Which Word Equation Represents A Neutralization Reaction

Ever wondered what happens when things get a little too... lively? Like when you've got a super sour lemon and you want to make it taste a bit more mellow? Well, in the world of science, there's this incredibly cool dance called a neutralization reaction. It's like a party where two opposites come together to create something perfectly balanced and, dare we say, a little bit magical!
Imagine you have two characters. One is a bit of a show-off, always bubbling with energy and ready to make things feel sharp and zesty. We call this character an acid. Think of things like lemon juice or vinegar – they have that distinct tang, right? Then, on the other side, you have a character that’s the complete opposite. This one is smooth, calming, and likes to take the edge off. This is our base. Think of things like baking soda – it feels a bit powdery and can help soothe a tummy ache.
Now, what happens when these two meet? It’s not a fight! Oh no, it’s a beautiful collaboration. They get together, and something amazing happens: they cancel each other out! It’s like they’re high-fiving and saying, "Okay, you take some of my zing, and I'll take some of your chill." The result? Something that’s neither strongly acidic nor strongly basic. It’s wonderfully neutral. And what do we get when we create something neutral? Usually, it's water! Yep, plain old H2O, the stuff we drink and that makes up most of our planet. How neat is that?
But wait, there's more! When an acid and a base hug it out, they don't just make water. They also create a little something extra, like a souvenir from their party. This souvenir is called a salt. Now, when we hear "salt," we usually think of the stuff you put on your fries. And sometimes, it is exactly that! But in chemistry, a salt is a much broader family of compounds. It's like a whole bunch of different flavors of souvenirs they can bring back from their reaction.
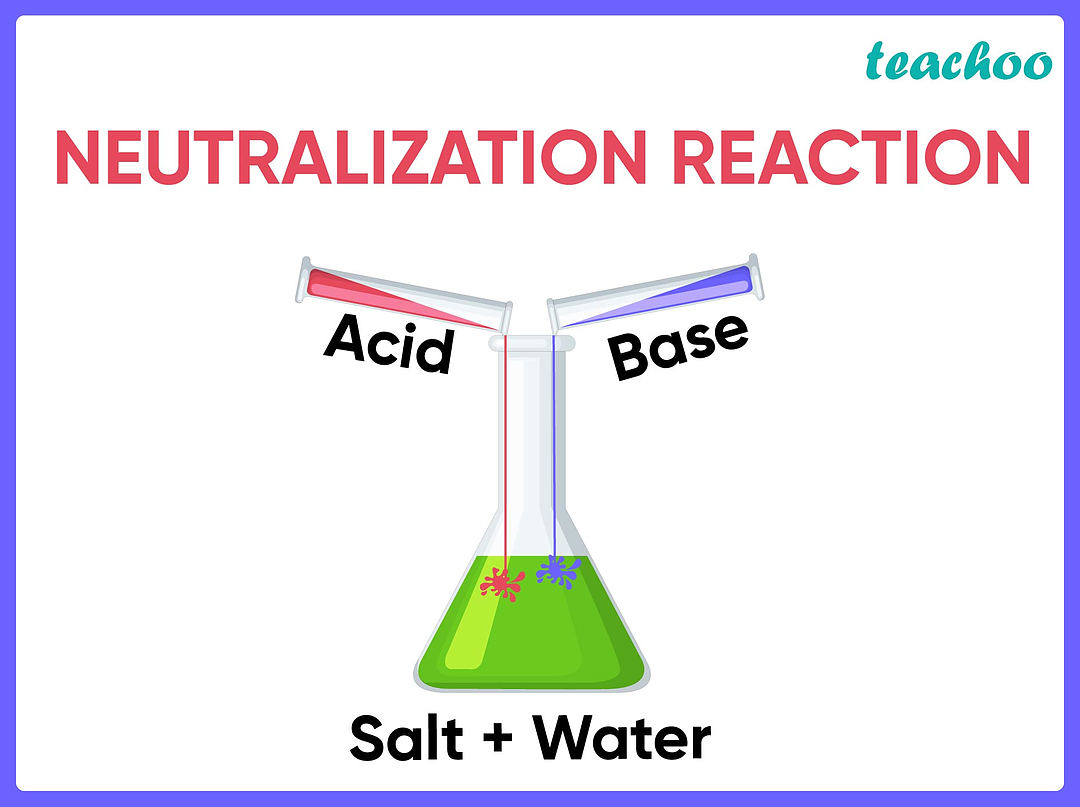
So, how do we write down this whole exciting party in a way that scientists understand? We use something called a word equation. It's like writing down the guest list and the main events of the party. It tells us who showed up and what they made. For a neutralization reaction, the word equation is super straightforward. It’s like the simplest, most elegant way to capture this whole event.

The general form of this epic word equation goes like this:
Acid + Base → Salt + Water
See? It’s as simple as that! You put the acid and the base on one side, and with a fancy arrow that means "turns into" or "produces," you have the salt and the water on the other side. It's like a recipe for balance!

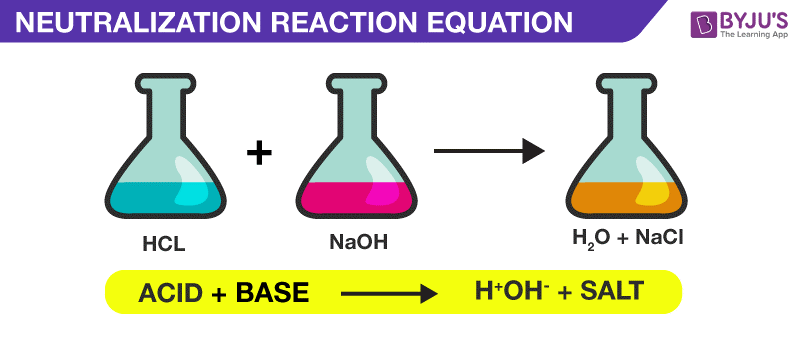
Let's imagine a specific party. What if we have our super-sour friend, hydrochloric acid (that's HCl, if you're feeling fancy, but we'll stick to words for now!). And let's invite our smooth operator, sodium hydroxide (that's NaOH, but again, words are friendlier!). What do they create?
When hydrochloric acid meets sodium hydroxide, they have their little chemical rendezvous. The acid's "H" part gets together with the hydroxide's "OH" part, and poof! They form water. What's left? The "Cl" from the acid and the "Na" from the base decide to hang out. And guess what? They form sodium chloride. That's the scientific name for the table salt we all know and love!
So, the word equation for this specific shindig would be:
Hydrochloric Acid + Sodium Hydroxide → Sodium Chloride + Water
Isn't that just delightful? It's like watching a tiny drama unfold where everyone ends up happy and balanced. This word equation is so special because it’s a universal language for this fundamental chemical process. It doesn't matter if you're in a bustling lab or just curious in your kitchen, this equation tells the same story. It’s a peek into the hidden world of molecules and how they interact to create the world around us.
The beauty of a neutralization reaction is its predictability and its usefulness. We use it all the time without even realizing it! That antacid you take for heartburn? It's a base neutralizing the excess acid in your stomach. That baking soda paste you use to clean a greasy pan? It's helping to neutralize and lift away the grime. It’s the unsung hero of everyday balance!
So next time you hear about an acid and a base getting together, remember their word equation: Acid + Base → Salt + Water. It’s a simple phrase, but it captures a world of chemical harmony and everyday magic. It's a reminder that even the most opposite things can come together to create something wonderfully neutral and essential.
This equation isn't just a string of words; it's an invitation to see the chemistry that's constantly happening all around us, turning the sharp and the tangy into the calm and the balanced. It’s a little science party that makes the world work just right!
