Why Are Metallic Crystals Malleable And Ductile
Alright, settle in, grab a cuppa, because we’re about to dive into something that sounds super science-y but is actually way cooler than you think. We’re talking about why your favorite metal spoon can bend without snapping like a dry twig, and why that electrical wire isn’t just a solid rod. It’s all about the secret life of metallic crystals, and trust me, they have more drama than a daytime soap opera.
So, imagine you’ve got a bunch of tiny little Lego bricks, right? But instead of just sticking together randomly, these Lego bricks in a metal are arranged in a super neat, super orderly pattern. Think of it like a perfectly stacked army of soldiers, all standing at attention. This, my friends, is a crystal lattice. Metals are basically made of these tiny, organized armies of atoms, all chilling together in a predictable grid.
Now, you might be thinking, “Okay, orderly armies, so what? Sounds kinda boring.” But here’s where the magic, and the malleability, comes in. Unlike, say, a bunch of salt crystals that are all brittle and will shatter if you sneeze too hard, these metal armies have a secret weapon: delocalized electrons. Imagine the electrons in these metal atoms are not strictly tied to their individual soldiers. Nope! They’re more like the paparazzi, flitting around everywhere, a whole swarm of them, creating this awesome, super-sticky, electron-gluey atmosphere.
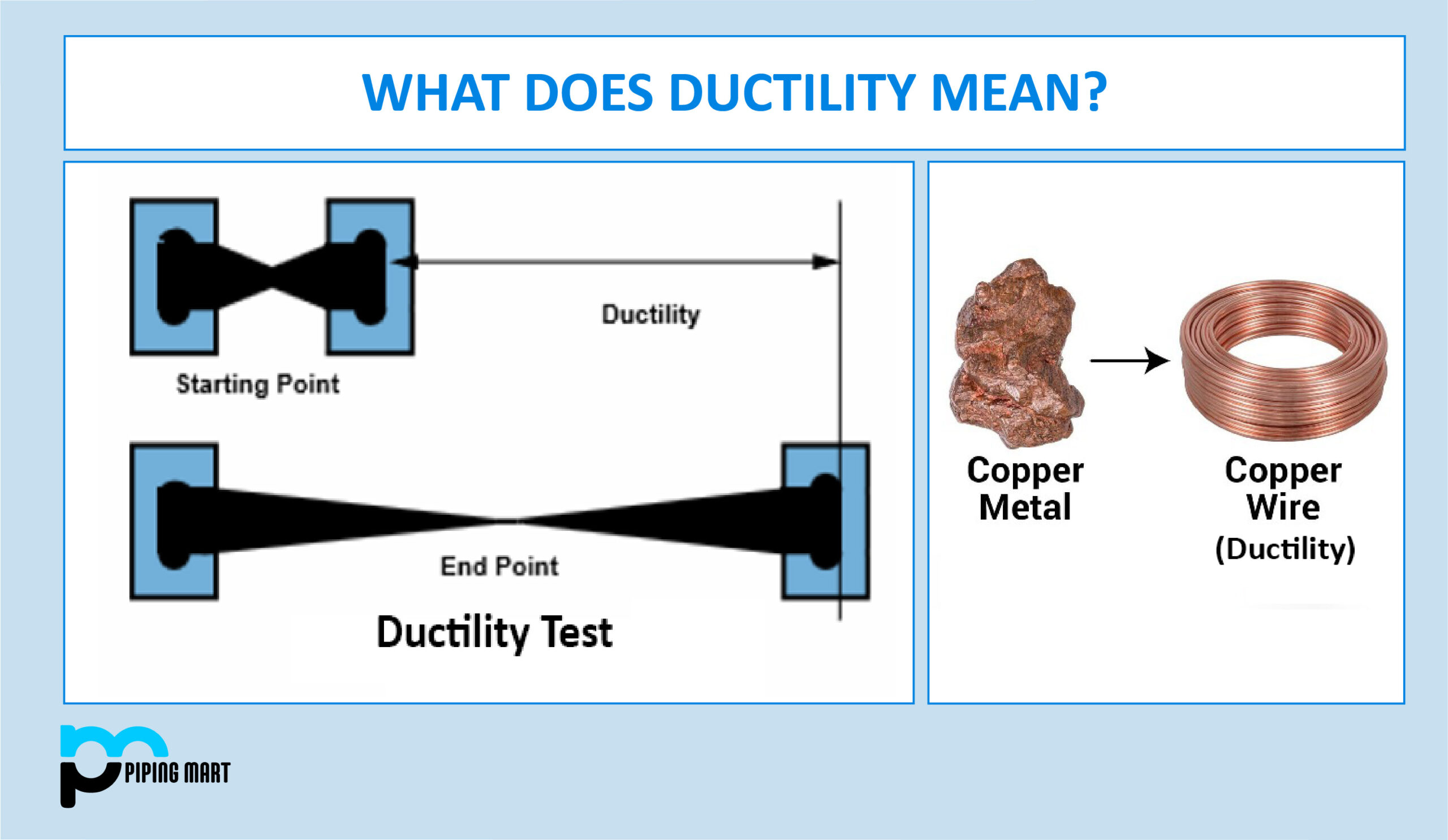
This cloud of free-roaming electrons is what holds the whole metal structure together. It’s like a gigantic, energetic hug from all the atoms. Now, when you try to persuade a metal to change shape – say, you’re hammering on it (that’s malleability, by the way, or being able to be hammered into thin sheets, like gold foil on fancy cakes!) or stretching it (that’s ductility, the reason we have all those lovely wires and cables!) – something really neat happens.
Instead of the orderly rows of atoms just cracking and breaking apart when you push them, they can actually slide past each other. It’s like those Lego soldiers, instead of toppling over when you nudge the pile, they can just sort of shimmy and slide into a new formation. The electron-glue is so strong and flexible that it just keeps holding everything together, even as the atoms rearrange themselves. It’s like the electrons are saying, “Whoa there, buddy, you’re moving? No problem, we got this!”

