Which Statements Accurately Describe Density Check All That Apply
Ever wondered why some things float and others sink? It's not magic, folks! It's all about something super cool called density. Think of it as how much "stuff" is packed into a certain space. We're diving into what that really means, and we've got some statements about density for you to ponder. Ready to become a density detective?
So, what's the big idea with density? Imagine you have a box. You can fill that box with a bunch of fluffy marshmallows, right? Or, you could fill the same exact box with heavy rocks. Which box would feel heavier? The one with the rocks, obviously! That's because the rocks are more densely packed than the marshmallows. There's more mass (that's the amount of stuff) in the same volume (the amount of space). Simple, huh?
Let's Get Down to Business: What's Density Really About?
Density is basically a measure of how compact something is. It's a ratio, meaning we compare two things: the mass of an object and its volume. The formula is pretty straightforward: Density = Mass / Volume. So, if you have a lot of mass crammed into a small volume, you've got high density. If you have less mass spread out over a larger volume, you've got low density.
Think about it like a crowded concert versus a wide-open park. The concert hall, with all those people packed in, is much denser than the park. You've got more bodies (mass) in a smaller space (volume) at the concert. Makes sense!
Now, for the Fun Part: Which Statements Ring True?
We're going to throw some statements your way about density. Your mission, should you choose to accept it, is to figure out which ones are spot on. Let's see if you can separate the scientifically sound from the… well, the less sound!
Statement 1: Density is a property of a substance that does not depend on the amount of the substance.
This one is a bit of a brain tickler, but it's super important! Let's say you have a big ol' chunk of gold. It has a certain density. Now, what if you cut that chunk in half? Does the density of that smaller piece change? Nope! The gold itself, whether it's a tiny fleck or a giant bar, has the same inherent compactness. It's like the "goldness" doesn't change just because you have less of it. So, density is an intensive property – meaning it doesn't change based on how much of the substance you have.
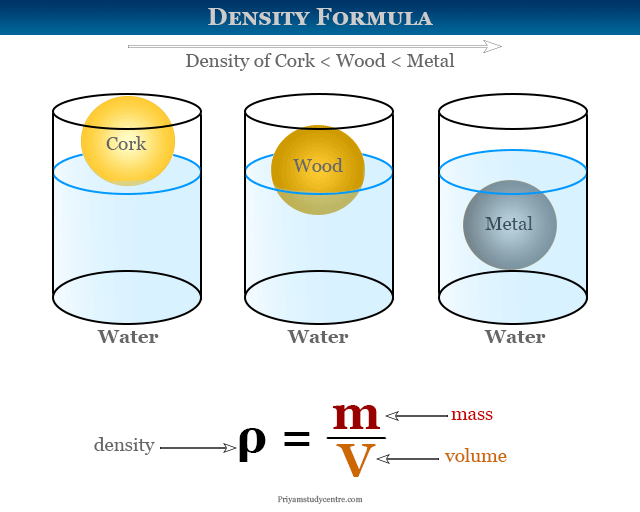
Statement 2: An object will float in a fluid if its density is greater than the density of the fluid.
Hold on a second, does this sound right? Think about a big, heavy battleship. It's made of steel, which is pretty dense. But it floats! And then you have a tiny pebble that sinks. This statement seems to go against our everyday experience, doesn't it? This is a classic trick! For something to float, its density must be less than the density of the fluid it's in. If it's denser, it sinks. If it's less dense, it floats. So, this statement is actually inaccurate.
Let's use a different analogy. Imagine you're trying to swim in a pool full of Jell-O. If you're super light and wiggly (low density), you'll probably bob around on top. If you're a heavy, dense boulder, you're going straight to the bottom! The Jell-O has its own density, and your body's density compared to the Jell-O's density determines whether you float or sink.
Statement 3: Density is calculated by dividing the mass of an object by its volume.

Remember our formula from earlier? Density = Mass / Volume. This statement is the absolute, undeniable truth! It's the very definition of density. If you want to know how dense something is, you need to know how much "stuff" is in it (mass) and how much space that "stuff" is taking up (volume). So, if someone asks you to calculate density, you know exactly what to do. This statement is definitely accurate.
