Which Statement Is True Of All Atoms That Are Anions
Ever felt a little… charged? Like you’ve got a bit of extra zing? Well, atoms can feel that way too! And when they do, they become something pretty special: an anion. Think of them as the atoms who’ve decided to embrace their inner drama queen.
Now, you might think all atoms are basically the same, just chilling in their electron shells. But nope! Some are divas, demanding more. They’re the ones that are ready to collect extra goodies.
So, what’s the one big, undeniable truth about all these little drama queens, these fabulous anions? It’s surprisingly simple, and honestly, a little bit sassy.
The core of it all boils down to their party trick. They’ve all done the same thing. It’s not like one anion is collecting a single electron and another is hoarding a whole baker’s dozen. There’s a universal rule they all follow.
It’s about gaining something. Not losing, not sharing (though some atoms do that too, bless their hearts), but actively acquiring. They’re the ultimate collectors of the atomic world. Always looking for that next addition.
And what exactly are they collecting? These little opportunists are grabbing something negative. They’re not picking up positive vibes, oh no. That would be a whole other story.
We’re talking about electrons. Tiny, negatively charged particles. They’re like the atomic equivalent of finding a twenty-dollar bill on the sidewalk. A little unexpected bonus!
So, here’s the big reveal, the juicy gossip of the periodic table. Every single anion has done this one thing:

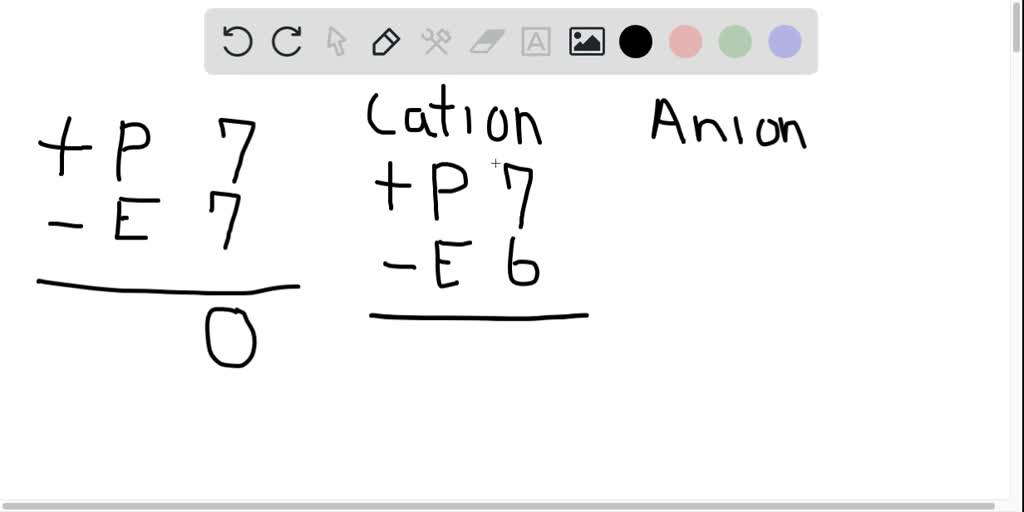
All anions have gained one or more electrons.
There. It’s as simple as that. No fancy jargon needed, no need to consult a crystal ball. Just a straightforward, universally accepted fact.
Think about it. A fluorine atom, the ultimate electron hog, is an anion because it’s snagged an extra electron. It's practically vibrating with negativity (in a good way!).
Then there’s oxygen. It’s pretty happy with its electron count, but if it sees an opportunity, it’ll grab not one, but two extra electrons. Talk about being greedy! But hey, that makes it an anion.
Even the big, lumbering atoms can get in on the action. Some of them, when they decide to be anions, might pick up a couple of electrons. They’re just more… substantial about it.
It’s like going to a potluck. Some people bring a small salad. Others bring a whole roasted turkey. But everyone who brings something to the table is participating in the communal feast. Anions are just the ones who brought extra plates… of electrons!
