Which Statement Correctly Describes The Electrons In A Water Molecule

Get ready to dive into the amazing world of water molecules, folks! We're going on a thrilling adventure to uncover a tiny secret about these everyday heroes. You know, the stuff that makes up oceans, your morning coffee, and even that refreshing sip on a hot day. It's everywhere, and today, we're going to shine a spotlight on the tiny, zippy things that make it all happen: electrons!
Think of a water molecule like a super cute, microscopic party. It's got a main star, a big ol' oxygen atom, who's kind of the life of the party. And then there are two smaller, enthusiastic attendees, the hydrogen atoms, who are just bouncing with excitement. They're all huddled together, holding hands, and doing a little dance that makes water, well, water!
Now, what are these electrons we keep talking about? Imagine them as the tiny, super-energetic party favors at our water molecule bash. They're so small you can't even see them with your regular eyeballs, but they're incredibly important. These little guys are the real movers and shakers of the molecular world!
So, the big question is, how are these electrons behaving in our little water party? Are they all chilling in one spot, maybe hogging all the good snacks? Or are they spread out evenly, like a perfectly portioned dessert platter? This is where things get really interesting, and a little bit like a thrilling detective story.
Let's break down the players. We have our central oxygen atom. Think of oxygen as the generous host, but with a bit of a clingy personality when it comes to electrons. It really, really likes to have them around.
Then we have our two hydrogen atoms. These guys are happy to share, but they're also a little bit shy when it comes to electrons. They don't have as much "pull" as the oxygen.
So, when the oxygen and hydrogens decide to buddy up and form a water molecule, something super cool happens with their electrons. It's like a tug-of-war, but a friendly one, where everyone still gets to play. The electrons aren't completely stolen; they're just a bit more drawn to one atom than the others.

Imagine you're sharing a big, delicious pizza with your friends. The oxygen is like the friend who really, really loves pizza and has a slightly bigger slice. The hydrogens are like the friends who are happy with their slices, but maybe a little less enthusiastic about the pizza itself.
In our water molecule, the electrons are like the cheese and toppings that make the pizza so amazing. The oxygen atom is a bit greedier for these electron toppings. It pulls them closer to itself, making that side of the molecule a little bit more "negative" in charge. It's like the oxygen is saying, "Mmm, more electrons for me, please!"
The hydrogen atoms, on the other hand, are left with a little less electron attention. This makes their sides of the molecule a bit more "positive" in charge. They're not left completely bare; they still have some electrons visiting, but not as many as the oxygen. It's like they're saying, "Okay, I'll take what's left, thanks!"
So, what statement correctly describes this electron situation? It's not that the electrons are completely transferred from the hydrogens to the oxygen, like giving away your entire toy collection. That would be a whole different story!
Nor is it that the electrons are shared perfectly equally, like dividing a bag of candies into exactly identical piles for everyone. That would be a super chill, perfectly balanced scenario. But that's not quite what's happening in a water molecule.
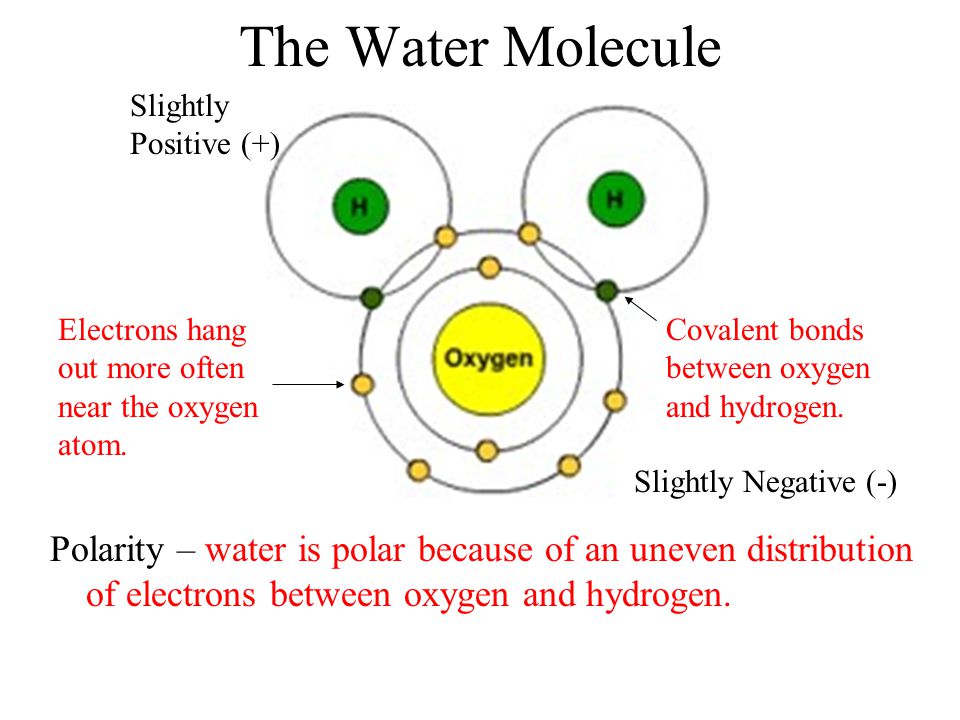
Instead, the electrons are shared, but in a way that's a little bit lopsided. They spend more time hanging out with the oxygen atom than with the hydrogen atoms. This unequal sharing is the secret sauce that gives water its unique properties.
This is why a water molecule is called a polar molecule. Think of it like a tiny magnet, with a slightly positive end and a slightly negative end. That uneven distribution of electron "goodness" is what makes it so.
So, when you see a statement that says the electrons are unequally shared, and that they are more attracted to the oxygen atom, you've hit the jackpot! You've found the correct description of the electrons in a water molecule. They're not just lounging around; they're actively participating in a dynamic, slightly biased, but ultimately harmonious arrangement.
This electron dance is what allows water molecules to stick together, to form waves, to dissolve so many things, and to be the incredible life-giver it is. It’s all thanks to this little tug-of-war over electrons. It’s a beautiful display of atomic cooperation, with a touch of favoritism!
So next time you take a drink of water, or see a rain drop, remember the tiny, zippy electrons and their important, unequal sharing. They're the unsung heroes of our watery world, and their behavior is truly something to marvel at. It's a small detail that makes a huge difference, proving that even the tiniest things can have the biggest impact.

