Which Statement Describes The Electron Sharing Between Hydrogen And Fluorine
Hey there, fellow chill seekers and curious minds! Ever find yourself staring at a lightbulb and wondering, "What's actually going on in there?" Or perhaps you've been mixing up some baking soda and vinegar for a science fair volcano (or just for fun, no judgment here!), and pondered the magic behind it all? Today, we're diving into the tiny, yet epic world of atoms, specifically the fascinating dance between two energetic buddies: hydrogen and fluorine. Forget stuffy textbooks; we're talking about this like we're swapping stories over a perfectly brewed cup of coffee.
So, the big question is: Which statement describes the electron sharing between hydrogen and fluorine? It’s a bit like asking how two people become best friends, or how a dynamic duo gets their super powers. And the answer, my friends, is all about the art of covalent bonding. Specifically, it’s about something called a polar covalent bond. Pretty cool, right? Let's break it down, no complex jargon allowed, just good vibes and a sprinkle of science.
The Atomic BFFs: Hydrogen and Fluorine
Imagine atoms as tiny little characters, each with their own personality and preferences. Hydrogen, our first pal, is the simpler one. Think of it as a single, enthusiastic person always looking for someone to hang out with. It has just one electron, and it’s really eager to complete its outer shell, which is like its personal bubble of happiness. Just one more electron, and hydrogen is content.
Now, meet Fluorine. This atom is a bit more intense. It’s got a whopping seven electrons in its outer shell. Fluorine is like that friend who’s always a little anxious about not having enough – it’s desperately seeking that one missing electron to feel complete. It’s got a really strong pull, a magnetic personality, if you will, for electrons.
When these two meet, it’s not about one atom completely stealing the other’s electron. That’s a different kind of relationship, called an ionic bond, where one atom is the generous giver and the other is the grateful receiver. Think of it like a parent giving a child an allowance. But with hydrogen and fluorine, it’s more of a partnership.
The Sharing Economy of Electrons
This is where the magic of covalent bonding comes in. Instead of one atom hogging an electron, they decide to share. It’s like two friends deciding to share a Netflix password instead of each buying their own subscription. They both get access, and everyone’s happy, right? Well, sort of. In the case of hydrogen and fluorine, this sharing isn't perfectly equal.
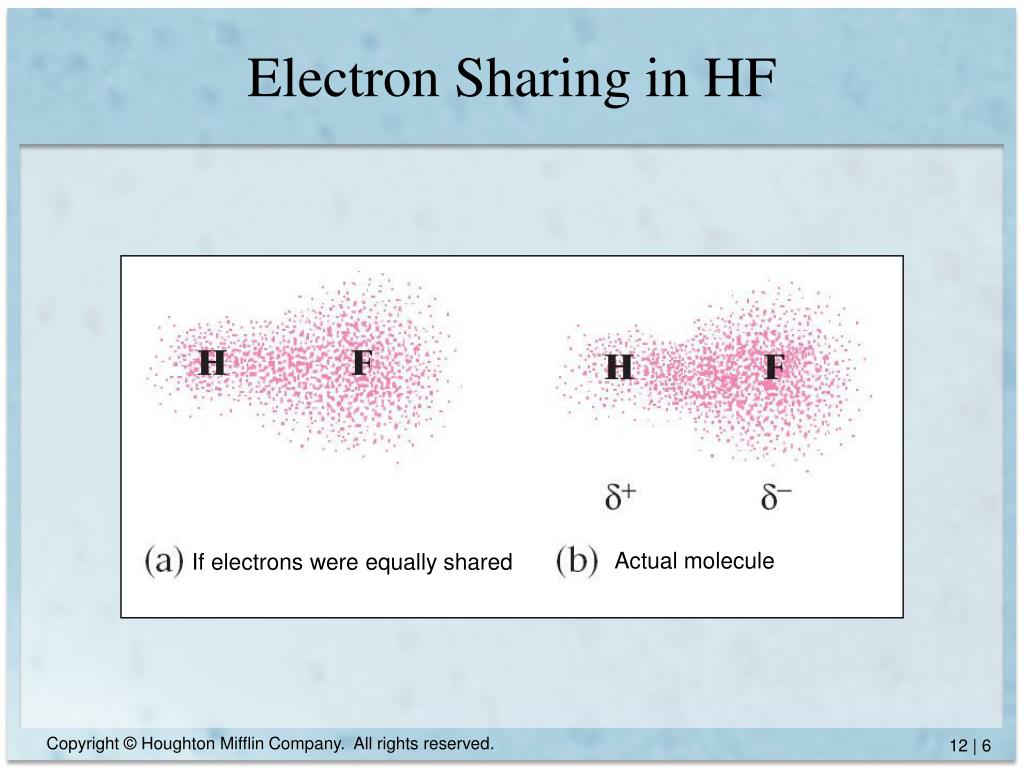
This is the crucial part: the electron sharing between hydrogen and fluorine is unequal. Fluorine, with its super strong pull, tends to hog the shared electrons a little bit more than hydrogen does. Imagine two people sharing a blanket on a chilly evening. One person might be a little more prone to pulling the blanket closer, especially if they tend to get colder faster.

