Which Would You Expect Of A Eukaryotic Cell Lacking Telomerase
Ever wondered what makes us tick, or more accurately, what keeps our cells in tip-top shape? It's a microscopic world out there, and within it, our eukaryotic cells are the rockstars. These are the sophisticated cells that make up all plants, animals, fungi, and even some algae – essentially, anything that isn't a simple bacterium. They’re like tiny, bustling cities with specialized neighborhoods (organelles) and intricate management systems. But even the best-managed cities can face challenges, and understanding these challenges helps us appreciate the incredible resilience and complexity of life. Today, we're diving into a particularly juicy thought experiment: what would happen if these cellular powerhouses suddenly lost a key player, the amazing enzyme called telomerase?
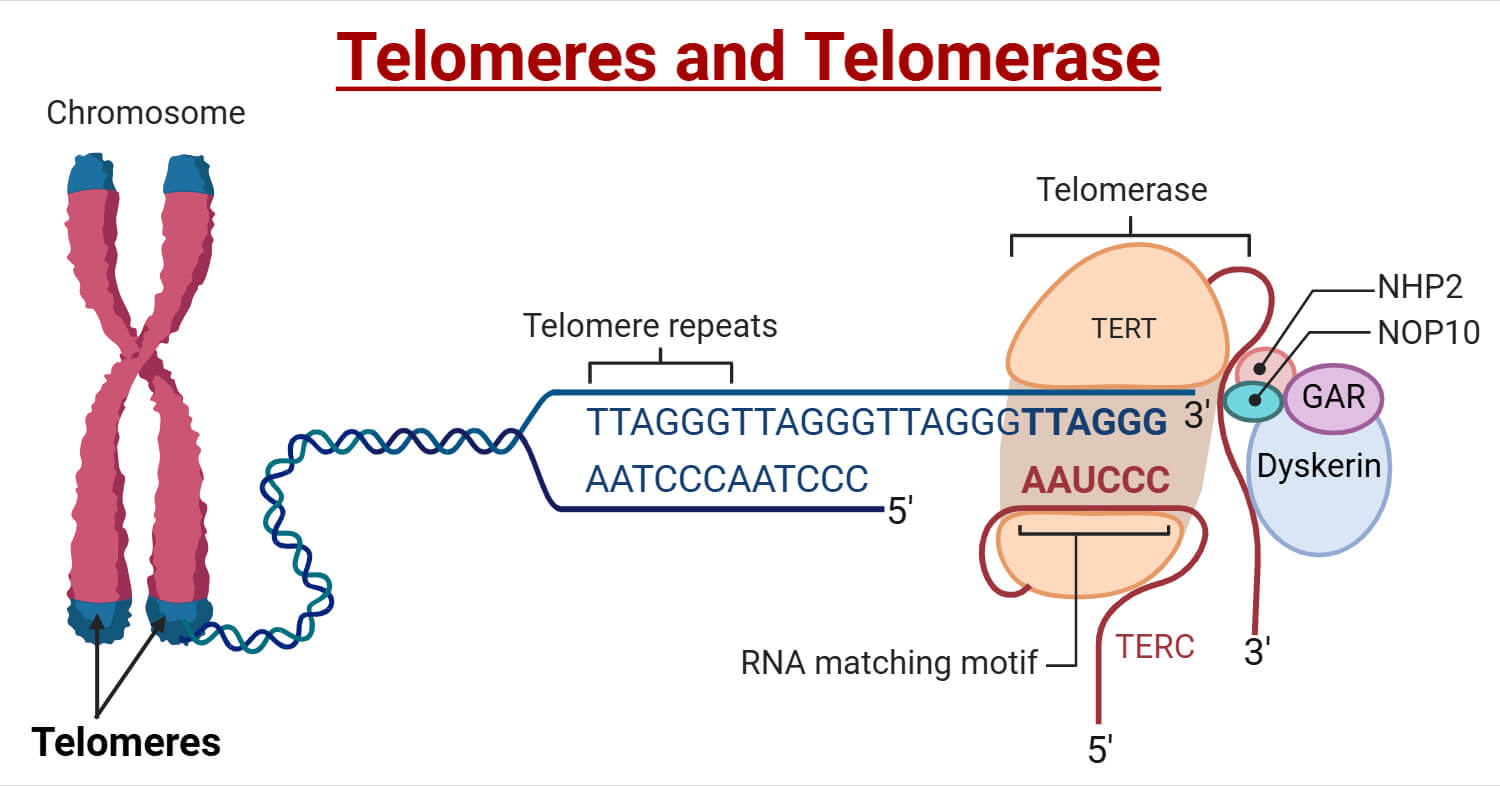
This isn't just some dry biology quiz question; it's a fascinating peek into the fundamental processes that govern aging, reproduction, and even the fight against diseases like cancer. Think of our chromosomes, the structures that hold our genetic blueprint, like the instruction manuals for our cells. At the very tips of these manuals are protective caps called telomeres. These telomeres are crucial because they prevent the important genetic information from fraying or being lost during the cell's constant process of division. Imagine the end of a shoelace – the plastic tip (the telomere) keeps it from unraveling. Without these caps, our chromosomes would be in serious trouble, leading to errors in our DNA and eventually, cell malfunction.