Isn't science just wonderfully wild? It's like a cosmic game of show and tell, where even the most basic substances have dazzling secrets to reveal. And the electrons in water? They're definitely putting on a fantastic show!
So, to recap the excitement: the electrons in a water molecule are shared, but with a clear favorite. They're not running away, and they're not perfectly balanced. They're just a little bit more cozy with the oxygen, creating that special, polar personality that makes water so utterly fantastic.
It's this precise electron arrangement that allows water to do all its amazing tricks. From its ability to act as a universal solvent (dissolving your sugar in tea!) to its role in keeping us alive, it's all down to this electron shuffle. They’re the tiny conductors of a grand symphony!
The oxygen atom, with its eight protons, has a stronger positive pull in its nucleus. This stronger pull is what tugs those shared electrons closer. Think of it like a stronger magnet attracting metal filings.
The hydrogen atoms, with their single proton, have a weaker pull. So, the electrons don't get as close to them. It's like having a weaker magnet trying to attract the same filings – they just don't grab on as tightly.

This creates a partial negative charge on the oxygen side of the molecule, making it a bit like a tiny, friendly vacuum cleaner for electrons. And on the hydrogen sides, you get partial positive charges, like little spots that are slightly "less" electron-rich. It's a subtle but powerful difference.
So, the statement that captures this beautifully is the one highlighting unequal sharing and a stronger attraction to oxygen. This is the key to understanding water's incredible behavior. It's the reason why water molecules can form those super-important hydrogen bonds, which are like little intermolecular hugs that hold water together.
These hydrogen bonds are what give water its high boiling point (it takes a lot of energy to turn liquid water into gas!), its surface tension (why bugs can walk on water!), and its ability to expand when it freezes (why ice floats!). All of this amazingness stems from that simple, unequal sharing of electrons. It’s a testament to the profound impact of tiny things.
So, congratulations, you've unlocked a secret of the universe, a tiny detail about the most common substance on Earth! You've learned about the electron dance in a water molecule, and how it makes our world so wonderfully wet and alive. Keep exploring, keep questioning, and keep enjoying the marvels of science!
The statement that correctly describes the electrons in a water molecule is that they are unequally shared, spending more time near the oxygen atom than the hydrogen atoms.