Think of it this way: if the electrons were all strictly belonging to one atom, like a possessive ex, and you tried to move the atoms, they’d be like, “Hey! That’s my electron!” and BAM! Instant fracture. But because these electrons are communal property, shared by everyone like free Wi-Fi at a coffee shop, they don’t get all bent out of shape when the atoms decide to do a little dance.
This sliding happens along specific planes within the crystal lattice, called slip planes. It’s like there are designated dance floors where the atoms can glide and glide without causing a structural meltdown. The more slip planes a metal has, and the easier it is for atoms to slide along them, the more malleable and ductile it’s going to be. This is why gold, with its relatively easy-to-slide atomic layers, is so incredibly bendy. You can literally hammer a sheet of gold so thin you can see through it – that’s some serious malleability!

Now, not all metals are created equal in the malleability department. Some are tougher nuts to crack than others. Think of iron. It’s strong, sure, but it’s not as delightfully bendy as, say, copper. This has to do with the specifics of their crystal structures and how easily those atoms can shimmy. Some atomic arrangements make it harder for those slip planes to do their thing.
And here's a fun fact: the reason why you can’t really bend a piece of plastic or glass into a wire is because their atoms are arranged differently. They’re more like jumbled piles of rocks, not neat Lego armies. When you try to deform them, the bonds between the atoms are rigid and break easily, leading to shattering. No electron-glue to save the day there!

So, next time you’re using a metal fork, or admiring a shiny piece of jewelry, give a little nod to those amazing metallic crystals. They're not just passive lumps of stuff; they're dynamic, flexible structures held together by a sea of electrons, allowing them to be hammered, stretched, and shaped into all the wonderful things we use every day. It’s a tiny atomic party, and everyone’s invited to slide and shimmy!
It’s a beautiful dance of atoms and electrons, a testament to the fact that even in the most rigid-looking materials, there’s often an incredible capacity for movement and transformation. Who knew that such a fundamental property of metals, something we often take for granted, was down to such an elegant and communal atomic arrangement? It’s like the universe decided to make metals really good at giving hugs – a strong, flexible, all-encompassing electron hug.
