Which Type Of Model Best Represents Simple Molecules

Ever looked at a tiny speck of something, like a drop of water or the puff of air you breathe, and wondered what it's actually made of? It’s like peering into a miniature universe! These super-duper small bits are called molecules, and even though we can't see them with our eyes, they're doing all sorts of amazing things to make our world go 'round.
Now, imagine you have a bunch of these tiny building blocks. How do you show someone what they look like? You can't just whip out a magnifying glass! Scientists, bless their clever socks, have come up with all sorts of cool ways to represent these invisible wonders. It’s a bit like trying to describe your favorite cartoon character to someone who’s never seen it.
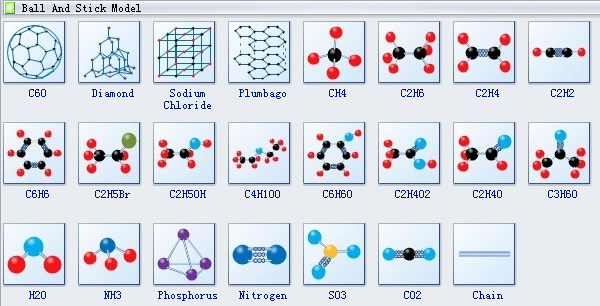
One of the most common and, dare I say, cutest ways to imagine a simple molecule is with what we call the "ball-and-stick" model. Think of it like a tiny molecular LEGO set! You’ve got little colorful balls, and they’re connected by sticks. It’s so straightforward, even your little cousin could probably understand it.
The balls, you see, represent the different types of atoms that are holding hands to form the molecule. Like, a bright red ball might be oxygen, and a smaller white ball could be hydrogen. And those little sticks? They're like the super-strong glue, the chemical bonds, that are keeping them all together in a hug. It’s a wonderfully simple picture of how things are connected.
This ball-and-stick approach is fantastic for showing off the shape of a molecule. You can see if it's bent like a little boomerang or if it’s all in a straight line. It’s like looking at a tiny, three-dimensional sculpture. You get a real sense of how these atoms are arranged in space, which is super important for understanding how they behave.
For example, think about water. We all know and love water, right? It’s essential for life! A water molecule is made of one oxygen atom and two hydrogen atoms. In a ball-and-stick model, you’d see that big red oxygen ball with two smaller white hydrogen balls attached, kind of like Mickey Mouse ears! This "bent" shape is actually a big deal for why water acts the way it does – why it’s so good at dissolving things and why ice floats.
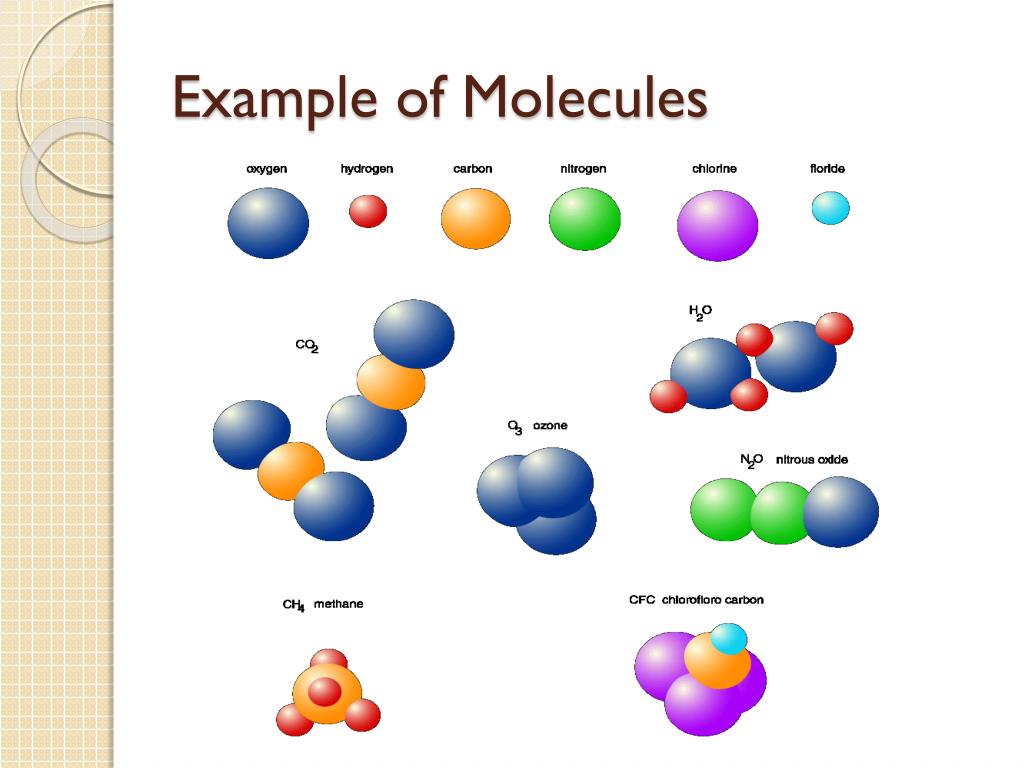
Another way to visualize these tiny worlds is with the "space-filling" model. This one is a bit more like a molecular sculpture made of soft, squishy marshmallows. Instead of just showing the connections, the balls are actually drawn to the correct size relative to each other. So, an oxygen atom looks bigger than a hydrogen atom, and they all merge together to form a sort of blob.
This model is great for showing how much space a molecule actually takes up. It gives you a better idea of the molecule’s overall size and its outer surface. Imagine trying to pack a bunch of these into a small box – the space-filling model helps you see if they’d all fit snugly or if there’d be a lot of awkward gaps. It’s a bit more realistic about the "real estate" these molecules occupy.
The space-filling model really emphasizes that atoms in a molecule aren't just little points connected by lines; they actually have a volume. This is super important when molecules start bumping into each other. It helps us understand things like how easily one molecule can get close enough to another to react with it.