Fluorine is much more electronegative. That’s a fancy science word for "really good at attracting electrons." It’s got this powerful charisma that draws the shared electrons closer to its nucleus. Hydrogen, being less electronegative, has to let its buddy Fluorine have a bit more of the electron attention. This creates a subtle, yet significant, imbalance.
Polarity: The Imbalance That Makes Things Interesting
Because fluorine pulls the shared electrons closer, the area around the fluorine atom becomes slightly negative. Think of it like a magnet. The electron cloud is denser, more concentrated, around fluorine, giving it a partial negative charge. Conversely, the area around the hydrogen atom, where the electrons are spending less time, becomes slightly positive. It's like the opposite pole of the magnet.
This unequal sharing is what we call a polar covalent bond. The molecule, in this case, hydrogen fluoride (HF), isn't perfectly neutral. It develops poles, just like a tiny bar magnet: a slightly negative end and a slightly positive end. This polarity is the secret sauce that makes water, for instance, such an amazing solvent, and it’s fundamental to all sorts of chemical reactions and biological processes.
Think about it like this: you and your best friend are sharing a pizza. If one of you eats a slightly larger slice (but you still share the pizza!), that doesn't mean one person isn't eating. It just means there's a slight preference. In HF, fluorine is the one taking the slightly larger bite of the electron pie.

Why Does This Matter? Practical Perks of Polarity
Okay, so atoms are sharing unevenly. Big deal, right? Well, this seemingly small detail has huge implications for how molecules interact and behave. It’s the foundation of so many things we see and use every day.
The Water Wonder
You know how water is often called the "universal solvent"? That’s largely thanks to the polar nature of its hydrogen and oxygen atoms. Because water molecules are polar, they can attract and dissolve other polar substances. It’s like having a really friendly host at a party – they can easily connect with lots of different guests. This is why salt dissolves in water, why your tea bag works, and why life as we know it is even possible on Earth!
Imagine trying to mix oil and water. They don't mix, right? That's because oil molecules are nonpolar – they're like a group of people who all have the same interests and don't really interact with those who are different. Polar molecules, like water, have those slight charges that allow them to attract and surround other polar molecules, pulling them into solution. It’s a beautiful dance of intermolecular forces.
From Your Toothpaste to Your Biology
Even in simple things like toothpaste, polarity plays a role. Fluoride ions (which come from compounds containing fluorine) are added to toothpaste to strengthen tooth enamel. This works because of the way fluoride interacts with the minerals in your teeth, a process influenced by its electronegativity and ability to form bonds.
In your own body, countless chemical reactions rely on polar bonds. The way proteins fold, how DNA replicates, and how your cells communicate – all of it involves the subtle tug-of-war of electrons and the resulting polarities of molecules. It's like the intricate wiring in a smart home; small differences in how signals are transmitted can lead to complex and vital functions.
Fun Facts and Analogies to Make it Stick
Let's sprinkle in some fun! You know how in some sitcoms, you have a couple where one person is super organized and the other is a bit more laid-back? The organized one might keep the shared calendar updated with all the details, while the laid-back one might just check it occasionally. That's a bit like the hydrogen-fluorine dynamic. Fluorine is the über-planner, keeping the electron-schedule packed!
Or think about a popular influencer on social media. They have a huge following, and their posts get a lot of attention (electrons). Their followers are drawn to them. Fluorine is like that influencer, drawing in those electrons because it’s just that popular and has a strong appeal. Hydrogen is like a dedicated fan, happy to be part of the influencer's orbit, even if they don't get all the spotlight.
The electronegativity difference between hydrogen and fluorine is one of the largest among common pairs. This is why the bond is so distinctly polar. It's not a mild disagreement about who gets the remote; it's more like a polite but firm insistence on a favorite channel!

The Science of Everyday Strength
The strength of a covalent bond, and its polarity, is a major factor in the stability of molecules. This means that the substances we rely on, from the air we breathe to the medicines we take, are stable and predictable because of these precise atomic interactions. It’s the invisible scaffolding holding our material world together.
Consider the humble glass of water. It seems so simple, but the way its molecules stick together (due to those polar attractions, called hydrogen bonds!) is what gives water its surface tension, allows plants to draw water up their stems, and creates the very oceans that cradle life.
A Final Thought on Sharing and Connection
So, back to our initial question: Which statement describes the electron sharing between hydrogen and fluorine? The most accurate description is that it’s an unequal sharing of electrons, forming a polar covalent bond. Fluorine, being more electronegative, attracts the shared electrons more strongly, creating a partial negative charge on fluorine and a partial positive charge on hydrogen.
It’s a beautiful illustration of how even in the smallest interactions, there’s a complex interplay of attraction, influence, and balance. It reminds me that in our own lives, relationships are rarely perfectly 50/50. We share our time, our energy, our resources, and sometimes, one person might naturally take the lead or offer more support in certain moments. It doesn't make the sharing any less valuable or the connection any less strong. It just makes it real.
Just like hydrogen and fluorine find a stable, albeit unequal, partnership, our own connections thrive on understanding these subtle dynamics. So next time you’re enjoying a glass of water or even just breathing in the air around you, take a moment to appreciate the incredible, invisible world of atomic relationships and the polar magic that makes it all happen. Keep that curiosity flowing, and stay wonderfully balanced!
