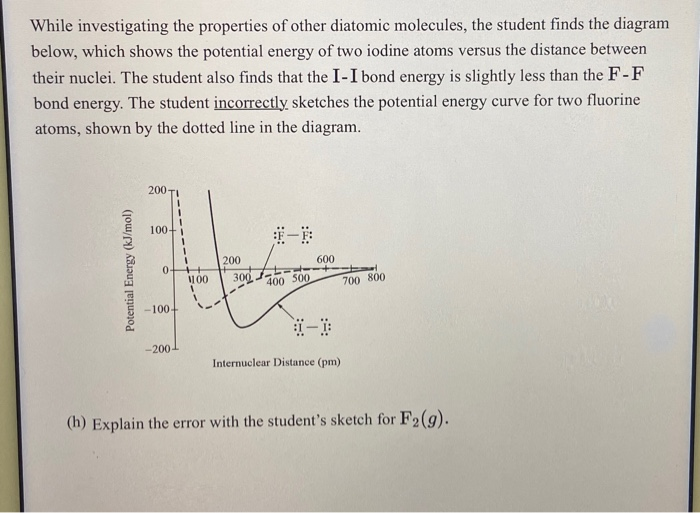
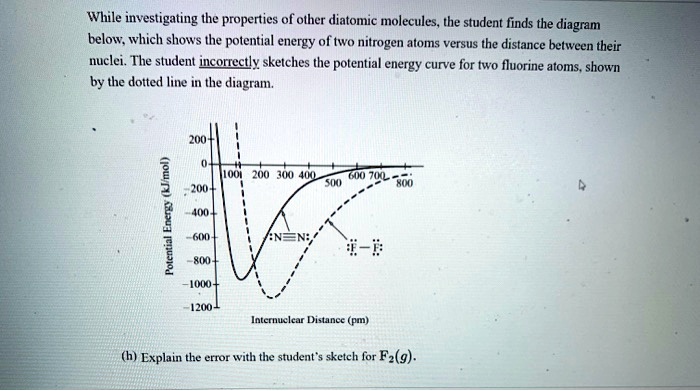
While Investigating The Properties Of Other Diatomic Molecules
Hey there, fellow curious minds! Ever find yourself staring at a perfectly brewed cup of coffee, or maybe watching a little kid blow bubbles, and wonder what’s really going on with all the tiny, invisible stuff that makes those things happen? Well, today we’re going to dive into a surprisingly cool topic that’s a bit like being a detective for the really, really small. We’re going to chat about something called diatomic molecules. Now, don't let that fancy word scare you! Think of it as a secret handshake for pairs of atoms that just love sticking together.
So, what exactly is a diatomic molecule? Imagine you’re at a party, and everyone’s mingling. Most people are happily chatting in little groups, but some folks just find their perfect partner and decide to stick together, almost like they're holding hands. That’s kind of what’s happening with diatomic molecules. They’re made of just two atoms that are bonded together. Simple, right?
Now, you might be thinking, "Okay, that’s neat, but why should I care about a couple of atoms holding hands?" Ah, my friend, that’s where the magic happens! These little two-atom buddies are everywhere. They’re in the air we breathe, the water we drink, and even in the snacks we munch on. Understanding them is like getting a backstage pass to how the world around us actually works.
Let’s take the most famous diatomic molecule of all: oxygen (O2). Yep, that stuff that keeps us all alive and kicking! When you take a deep, refreshing breath after a brisk walk or a good laugh, you’re filling your lungs with these O2 pairs. They’re like little energy boosters, ready to help your body do all its amazing things. Without these diatomic oxygen molecules, well, life as we know it wouldn’t be so… lively.
Think about it like this: imagine you’re building with LEGOs. You can have individual LEGO bricks, but when you snap two together, they become a stronger unit. Diatomic molecules are kind of like those snapped-together LEGOs. They've found a way to be more stable, or at least more comfortable, when they’re paired up.
It's not just oxygen, though! There's another equally important diatomic molecule we encounter constantly: nitrogen (N2). This is actually the most abundant molecule in our atmosphere – like the quiet, steady friend in the background of every gathering. While oxygen is the energetic one that gets things going, nitrogen is the more reserved, stable partner. It makes up about 78% of the air we breathe! Imagine a big, comfy couch that’s mostly made up of nitrogen. Oxygen is like the fun pillow you can grab when you want some excitement.
These two, oxygen and nitrogen, are the kings and queens of the diatomic world when it comes to our everyday lives. But there are others! We've got hydrogen (H2), which is incredibly important in stars and for making things like water (H2O – see, more pairs!). We’ve got fluorine (F2), chlorine (Cl2), bromine (Br2), and iodine (I2). These last four are a group called halogens, and they’re like the very reactive, exciting guests at the molecular party. They’re very good at making friends with other elements, sometimes a little too good!

Why do some elements form these diatomic pairs and others don't? It all comes down to something called electron configuration. Don't worry, we're not going to get bogged down in quantum mechanics! Just think of electrons as tiny, energetic dancers around the center of an atom. These dancers like to be in specific spots, and sometimes, the best way for an atom to get its dancers in the "happy" spots is to share them with another atom. When two atoms share electrons to form a bond, they become a diatomic molecule.
It's like a dance competition where two dancers realize they can get a better score if they perform a synchronized routine, rather than dancing solo. They hold hands, move together, and create a more harmonious performance. In the same way, two atoms bond to achieve a more stable electron arrangement.
Let's consider water (H2O) again. While the whole molecule isn't diatomic (it has three atoms), it's made up of oxygen bonded to two hydrogen atoms. Each of those oxygen-hydrogen bonds is a little two-atom partnership. So, even in more complex molecules, the diatomic bond is a fundamental building block. It’s like the fundamental chords in music; they can be combined to make symphonies, but the basic chord is still there.
What’s so cool about studying these diatomic molecules specifically? Well, for starters, they’re like the simplest test subjects for understanding chemical bonds. Since they’re just two atoms, scientists can study their properties – how they vibrate, how they rotate, how they absorb light – in a much more focused way. It's like studying a single note before trying to understand an entire orchestra.
This fundamental understanding is what allows us to do amazing things. For instance, when we look at the light from distant stars, scientists can analyze the "fingerprints" of diatomic molecules like carbon monoxide (CO) or hydrogen cyanide (HCN) in their atmospheres. These fingerprints tell us what those stars are made of and what conditions are like light-years away! It's like reading a cosmic postcard.
Or think about industrial processes. Understanding how nitrogen and oxygen behave when heated or pressurized is crucial for everything from making fertilizers (which help our food grow) to designing rocket engines. These diatomic molecules are the unsung heroes of many technologies we rely on every single day.
Even in our own bodies, diatomic molecules play vital roles beyond just breathing. For example, nitric oxide (NO) is a diatomic molecule that acts as a signaling molecule in our bodies, affecting blood pressure and nerve function. It's like a tiny messenger delivering important instructions!
So, the next time you're enjoying a slice of pizza, taking a breath of fresh air, or simply gazing up at the night sky, remember these tiny, two-atom partnerships. They’re not just abstract scientific concepts; they’re the fundamental building blocks of our universe, the silent partners in countless everyday phenomena. They’re a constant reminder that even the simplest arrangements of matter can hold immense importance and unlock incredible wonders about the world around us. Pretty neat, right?
