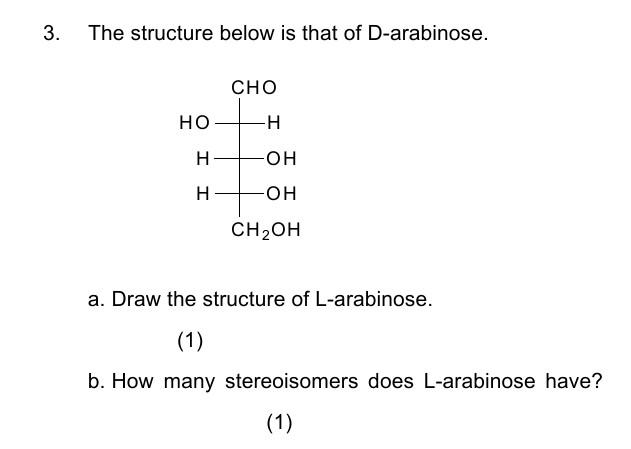
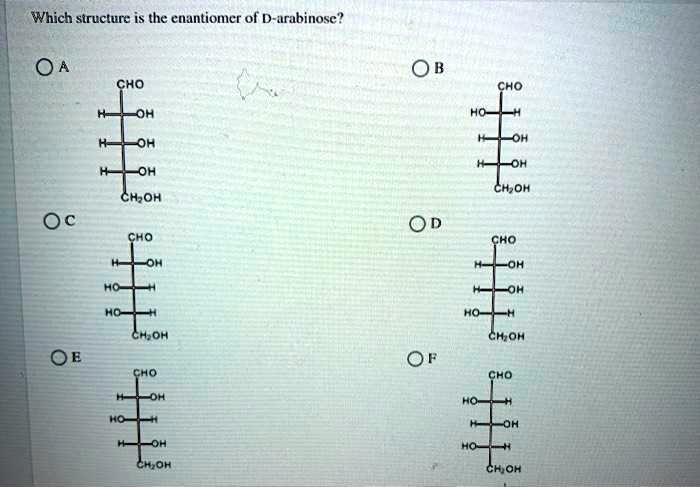
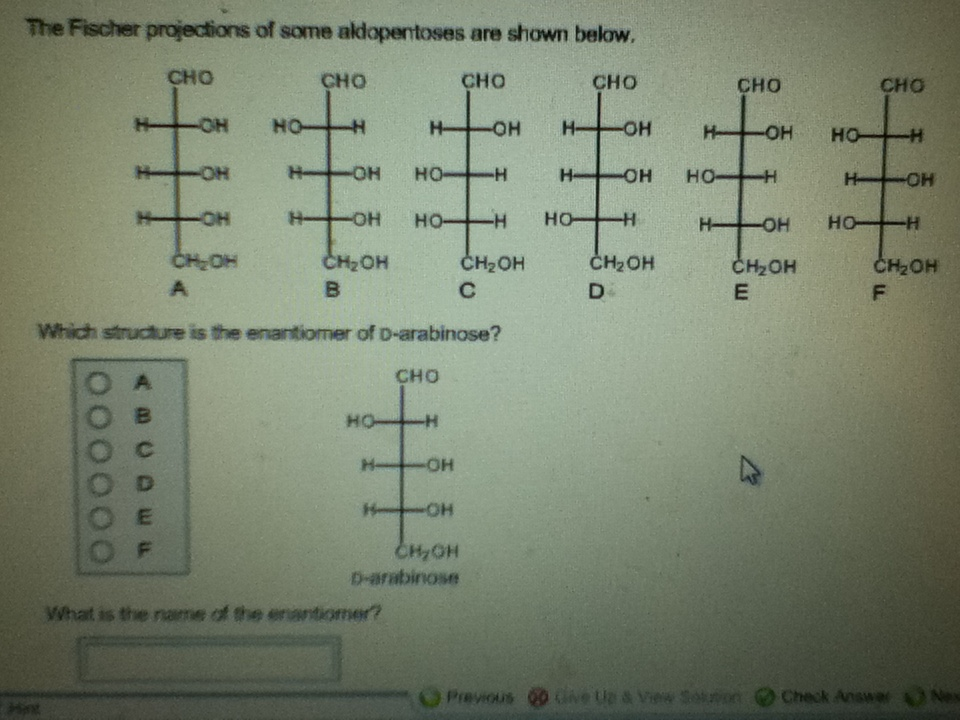
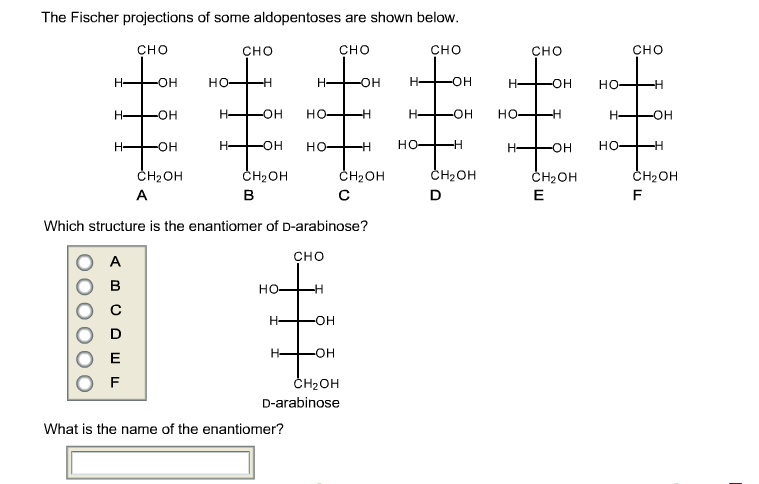
Which Structure Is The Enantiomer Of D Arabinose

Hey there, coffee buddy! So, you’re wondering about enantiomers, huh? Especially when it comes to D-arabinose? Totally get it. It’s like trying to figure out which sock is the other sock, right? That perfectly mirrored, yet totally different, counterpart. And when we’re talking about sugars, things can get a little twisty. Get it? Twisty? Okay, maybe I’ll stick to chemistry.
So, D-arabinose. Let's break this down before we dive into its mirror image. Think of D-arabinose as our starting point, our “original” coffee mug, if you will. It’s a simple sugar, a pentose, meaning it’s got five carbons. Pretty standard stuff, but those carbons are arranged in a very specific way. And it’s that specific arrangement that matters, oh boy, does it matter!
Now, when we talk about enantiomers, we're essentially talking about molecules that are non-superimposable mirror images of each other. This is the golden rule, the unbreakable law of enantiomery. Imagine your left hand and your right hand. They look pretty darn similar, right? They’re both hands! But try as you might, you can’t perfectly stack your left hand on top of your right hand. They just don't match up. That, my friend, is the essence of enantiomers. They are reflections, but they are not identical twins who can swap outfits. Nope, not at all.
So, what’s the deal with D-arabinose and its shadowy twin? Well, it’s all about the chiral centers. Chiral centers are basically carbon atoms that are bonded to four different groups. Think of them as the little crossroads where the molecule’s identity can take a left turn or a right turn. D-arabinose, being a pentose, has a few of these potential crossroads. And the configuration at these crossroads dictates whether it’s a D-sugar or an L-sugar. It's like the subtle tilt of a building that makes it face north or south. Same building, different orientation.
