Which Type Of Bond Represents A Weak Chemical Bond
Ever wondered what holds the world around us together, from the water we drink to the air we breathe? It's all thanks to the fascinating world of chemical bonds! While some bonds are like superglue, strong and unyielding, others are a bit more… gentle. Today, we're going to peek into the realm of the weakest chemical bonds. Why is this a fun detour? Because understanding these subtle connections unlocks a whole new appreciation for how matter behaves, and it's surprisingly relevant to everything from how your nose works to why some materials have unique properties!
So, what exactly is the point of a weak bond? You might think "weak" means "useless," but that's far from the truth. Weak bonds, often referred to as intermolecular forces, are crucial for a vast array of phenomena. They're the unsung heroes that allow molecules to interact with each other without completely merging or breaking apart. Think of them as the gentle nudges and playful shoves between molecules that create collective behaviors. The benefits are immense: they dictate the states of matter (solid, liquid, gas), influence solubility (why oil and water don't mix), and play a vital role in biological processes. Without these weaker interactions, life as we know it simply wouldn't be possible!
These weak bonds pop up everywhere, often in ways we don't consciously notice. In education, they're fundamental to understanding concepts like surface tension in water (think of a water strider gliding across a pond!), the boiling point of different liquids, and even how DNA strands can temporarily separate for replication. In our daily lives, the pleasant aroma of your morning coffee is due to volatile molecules interacting weakly with the air. The way a gecko's feet stick to a wall involves incredibly weak forces. Even the feel of silk between your fingers is a testament to these subtle molecular attractions.
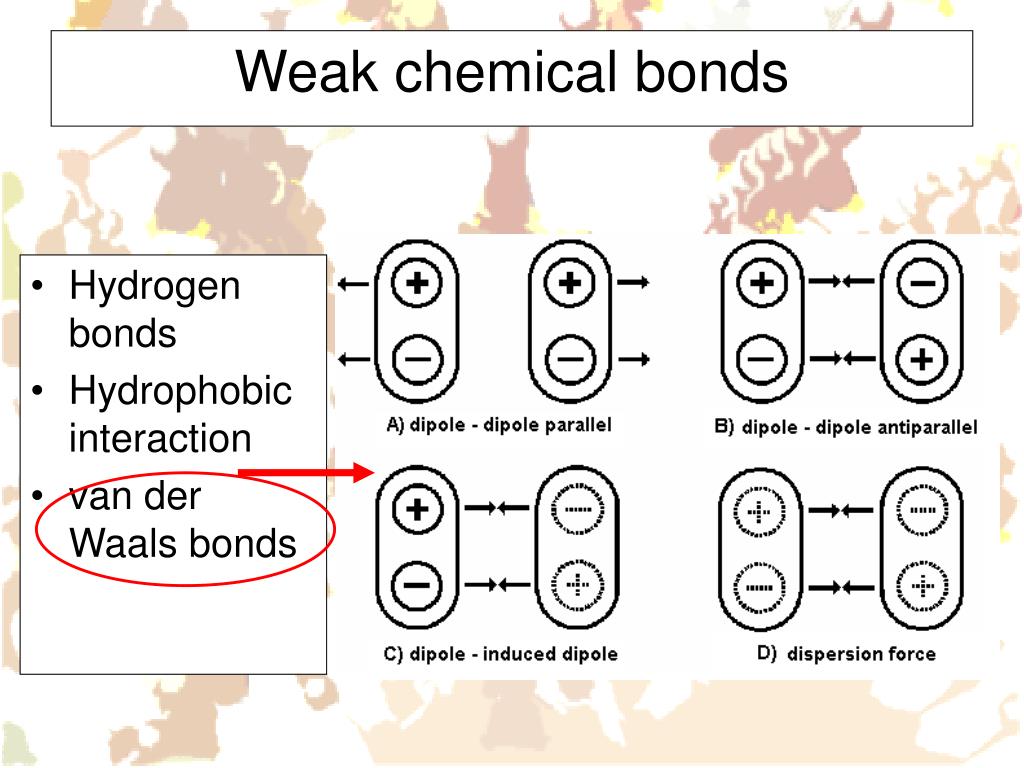
The standout champion for the title of the weakest chemical bond is often considered to be the London dispersion force (also known as van der Waals forces). These arise from temporary, fluctuating electron distributions in atoms and molecules, creating fleeting, weak attractions. While individually weak, when you have many of them working together, they can become surprisingly significant. Another important player in the weak bond arena is the hydrogen bond, which is stronger than London dispersion forces but still considered weak compared to covalent or ionic bonds. These occur when a hydrogen atom is bonded to a highly electronegative atom (like oxygen or nitrogen) and is attracted to another electronegative atom in a nearby molecule.
Ready to explore this fascinating world a little more? It's easier than you think! You can start by observing everyday phenomena. Notice how water forms droplets instead of spreading out thinly – that's intermolecular forces at play! Think about why ice floats on water (a consequence of hydrogen bonding and its unique structure). You can even do simple experiments at home, like seeing how different liquids evaporate at different rates. A quick search online for "intermolecular forces demonstrations" will reveal many fun and visual experiments you can try or observe. So next time you marvel at a dewdrop or enjoy a cool drink, remember the quiet, powerful influence of these gentle giants of the molecular world!
