Why Are Cations Smaller Than Their Parent Atoms

Okay, so let's talk about atoms. These tiny, invisible building blocks of everything are pretty neat. We've all heard about them, right? They're like the Lego bricks of the universe.
But have you ever thought about what happens when these atoms decide to get rid of some of their stuff? Like, when they become a bit… lighter? This is where things get interesting, and maybe a little counterintuitive.
So, imagine an atom. It's got a nucleus in the middle, like a grumpy old dad. And then it has electrons buzzing around, like hyperactive kids. These kids are important!
Now, sometimes, an atom feels like it has too many electrons. It’s like having too many toys and not enough space to play. So, it decides to shed a few. This is where our journey begins!
When an atom loses an electron (or a few!), it becomes something new. It's not an atom anymore, it's a cation! Think of it as the atom's "lite" version.
And here's the funny part, the thing that makes me shake my head and chuckle. These cations, these electron-shedding atoms, are actually smaller than their parent atoms. Yep, you heard me. Smaller!
It seems a bit unfair, doesn't it? You'd think losing something would make you weaker, or maybe more spread out. But nope. Not in the atom world.
Think about it like this. Imagine a crowded room full of people. Everyone's bumping into each other. That's kind of like an atom with all its electrons. Lots of electron-electron repulsion going on.
Now, a few people leave the room. Suddenly, there's more personal space, right? Everyone can spread out a little more comfortably.
But with atoms, it's the opposite! When electrons leave, the remaining ones feel… less crowded. They're not pushed around as much by their departing pals.

The nucleus, that grumpy old dad in the center, still has its same positive charge. It's like its pulling power hasn't changed.
But now, with fewer electrons to pull, that positive pull gets concentrated. It’s like a vacuum cleaner with less fluff inside – it can suck up what’s left more effectively.
So, the remaining electrons get pulled in tighter. They snuggle up closer to the nucleus. And voilà! The cation shrinks. It’s like a deflated balloon that gets squeezed.
It’s an unpopular opinion, I know. My chemistry teacher might have given me a stern look. But it just makes sense in a weird, atom-logic kind of way.
We are used to thinking that removing things makes something less substantial. Less substance, less size. But atoms are rebels. They defy our everyday logic.
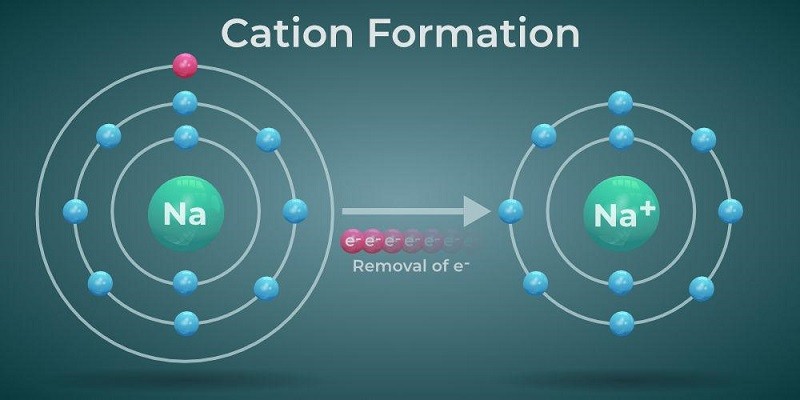
Consider Sodium (Na). It's a regular, decent-sized atom. It's got its nucleus and its electron entourage.
But when Sodium decides to become a cation, it becomes Na+. It’s lost an electron. And poof! It’s a lot smaller.

It's like shedding a winter coat. You might feel a bit lighter, but you also feel more… compact. More streamlined. Ready to move faster.
And it’s not just Sodium. This happens with pretty much all atoms that become cations. It’s a universal rule in the tiny world of chemistry.
Think about Magnesium (Mg). It's a perfectly fine atom. Then it decides to get rid of two electrons and becomes Mg2+.
And guess what? It’s even smaller than the sodium cation. The more electrons it loses, the tighter things get pulled in. It’s like a super-concentrated atom.
It’s almost like the atom is showing off. "Look at me! I've gotten rid of some baggage, and I'm looking good and small."
Perhaps it’s the atom’s way of saying, "Less is more!" It’s a minimalist philosophy for the atomic world.
The electrons, those energetic youngsters, are held more tightly by the nucleus’s charm. They are no longer playing in a vast, sprawling playground. They are in a more exclusive, cozy club.
This closer proximity means the overall "size" of the electron cloud is reduced. The outer boundaries of the atom get pulled inward.

It's a bit like a group of friends who used to hang out all over town. But when a few friends move away, the remaining friends stick closer together, maybe just meeting at one favorite cafe.
The nucleus, with its positive protons, is like the magnetic force. It draws the negative electrons. When there are fewer electrons, this magnetic pull is more intense per electron.
So, the electrons don't have as much freedom to roam. They are more confined, and this confinement makes the cation a more compact package.
It's a fascinating dance of forces. The attraction between the nucleus and electrons, and the repulsion between the electrons themselves.
When electrons are removed, the repulsion decreases. This allows the attractive forces to win, pulling the remaining electrons closer.
It’s a bit like a tug-of-war. If you have fewer people on one side of the rope, the few people on the other side can pull harder.
So next time you hear about cations, remember this little secret. They’re not just atoms that lost a bit of their "stuff." They are leaner, meaner, and undeniably smaller.
It's a charming quirk of the universe. A reminder that things aren't always as they seem, especially when you're dealing with the incredibly small.
And if anyone tells you otherwise, just smile. You know the truth. Cations are the compact celebrities of the atomic world. They've shed the excess and embraced a more streamlined existence.
So, the next time you see a chemical formula with a little plus sign, give a nod to the cation’s shrinking prowess. It’s a tiny but mighty marvel!
It's a delightful paradox. Losing something makes you smaller, not bigger. It’s like a diet that actually works wonders.
The parent atom is like a person with a full shopping cart. A bit cumbersome. A cation is that same person with just a few items, zipping through the aisles with ease.
This difference in size is actually super important in chemistry. It affects how atoms bond and interact. It's the little details that make big things happen.
So, while it might seem a bit odd, the shrinking cation is a fundamental concept. And honestly, a rather amusing one. It’s the universe’s way of keeping us on our toes.
Just remember: less electrons equals a tighter, smaller atom. Simple as that. Well, as simple as atomic physics gets, anyway!
It's a bit like a celebrity shedding their entourage. They might have fewer people around them, but they become more focused, more streamlined, and in the atom's case, smaller. A curious but captivating phenomenon.
