Which Structural Formula Is Correct For 2 Methyl 3 Pentanol
Alright folks, gather ‘round, grab a virtual croissant, and let’s dive into a little chemistry caper that’s been causing some serious head-scratching. We’re talking about a molecule, a rather unassuming one, called 2-methyl-3-pentanol. Now, you might be thinking, “Chemicals? Sounds like my uncle Barry after his third sherry.” But stick with me, because this isn’t about lab coats and beakers; it’s more like a detective story with atoms as the suspects and a structural formula as the crime scene blueprint.
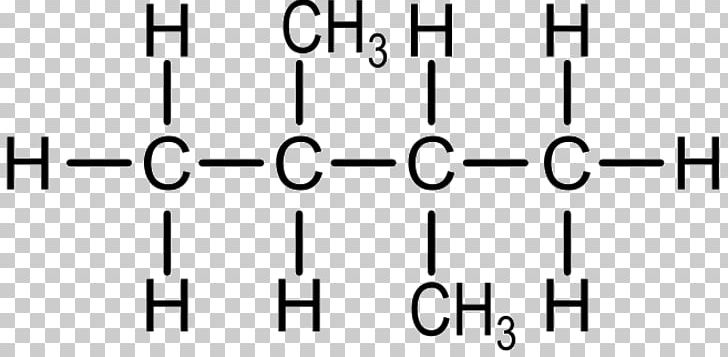
So, what’s the deal with 2-methyl-3-pentanol? Well, the name itself is like a cryptic crossword clue. Let’s break it down, shall we? Pentanol, that’s our main stage. Think of it as a five-carbon chain, the backbone of our operation. Like a nice, long baguette. And the ‘ol’ at the end? That’s chemistry-speak for an alcohol. So, we’ve got a five-carbon baguette with a hydroxyl group (that’s an OH, for the uninitiated, the equivalent of a tiny little life-giving disco ball) attached somewhere along the line.
But wait, there’s more! We’ve got a methyl group tagging along for the ride. That’s a single carbon atom with three hydrogens, CH3. Think of it as a tiny, mischievous gremlin, clinging to the baguette for dear life. And where is this gremlin hanging out? The name tells us: at position 2. So, on the second carbon of our five-carbon baguette, there’s a little methyl gremlin.
And then, the grand finale, the 3! This tells us the location of our precious OH disco ball. It’s chilling on the third carbon of the baguette. So, we’ve got a five-carbon chain, a methyl gremlin on carbon 2, and an OH disco ball on carbon 3. Easy peasy, right? Well, if you’re fluent in Chemicalese, maybe. For the rest of us, it’s like trying to assemble IKEA furniture without the instructions, but with slightly more potential for… explosions (okay, probably not explosions for this guy, but you get the dramatic flair).
The Suspects Emerge: Which Formula is the Real Deal?
Now, the fun part. When chemists draw these molecular blueprints, they have a few ways of doing it. You’ve got your super-detailed, every-atom-is-accounted-for kind of drawing. Then you have your more condensed versions, which are like the CliffsNotes of molecular structures. And sometimes, in the heat of the moment (or perhaps after one too many caffeine-fueled nights), things can get a little… ambiguous. This is where our structural formula mystery kicks in.
Imagine you’re at a café, and a chemist slams down two drawings. “Which one is 2-methyl-3-pentanol?” they exclaim, wild-eyed. One drawing might look something like this (imagine a bunch of lines and letters here, because I can’t actually draw them for you, but trust me, it’s a masterpiece of squiggles): a straight line of five carbons, a CH3 sticking off the second one, and an OH on the third. Simple, elegant, like a perfectly brewed espresso.
But then there’s the other drawing. And this is where the plot thickens. This second drawing might also look like it represents 2-methyl-3-pentanol. How can this be, you ask? Is it a quantum superposition of molecular structures? Did a rogue atom sneak in and change everything? Not quite. It’s usually a matter of perspective, or more accurately, how you number the carbon chain.
See, in the world of organic chemistry, we have rules for numbering carbon chains. It’s like giving everyone a seat at the dinner table. We usually start numbering from the end that gives our functional groups (like that OH disco ball) the lowest possible number. And substituents (like our methyl gremlin) also get the lowest numbers if there’s a choice. It’s all about finding the most efficient seating arrangement, or in chemistry terms, the most economical nomenclature.
The Case of the Wandering Numbers
So, let’s say we have a molecule. We can start numbering our five-carbon chain from the left, or from the right. If we number from the left, our OH group might be on carbon 3, and our methyl group on carbon 2. That fits our name, right? 2-methyl-3-pentanol. Huzzah!
But what if we were to number that exact same molecule from the right? Suddenly, our OH group might be on carbon 3 (hey, that’s the same!), but our methyl group would be on carbon 4! Now, that would be called 4-methyl-3-pentanol. Uh oh. This is where the confusion creeps in. It’s like calling your dog "Fido" one day and "Spot" the next – technically the same creature, but potentially leading to some very awkward introductions.
The key is that the name dictates the numbering. When we say 2-methyl-3-pentanol, we are prescribing a numbering system. We are saying, “For this specific molecule, I am declaring that the longest chain has five carbons, and when I number it according to the rules (lowest numbers for OH and methyl), the methyl is on 2 and the OH is on 3.”
So, if you see a drawing that, when numbered from the right according to the rules, gives you a methyl on carbon 4 and an OH on carbon 3, that drawing is not 2-methyl-3-pentanol. That would be its isomeric twin, hanging out in a parallel universe of chemical structures. The drawing that is 2-methyl-3-pentanol is the one that, when you apply the IUPAC (that’s the International Union of Pure and Applied Chemistry, the folks who make up all these rules – probably after a very long committee meeting fueled by lukewarm coffee) naming conventions, correctly yields the name 2-methyl-3-pentanol.

It’s a bit like a magic trick, but instead of a rabbit disappearing, it’s a structural formula that magically aligns with its proper name when you look at it through the lens of proper chemical etiquette. The surprising fact here is that, for even simple molecules, there can be multiple ways to draw them, and it’s the rules of naming and numbering that tell us which drawing is the official representation of that name.
Think of it this way: You can draw a picture of a person from the front or from the side. Both are valid representations, but they highlight different features. In chemistry, we have a preferred "pose" for our molecules, dictated by the naming rules, to ensure everyone is talking about the exact same chemical entity. So, the correct structural formula for 2-methyl-3-pentanol is the one that, when deciphered according to the established chemical language, unequivocally shouts, “I am 2-methyl-3-pentanol and not a moment sooner!” It’s a beautiful, if slightly pedantic, system.
And that, my friends, is the epic saga of 2-methyl-3-pentanol’s structural identity. It’s a reminder that even in the seemingly rigid world of chemistry, there’s a story, a logic, and a set of rules that make even the most complex molecules understandable. Now, who’s up for another round of molecular mysteries? Perhaps we can tackle 1,2-dimethylcyclopentane next. Just kidding… unless?