The "D" in D-arabinose? It refers to the configuration of the hydroxyl group (-OH) on the highest numbered chiral carbon. This is a convention, a way for chemists to keep things organized, like a filing system for molecules. If that -OH group is on the right side in a specific standard projection (usually the Fischer projection, but let’s not get bogged down in too many acronyms just yet!), then it’s a D-sugar. If it’s on the left, then it’s an L-sugar. Simple as that, or is it? Ah, the sweet, sweet complexity of chemistry!
So, if D-arabinose has its highest-numbered chiral carbon's -OH group pointing to the right, what would its enantiomer look like? You guessed it! Its enantiomer would have that same highest numbered chiral carbon's -OH group pointing to the left. It’s like flipping the whole molecule over in a mirror. The rest of the arrangement of groups around that specific carbon would be reversed, creating that mirror image effect.
Now, here’s where things get really interesting, and a little mind-bending. D-arabinose is a specific isomer. And its enantiomer? It’s also a specific isomer. We’re not just talking about any random sugar. We’re talking about a very particular spatial arrangement. And this spatial arrangement is crucial. It affects how these sugars interact with other molecules, like enzymes in our bodies. Imagine trying to fit a left glove onto a right hand. It just doesn’t work, does it? Same principle applies here, on a molecular level. Our bodies are incredibly picky about which “hand” of a sugar they’ll accept.
So, what’s the actual name of this mirror-image counterpart to D-arabinose? Drumroll, please! It is L-arabinose. Yep, it's that straightforward and that complicated all at once! L-arabinose is the enantiomer of D-arabinose. It has the exact same chemical formula, the same atoms connected in the same order, but the spatial arrangement is flipped. Think of it as D-arabinose’s identical twin who was born in a parallel universe where everything is reversed.
