Which Statement Is Always True For Exothermic Reactions

Have you ever wondered what happens when things get a little heated? Not just in a "heated debate" kind of way, but in a real, actual, science way. We're talking about something called an exothermic reaction. It sounds super fancy, right? But honestly, it's one of the coolest and most dependable things in the whole universe.
Think about it like this: sometimes, when chemicals get together, they decide to have a little party. And during this party, they release a whole bunch of energy. It's like they're saying, "Here, have some of this!" It’s this release of energy that makes exothermic reactions so predictable and, dare we say, a little bit magical.
So, what's the one thing you can absolutely, positively, 100% count on with these energetic chemical celebrations? There's a statement that's as solid as a rock, and it tells us exactly what's going on under the hood. It’s the secret handshake of the exothermic world.
The Big Reveal: What's Always True?
Alright, drumroll please! The statement that is always true for an exothermic reaction is this: Energy is released into the surroundings.
That's it! Simple, right? No complex jargon, no confusing formulas (for now, anyway!). Just a straightforward observation about what these reactions do. They give off energy. It’s like a chemical generosity.
Imagine you're baking cookies. When you mix the ingredients and put them in the oven, something happens. The cookies bake, and they give off a lovely aroma. That aroma is a bit like the energy released in an exothermic reaction. It's a sign that something significant has occurred.
But with exothermic reactions, it's not just a smell. It's heat! Or light! Or sometimes even sound. They're basically shouting, "Look at me, I'm doing something awesome!" And that "awesome thing" is always accompanied by a giveaway of energy.
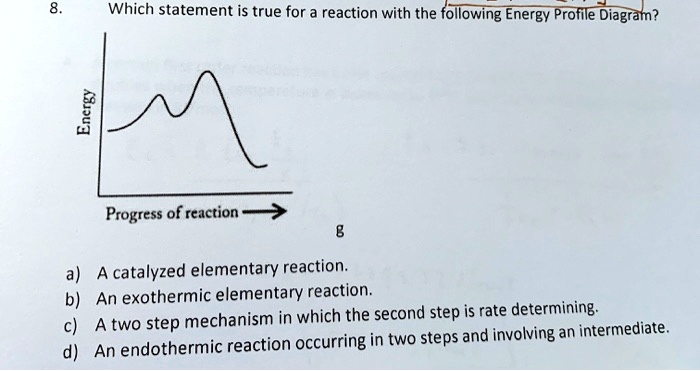
Why This Is So darn Cool
Now, why is this so entertaining? Because in science, finding something that's always true is like finding a unicorn. There are so many variables, so many "it depends" scenarios. But with exothermic reactions, this one thing is a constant. It's the anchor in a sea of chemical possibilities.
Think about it from a scientist's perspective. If you're trying to build something new, or understand a process, having a reliable rule to fall back on is incredibly valuable. It helps you predict outcomes and design experiments with confidence. It’s like having a cheat code for understanding nature.
And for us regular folks, it helps us understand the world around us a little better. When you feel warmth from a hand warmer, or see a bright spark from a firework, you're witnessing an exothermic reaction in action. That "energy released" statement is the key to unlocking that understanding.
It’s also incredibly fun because it’s so tangible. We can feel the heat, we can see the light. It’s not some abstract concept that’s floating around in the ether. It’s happening right in front of us, and we know, without a doubt, that energy is being let loose.

"The universe loves a good giveaway, and exothermic reactions are its enthusiastic participants!"
This fundamental truth about exothermic reactions is what makes them so widely used. From the fuel that powers our cars to the reactions that keep our bodies alive, energy release is a key player. It’s the engine that drives so many processes we take for granted.
Let's Get a Little More Specific (But Still Fun!)
When we say "energy is released," what kind of energy are we talking about? Mostly, it's in the form of heat. That's why many exothermic reactions feel warm to the touch. They're essentially heating up their surroundings.
But sometimes, it’s more dramatic! Think of a chemical reaction that produces a bright flash of light. That's also energy being released, just in a different form. Fireworks are a perfect example of exothermic reactions showing off their light-producing capabilities.
And occasionally, you might even hear a little pop or fizz. That sound is also a manifestation of the energy being let go. So, it's a multi-sensory experience, this exothermic business! It engages more than just one of our senses.
This ability to release energy is crucial. It means that the products of the reaction have less energy than the starting materials. Think of it like a pendulum at the top of a swing versus at the bottom. The pendulum at the bottom has less stored energy.

So, the reactants are like coiled springs, full of potential energy. When the reaction happens, they uncoil, and that stored energy has to go somewhere. And where does it go? Out into the world, in the form of heat, light, or sound. It’s a chemical unburdening.
This fundamental principle is often represented by a concept called enthalpy change. For exothermic reactions, this change is negative. It’s a mathematical way of saying that energy has left the system. It's the scientific nod to the energy giveaway.
But don't let the fancy term scare you! At its heart, it's just another way of saying that energy is being released. The negative sign is just a reminder that something is leaving, like a withdrawal from a bank account of energy. It’s a depletion of internal energy.
Why Does This Make Us Curious?
What makes this so engaging is its implication of power. Exothermic reactions are the workhorses of the chemical world. They provide the energy for so many things we rely on. From the gentle warmth of a burning candle to the immense power of an explosion, it's all about energy release.

Imagine the potential! Scientists and engineers harness this predictable energy release to create everything from medicines to new materials. They understand the rule, and they use it to their advantage. It’s like knowing the secret to a magic trick and using it to perform amazing feats.
It also makes us think about the opposite: endothermic reactions. Those are the ones that absorb energy. They get cold instead of hot. Comparing the two helps us appreciate the distinct personalities of different chemical reactions. It's the contrast that makes the story more interesting.
So, the next time you feel warmth from something, or see a bright light, take a moment to appreciate the exothermic reaction at play. Remember that statement: energy is released into the surroundings. It’s a simple truth, but it’s the key to understanding a vast and energetic part of our world. It's a fundamental law of chemical interaction.
And who knows, maybe this little glimpse into the energetic world of exothermic reactions will spark your own curiosity. Perhaps you’ll start noticing these energy releases everywhere. It's a fascinating journey, and it all starts with knowing that one, always-true statement. It’s a gateway to a universe of chemical wonders.
So, go forth and observe! Notice the warmth, the light, the energy. It’s all around you, a constant reminder of the power and predictability of exothermic reactions. They're the unsung heroes, providing the spark, the heat, and the energy that makes our world go 'round. It's a truly electrifying concept.