So, which one is best for simple molecules? Well, it's a bit like asking if a drawing or a sculpture is better. They both show you something different and equally fascinating! For understanding the basic connections and the general shape, the ball-and-stick model is usually the star. It’s clear, easy to draw, and immediately tells you how the pieces are put together.
It’s like getting a simple diagram of how your favorite toy is assembled. You see the main parts and how they snap into place. For a beginner trying to grasp the concept of molecules, the ball-and-stick model is often the friendliest introduction. It takes a complex idea and makes it as approachable as building with blocks.
However, if you want to get a better feel for the molecule's actual "footprint" or how it interacts with its neighbors, the space-filling model takes the cake. It’s like seeing a more detailed blueprint that includes how much room each part takes up. This can be really helpful when you start thinking about how molecules fit together in larger structures.

Think about a very simple molecule like methane, the main ingredient in natural gas. It's just one carbon atom and four hydrogen atoms. A ball-and-stick model shows you the central carbon ball with four hydrogen balls sticking out in a sort of tripod shape. It's a clear and satisfying picture.
Then, the space-filling model of methane shows you a central, larger blob (the carbon) with four smaller, fuzzier blobs (the hydrogens) hugging it, creating a roughly spherical shape. You can really see how it would roll around! Both models are telling you about methane, but in slightly different, yet equally valuable, ways.
Sometimes, scientists also use simplified drawings that look a bit like a skeleton. These are called skeletal structures. For simple molecules, they're like a shorthand. You don't draw every single atom and bond. Instead, the lines represent bonds, and the corners and ends of lines are assumed to be carbon atoms unless something else is specified.
It’s like seeing a secret code that chemists use. For example, a zig-zag line might represent a chain of carbon atoms, with hydrogen atoms attached to fill up all the empty spots. It's super efficient for drawing longer, more complex molecules, but for really simple ones, the ball-and-stick or space-filling models are often more intuitive for us regular folks.
The beauty of these models is that they help us visualize the invisible. They take abstract chemical formulas and turn them into something we can picture, even if it’s just in our minds. It’s like having a mental toy box filled with tiny, intricate molecular constructions.
So, next time you’re enjoying a glass of water or breathing in a lungful of air, remember that there are these incredible, tiny molecules whizzing around. And thanks to these clever models, we have wonderful ways to imagine their shapes and understand their importance. It's a little peek into the amazing world of chemistry, made fun and accessible, one molecule at a time!