It’s important to remember that while they share a name (arabinose), the "D" and "L" prefixes denote a significant difference. They are stereoisomers, specifically enantiomers. This means they have different physical properties, though some are very similar. For instance, their optical activity is equal in magnitude but opposite in sign. D-arabinose might rotate plane-polarized light clockwise, while L-arabinose would rotate it counterclockwise. Fancy, huh? It’s like one spin class makes you feel energized, and the other… well, maybe just dizzy.
Why does this matter? Well, think about the biological world. Our enzymes, those amazing molecular machines that drive all sorts of processes in our bodies, are often chiral themselves! They’re designed to work with specific enantiomers of molecules. So, while D-arabinose might be a building block for certain biological pathways, L-arabinose might be completely useless, or even interfere with things. It's like having a key that’s the wrong shape for the lock. It just won’t turn, no matter how hard you push.
D-arabinose itself is found in some natural products, like certain plant gums and glycosides. It's not as common as some other sugars, like glucose, but it’s definitely out there. And L-arabinose? Well, L-arabinose is actually found in nature too! It’s a component of some complex polysaccharides, like hemicellulose in plant cell walls. So, even though they are mirror images, both have their own little niches in the grand scheme of things. It's like having two different types of beautifully crafted cutlery; both are useful, but for different dining experiences.
Let’s visualize this a bit more. Imagine drawing D-arabinose on a piece of paper. You’ve got your carbon backbone, and then your –OH groups and –H atoms attached. Now, imagine holding that paper up to a mirror. The reflection you see is L-arabinose. If you tried to lay the original drawing perfectly on top of its reflection, you’d find that parts of it would stick out. The -OH on carbon 2 in D-arabinose might be on the right, but in L-arabinose, it would be on the left. The same reversal happens for the other chiral carbons. It’s a complete flip, top to bottom, left to right. It's not just a rotation; it’s a true reflection.
The concept of "D" and "L" comes from glyceraldehyde, a simpler sugar with just three carbons. D-glyceraldehyde has its hydroxyl group on the right in the Fischer projection, and L-glyceraldehyde has it on the left. All other D-sugars are related to D-glyceraldehyde by having the same configuration at their highest-numbered chiral center. And similarly, L-sugars are related to L-glyceraldehyde. It's like a family tree for sugars, and D-arabinose and L-arabinose are siblings, or rather, mirror-image siblings.
So, when you’re asked about the enantiomer of D-arabinose, you’re looking for its mirror image. And that mirror image, with all its spatial arrangements flipped, is L-arabinose. It’s a fascinating dance of molecules, a subtle ballet of atoms in three-dimensional space. It’s what makes organic chemistry so incredibly intricate and, dare I say, beautiful.
It’s like having two perfectly made shoes. One is for your left foot, and the other is for your right foot. You can't wear the left shoe on your right foot and expect it to fit comfortably, right? It just wouldn't work. Similarly, biological systems are often highly selective for one enantiomer over the other. This selectivity is what makes stereochemistry so important in fields like pharmacology and biochemistry. A drug might be incredibly effective in one enantiomeric form, while its mirror image is inactive or even harmful.
Think about how the taste receptors on your tongue work. They are chiral! They can distinguish between enantiomers. This is why, for example, one enantiomer of carvone smells like spearmint, while the other smells like caraway. Mind-boggling, isn't it? The subtle difference in three-dimensional shape leads to a completely different sensory experience. And it’s the same with sugars and their interactions with enzymes.
So, to recap our little chat over this imaginary coffee: D-arabinose is our starting point, a specific arrangement of atoms in space. Its enantiomer is its non-superimposable mirror image. And that, my friend, is L-arabinose. They are like two sides of the same coin, or rather, two hands that are reflections but can never perfectly align. It’s a fundamental concept in stereochemistry, and it explains so much about how molecules behave in the real world, especially in the intricate world of living organisms.
It’s not just about memorizing names; it’s about understanding the underlying principles of molecular structure and how those structures dictate function. D-arabinose and L-arabinose are a perfect example of this. They look so similar on paper, but their spatial orientations make all the difference. It’s a reminder that in the world of molecules, even the smallest differences in arrangement can have a huge impact. So, next time you see a D- or an L- prefix, remember that it’s not just a label; it’s a key to understanding the molecule’s unique three-dimensional personality!
And that’s it! We’ve navigated the chiral waters and found the mirror image of D-arabinose. It’s L-arabinose. Phew! Hope your coffee is still warm and you’re not too dizzy from all this molecular flipping. It’s a wild ride, but totally worth it, right? Keep those chemistry questions coming!