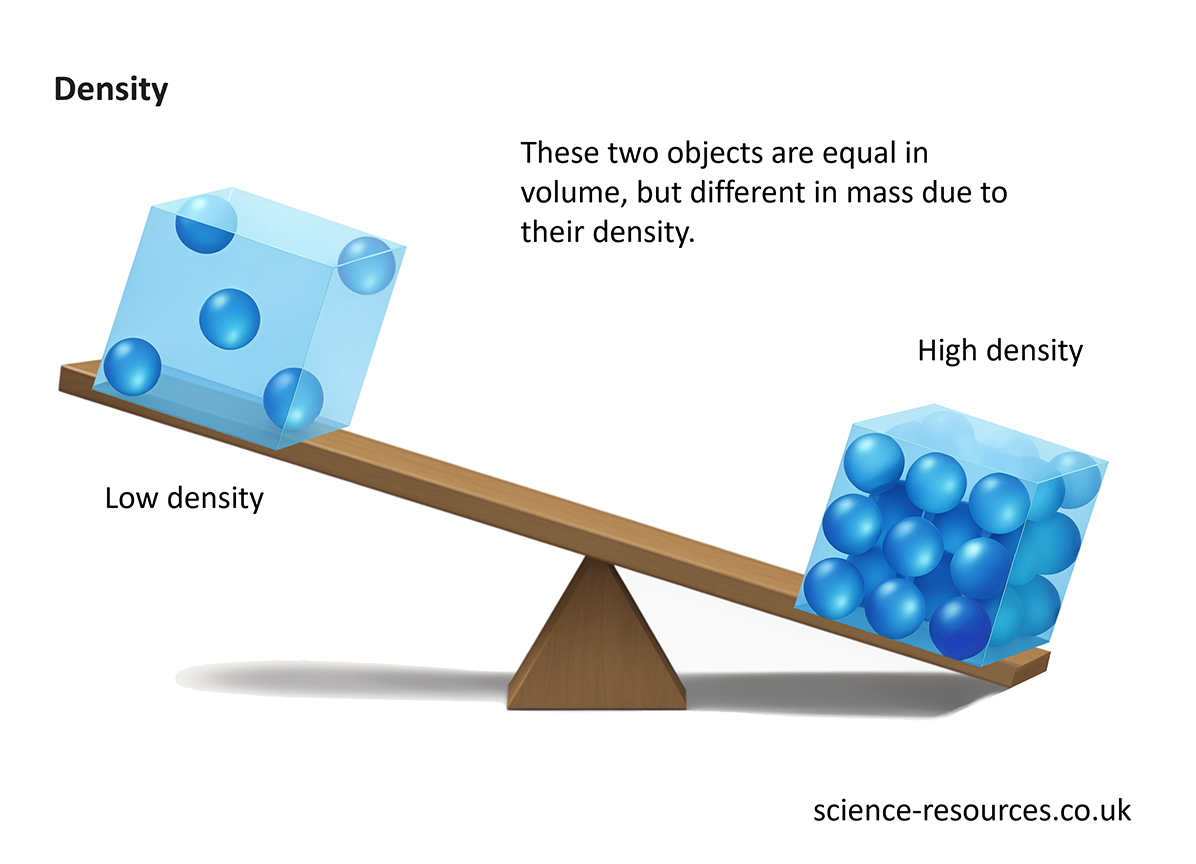
Statement 4: Two objects with the same volume will have the same density if they have the same mass.
Let's break this down. We know density is mass divided by volume. If two objects have the same volume, and we're also told they have the same mass, then what happens when we do the division? Mass / Volume will be the same for both! It's like saying if you have two identical boxes, and you fill both of them with exactly 10 ping pong balls, then both boxes will have the same "ping pong ball density." This statement is a direct consequence of the density formula and is therefore accurate.
Statement 5: An object will sink in a fluid if its density is less than the density of the fluid.
Okay, let's think about this one. This is the opposite of what we discussed for floating. If an object is less dense than the fluid it's in, it wants to rise to the top. It's like a helium balloon rising in the air. The air is denser than the helium. So, if an object is less dense, it's going to float, not sink. Therefore, this statement is inaccurate.
Think of it this way: if you throw a piece of cork into water, does it sink? No way! Cork is way less dense than water. It bobs happily on the surface. Now, imagine dropping a heavy rock into that same water. Splash! Down it goes. The rock is denser than the water.
Statement 6: Density is a measure of how much mass is contained in a given volume.
This is, in essence, the core concept of density! It's not just about mass, and it's not just about volume; it's about the relationship between them. How much "stuff" are you packing into a certain "space"? That's exactly what density tells us. It's a way to quantify that feeling of something being heavy for its size, or surprisingly light. This statement is a fantastic summary and is absolutely accurate.
So, there you have it! Density is a pretty neat concept that explains a whole lot of what we see in the world around us, from why ships float to why a tiny pebble sinks. It's all about how tightly packed that "stuff" is.
Which statements accurately describe density?
Based on our chat, the statements that accurately describe density are:
- Density is a property of a substance that does not depend on the amount of the substance.
- Density is calculated by dividing the mass of an object by its volume.
- Two objects with the same volume will have the same density if they have the same mass.
- Density is a measure of how much mass is contained in a given volume.
Keep pondering these ideas, and next time you see something float or sink, you'll have a clearer understanding of the amazing science behind it!
