is pulling it down, and if you let go, that pull will do something. It has the potential to move.

Now, you let go. Plop! As the ball starts to fall, its potential energy begins to decrease. Where does it go? Aha! It's transforming into kinetic energy. The faster the ball falls, the more kinetic energy it has. It's like the ball is trading its stored-up energy for the thrill of speed.
At the very moment the ball hits the ground (or just before, let's be precise!), its potential energy will be at its absolute minimum (close to zero if we consider ground level as our reference point). And, if everything were perfectly ideal (no air resistance, no bouncy floor), all of that initial potential energy would have been converted into kinetic energy. So, at that peak moment of falling, the kinetic energy is at its maximum, and it's equal to the initial potential energy.
This is key: when potential energy is at its lowest (near zero), kinetic energy is at its highest (equal to the initial potential energy). And when potential energy is at its highest (at the start), kinetic energy is at its lowest (zero, because it's not moving yet).
The "What If" Scenario: The Energy Cheat Code?
So, what if we could have kinetic energy greater than potential energy? What would that even look like? Imagine our falling ball suddenly going faster than it should, more energetic than it was when it was sitting up there. Where would that extra energy come from?
It would have to be pulled out of nowhere, violating our precious Law of Conservation of Energy. It's like trying to spend money you don't have – the universe has a way of saying, "Nope, not today, pal!"

This is why, in a closed system (meaning no energy is coming in or going out from the outside world), the total mechanical energy – which is the sum of potential and kinetic energy – remains constant. Potential Energy + Kinetic Energy = Constant Total Energy.
If kinetic energy increased beyond the initial potential energy, it would mean the total energy of the system also increased, which, as we’ve established, is a big no-no in physics class. It's like trying to have your cake and eat it too, and then somehow magically conjure another cake out of thin air. The universe just doesn't work that way.
The Not-So-Ideal World: Why It's Even Less Likely
Okay, so in a perfect, magical world with no friction or air resistance, kinetic energy can at best equal the initial potential energy, right at the point of maximum speed. But in our real world, things are a little messier. And this mess actually makes it even harder for kinetic energy to reach its theoretical maximum.
Think about our falling ball again. As it zips through the air, it's encountering air resistance. This is like a gentle (or not-so-gentle) pushback from the air molecules. This air resistance does work on the ball, and this work converts some of the ball's kinetic energy into heat. Yep, that’s right, the ball gets a tiny bit warmer as it falls!

Also, if the ball hits something that’s not perfectly rigid, like the ground, some energy is lost to sound (that thud!) and deformation of the ball and the surface. It's like the universe is saying, "Okay, you had a good run, but now it's time to spread that energy around a bit."
So, in the real world, at any point in its fall, the kinetic energy of the ball will actually be less than the potential energy it started with. Some of that potential energy has already been converted into other forms of energy – heat, sound, etc. – before it even gets a chance to become maximum kinetic energy.
This means that in our messy, wonderful reality, the kinetic energy is always less than or equal to the initial potential energy. It’s never greater. It can equal it in a perfectly idealized scenario at the lowest point of potential, but in reality, it's always a little bit less due to energy losses.
Beyond the Ball: Other Energy Shenanigans
This principle isn't just for falling balls! It applies to all sorts of things. Think about a pendulum swinging. At the highest point of its swing, it has maximum potential energy and zero kinetic energy. As it swings down, potential energy converts to kinetic energy. As it swings up the other side, kinetic energy converts back to potential energy.
If you've ever seen a pendulum swing for a long time, you might think, "Hey, it's going pretty high on the other side!" But over time, you'll notice it doesn't swing quite as high as it did initially. That's because of air resistance and friction at the pivot point, which are constantly taking little sips of energy from the system, turning it into heat.
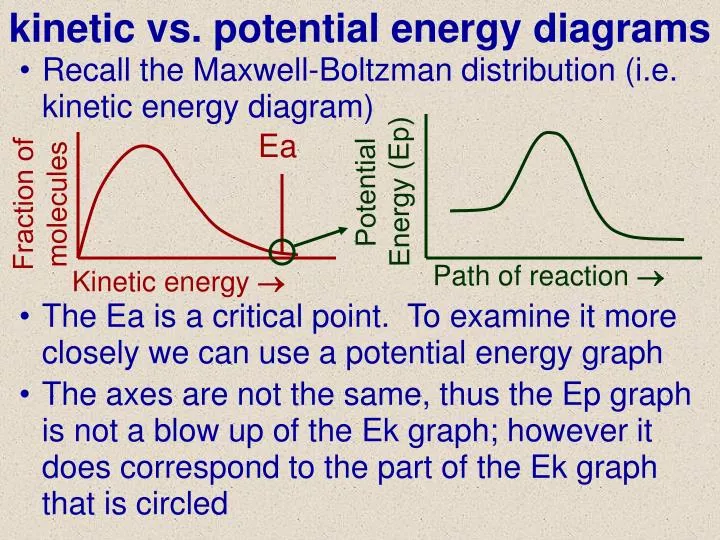
What about a roller coaster? When it’s at the top of the first big hill, it’s packed with potential energy. As it rushes down, that potential energy transforms into kinetic energy, making it zoom! But as it goes up and down other hills, you’ll notice it never reaches the same height as the first hill. Again, those sneaky energy losses at play.
The Takeaway: A Universe of Balance
So, to sum it all up in a way that won't give you a headache, the reason kinetic energy can't be greater than potential energy is all thanks to the Law of Conservation of Energy. Energy is a finite resource in any closed system; it just changes its fancy dress. It can’t magically multiply itself.
In a perfectly efficient world, kinetic energy can equal the initial potential energy at its peak. But in our real, wonderfully imperfect world, there are always little bits of energy escaping as heat, sound, or other forms. This means that, in practice, the kinetic energy is always less than or equal to the initial potential energy.
It’s a fundamental principle of how our universe operates, a constant dance of transformation and balance. And while it might seem like a limitation, think about it this way: this constant exchange, this beautiful transfer of energy, is what makes everything happen! From the tiniest atom buzzing with energy to the grandest star shining in the sky, it's all about energy finding new ways to express itself.
So, the next time you see something move, or something poised to move, remember this principle. It's not a restriction; it's an invitation to appreciate the incredible, interconnected, and endlessly fascinating flow of energy that makes our world, well, work. Isn't that a wonderfully uplifting thought? Keep exploring, keep wondering, and keep that awesome energy flowing!