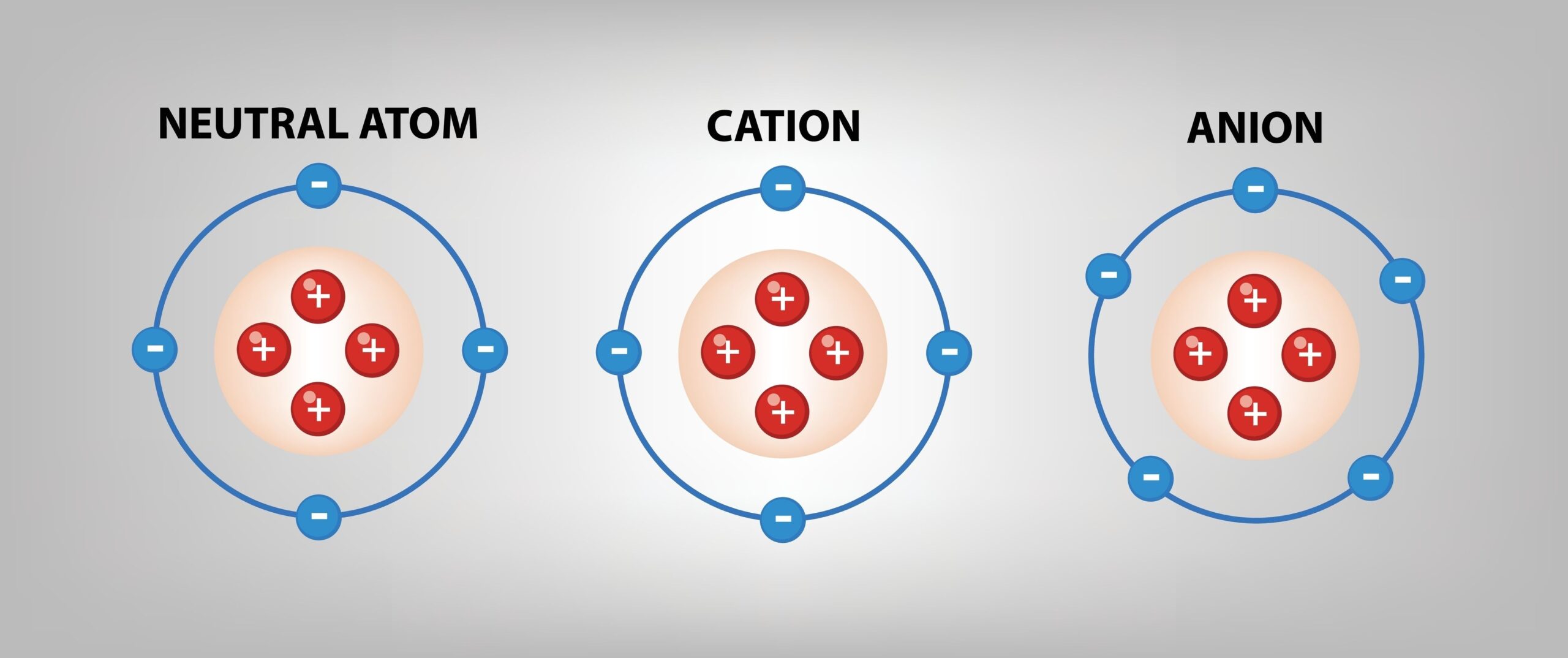
The key takeaway here is the gain. It’s the opposite of their cousins, the cations. Those guys are the ones who lose electrons, becoming positively charged. They’re the ones giving away the goodies, not hoarding them.
But anions? They’re the collectors. They’re the hoarders. They’re the ones who always seem to have a little something extra. It's a fundamental part of their identity.
Imagine an anion walking into a party. Everyone else is just existing, doing their atomic thing. But the anion? It’s got an extra electron tucked away. It's got a secret. It's a little bit more.
This gain of electrons is what gives them their characteristic negative charge. It’s the reason they’re attracted to positive things, like a moth to a flame. It’s their superpower, in a way.
So, next time you hear the word anion, just picture an atom who decided to hit the electron jackpot. They’ve got more than they started with. They’re a little bit heavier, a little bit more negative, and a whole lot more interesting.
It’s a simple concept, but it’s the bedrock of what makes an anion an anion. It’s not about how many protons they have, or how many shells they’ve filled. It’s all about that electron acquisition.
+anions+-+atoms+of+an+element+that+have+more+electrons+than+their+neutral+atom+(non-metals+form+anions).jpg)
And here’s my slightly unpopular opinion: I think anions are a little bit cooler. They’re the ones who are always evolving, always adding to themselves. They’re not content with the status quo.
They’re the ones who embrace change, who say, "You know what? I could use a little more something." And then they go out and get it! It’s a proactive approach to life, wouldn’t you say?
So, while cations might be all about letting go, anions are about grabbing on. They’re the accumulators of the atomic universe. And it’s this very act of accumulation that defines them.
It’s like finding a perfectly ripe berry on a bush. Some might just admire it. But an anion? It’s going to pluck it right off and make it its own. And that’s why it’s an anion.
The truth is, it’s a beautifully simple rule. No exceptions. No loopholes. Just a straightforward statement of atomic intention and action.

Every single atom that has, at some point, decided to bring more electrons into its orbit, is an anion. It’s their defining characteristic, their atomic swagger.
So, the next time you’re pondering the mysteries of the universe, the vastness of space, or even just what to have for dinner, remember the humble anion. They’ve got a little secret, a little extra something, that makes them unique. And that something is always, always, a gained electron (or more!).
It’s the ultimate underdog story. They weren't enough, so they made themselves more. And in the atomic world, that’s a pretty big deal.
So, let’s raise a metaphorical beaker to the anions of the world! The collectors, the accumulators, the ones who are always a little bit more. Because they’ve all done the same thing, and that’s pretty darn cool.
It’s not about being good or bad, it’s about a fundamental chemical action. And for anions, that action is always about gaining those precious, negatively charged electrons.
So there you have it. The undisputed, the undeniable, the slightly dramatic truth about all anions. They’ve all snagged themselves some extra electrons. End of story. Well, not really the end, because their story is just beginning with that extra negative charge!