Now, enter telomerase. This enzyme is like a highly skilled editor and repair crew rolled into one. Its primary job is to rebuild and lengthen these protective telomeres. Every time a eukaryotic cell divides, its telomeres get a little shorter. It’s a natural process. However, in most of our regular body cells, telomerase activity is very low. This means that over time, as cells divide, their telomeres shorten. This shortening acts like a cellular clock, eventually signaling the cell to stop dividing and enter a state of senescence (a sort of cellular retirement) or even to undergo programmed cell death, known as apoptosis. This is a good thing in general, as it prevents uncontrolled cell growth, which is the hallmark of cancer.
But what if a eukaryotic cell was completely devoid of telomerase? Imagine that shoelace without its tip, and no way to reattach or replace it. The consequences would be significant and, in the long run, quite detrimental for the cell. The most immediate and predictable outcome would be progressive telomere shortening with each subsequent cell division. In cells that rely on frequent replication, like those in the skin, the lining of the gut, or our immune cells, this would happen at an accelerated rate. The cells would quickly reach their limit for division.
The inability to maintain telomere length would lead to genomic instability, making the cell highly susceptible to mutations.
PPT - DNA Replication PowerPoint Presentation, free download - ID:1087536
As telomeres become critically short, they can trigger a cellular crisis. The chromosome ends, now unprotected, can fuse together or get misread by the cell's machinery. This chaotic situation can lead to the accumulation of significant DNA damage and errors. Think of a library where the protective covers of books are missing; they get damaged, pages get ripped, and it becomes impossible to find the right information. Similarly, a eukaryotic cell lacking telomerase would experience genomic instability, a dangerous state where the integrity of its genetic material is compromised. This instability can pave the way for various problems, including the development of diseases.
One of the most striking effects would be a dramatic reduction in the cell's replicative lifespan. Cells would become senescent much faster, meaning they’d stop dividing prematurely. This would have profound implications for tissue repair and regeneration. Imagine trying to heal a cut or recover from an illness if your cells simply couldn't divide enough to replace the damaged ones. This could lead to premature aging at a cellular level and impaired ability of the body to maintain itself. Tissues would degenerate more quickly, and the overall function of organs could be compromised.

However, the story isn't entirely bleak for every single cell. Interestingly, cancer cells often overcome this limitation by reactivating telomerase. This is one of the key reasons they can divide indefinitely, a characteristic known as immortality. So, a eukaryotic cell lacking telomerase would, in a way, be protected against becoming a cancerous cell itself, at least from this particular mechanism of immortalization. Yet, the other detrimental effects of telomere shortening, like genomic instability, could still contribute to cancer development through different pathways. It’s a complex dance between preventing cancer and maintaining cellular function.
In the context of an organism, if all or most of its cells lacked functional telomerase, the implications would be severe. Development might be severely hampered, and the organism would likely experience accelerated aging and a significantly shorter lifespan. Reproduction could also be affected, as germ cells (the cells that give rise to sperm and eggs) normally rely on mechanisms to maintain telomere length to ensure that offspring inherit healthy, functional chromosomes. Without telomerase, these cells would also face severe limitations.
So, while the loss of telomerase might seem like a simple switch being turned off, it triggers a cascade of events that are fundamental to life. It highlights the exquisite balance that eukaryotic cells maintain to ensure their survival, function, and the continuation of life itself. Understanding this process isn't just about memorizing enzymes; it's about appreciating the intricate machinery that makes us who we are, and the delicate pathways that, when disrupted, can lead to disease and aging. It’s a testament to the power of tiny, often unseen, molecular tools that work tirelessly to keep our cellular cities running.

