Which Statement Correctly Describes The Ka Of A Weak Acid

Hey there, fellow humans! Ever find yourself staring at a label on a cleaning product, or maybe even something you're eating, and you see all sorts of scientific-sounding words that make your brain do a little wobble? One of those tricky terms that pops up sometimes is Ka, especially when we're talking about weak acids. Now, before you start picturing lab coats and bubbling beakers, let's make this super simple and, dare I say, even a little fun. Because, believe it or not, understanding this "Ka thingy" can actually be kinda useful in our everyday lives!
So, what's the big deal with weak acids? Think about the stuff we use all the time. Lemon juice? Yep, that's a weak acid. Vinegar? Another one! Even the tiny bit of acid in your stomach to help digest food? You guessed it. These aren't the hydrochloric acid that'll melt through your sink (thank goodness!). These are the polite, well-behaved acids of the world. They do their job without being overly dramatic.
Now, these weak acids have a little secret weapon, a way of showing us just how willing they are to be acids. And that's where our friend, Ka, comes in. Think of Ka as a scorecard for how good a weak acid is at doing its acid thing. Specifically, it tells us how much that weak acid is willing to dissociate. Whoa, big word! Let's break that down.
Imagine you have a little group of friends, and they're all linked up. Let's call them the "Acid Buddies." When these Acid Buddies are hanging out in water (because most of our chemistry happens in water, like a giant communal swimming pool), some of them might decide to break away from the group and go their own way. They're still friends, but they're now doing their own thing in the water. This breaking away is called dissociation. The acid molecule splits into two smaller pieces, and one of those pieces is what gives acids their acidic properties (usually a hydrogen ion, but let's not get bogged down in that detail right now).
So, a weak acid is one that doesn't totally break apart. It's like a family reunion where most people stay together, but a few cousins might wander off to chat with other relatives. A strong acid, on the other hand, is like a confetti cannon – it just explodes into a million pieces! We're focusing on the ones that are a bit more reserved, the weak acids.
Here's where Ka becomes our hero. The Ka value is basically a number that tells us how easily that weak acid will let its buddies go off and do their own thing. A higher Ka value means the acid is more likely to dissociate. It's like saying, "Yeah, sure, go ahead and mingle! The more, the merrier!" These acids are pretty keen on being acids.

A lower Ka value means the acid is a bit more hesitant. It's more like, "Hmm, maybe we should all stick together? It's safer in the group." These acids don't dissociate as much. They're more content staying as one unit.
Let's try a little story. Imagine you have two types of party hosts. Host A is super enthusiastic and as soon as guests arrive, they're pushing everyone onto the dance floor, introducing them to strangers, and breaking up any little cliques. Host B is more chill. They let guests mingle, but most people tend to stick with their initial friends for a while before maybe, just maybe, striking up a conversation with someone new.
In this analogy, Host A is like a weak acid with a high Ka. They're really good at getting their "guests" (the parts of the acid molecule) to go their separate ways. Host B is like a weak acid with a low Ka. They're a bit more reserved, and not as many guests end up splitting off.

So, when we're looking at statements about the Ka of a weak acid, we're essentially looking for the one that accurately describes this willingness to dissociate. It's not about the acid being "good" or "bad," it's about its tendency to break apart in water.
Here's the tricky part, and where you might see some confusing statements. People might try to trick you by saying things like:
• "A weak acid with a high Ka is a strong acid." (Nope! A weak acid is always weak. A high Ka just means it's a stronger weak acid compared to others with lower Ka values.)

• "A weak acid with a low Ka doesn't dissociate at all." (Not true! It dissociates, it just does so to a lesser extent.)
• "Ka tells you how corrosive an acid is." (Not directly! While acidity relates to corrosiveness, Ka specifically measures the equilibrium of dissociation.)
The correct statement will always link Ka to the degree of dissociation of a weak acid in a specific solution (usually water).
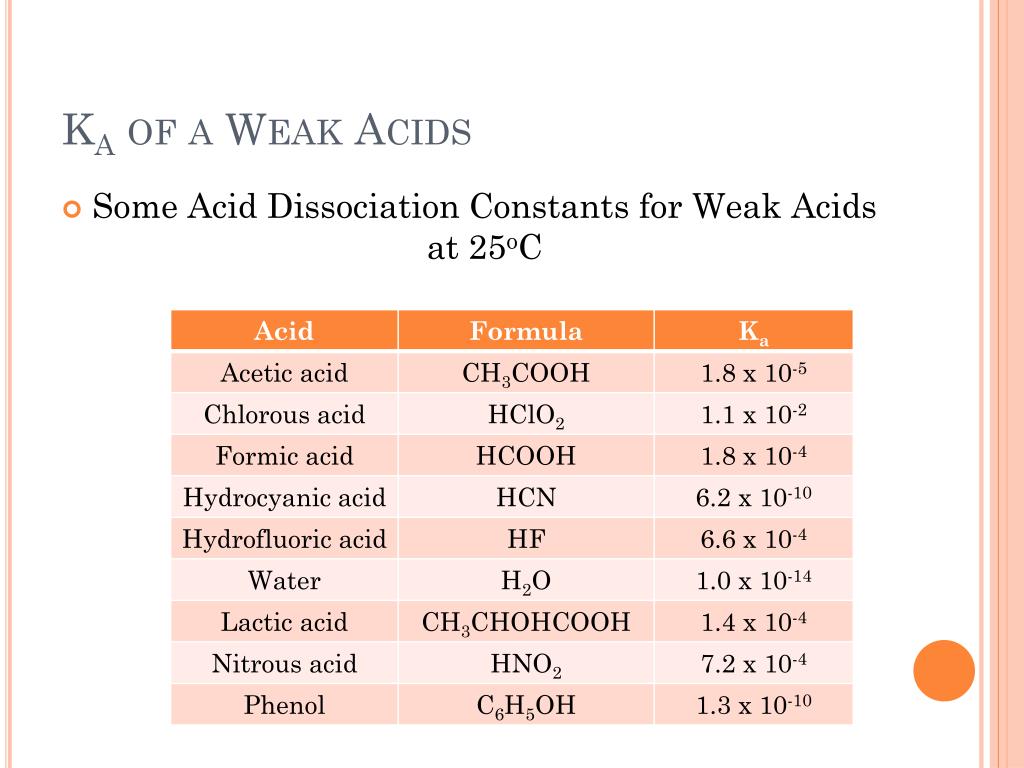
Why should you care about this little number, Ka? Well, think about cooking. If you're making a marinade with vinegar, understanding how acidic it is (and therefore how much of the acid is actually doing its job) can affect how tender your meat gets. Or, when you're buying skincare products, some ingredients are acids that help exfoliate your skin. Knowing the Ka of those acids can give you an idea of how potent they might be.
It also helps us understand things like how solutions will behave. If you're trying to make a buffer solution (that’s a whole other cool topic, but it involves keeping pH stable), the Ka of the acid you're using is super important.
Ultimately, the Ka value for a weak acid is a measure of its strength in terms of how much it dissociates in water. A higher Ka means more dissociation, and a lower Ka means less dissociation. It’s like a dimmer switch for acidity – some weak acids are on almost full brightness, while others are dialed down quite a bit.
So, the next time you see "Ka" in relation to a weak acid, just remember our party host analogy or the scorecard. It's all about how readily that acid buddy decides to go off and be an individual in the water. And knowing that can make you feel just a little bit more in control when deciphering those scientific labels!
