Which Statement Correctly Describes A Reaction In Dynamic Equilibrium

Ever feel like life is just a giant juggling act? You're trying to keep a million things in the air – work, family, that rogue sock that keeps disappearing in the laundry – and somehow, things just… stay up there, right? Well, get ready to have your mind tickled, because the amazing world of chemistry has a concept that's eerily similar, and it’s called dynamic equilibrium!
Imagine a super popular ice cream shop. On a hot summer day, there's a constant stream of people going in to buy their favorite scoops, right? But there's also a steady flow of people coming out after enjoying their delicious treats. Now, here's the magic trick: even though there are tons of people moving, the number of people inside the shop stays pretty much the same. It's not like the shop is a black hole that swallows people forever, and it's not like everyone magically teleports back outside. People are going in and people are coming out, at the same darn rate!
That, my friends, is the essence of a reaction in dynamic equilibrium. It’s not a boring, static situation where nothing is happening. Oh no! It’s a party, a bustling marketplace, a never-ending dance where two things are happening at the exact same speed, canceling each other out so the overall picture looks like it's not changing. It’s the ultimate masterclass in balance!
Let's dive a little deeper. In chemistry, we often talk about "reactions." Think of them like little chemical transformations, where one set of ingredients (we call them reactants) turns into something new (we call them products). But here's the kicker: sometimes, those new things (the products) can turn back into the original ingredients (the reactants)! It's like baking a cake and then, poof, it magically turns back into flour, eggs, and sugar. Except, in chemistry, this can happen all the time, like a chemical boomerang.
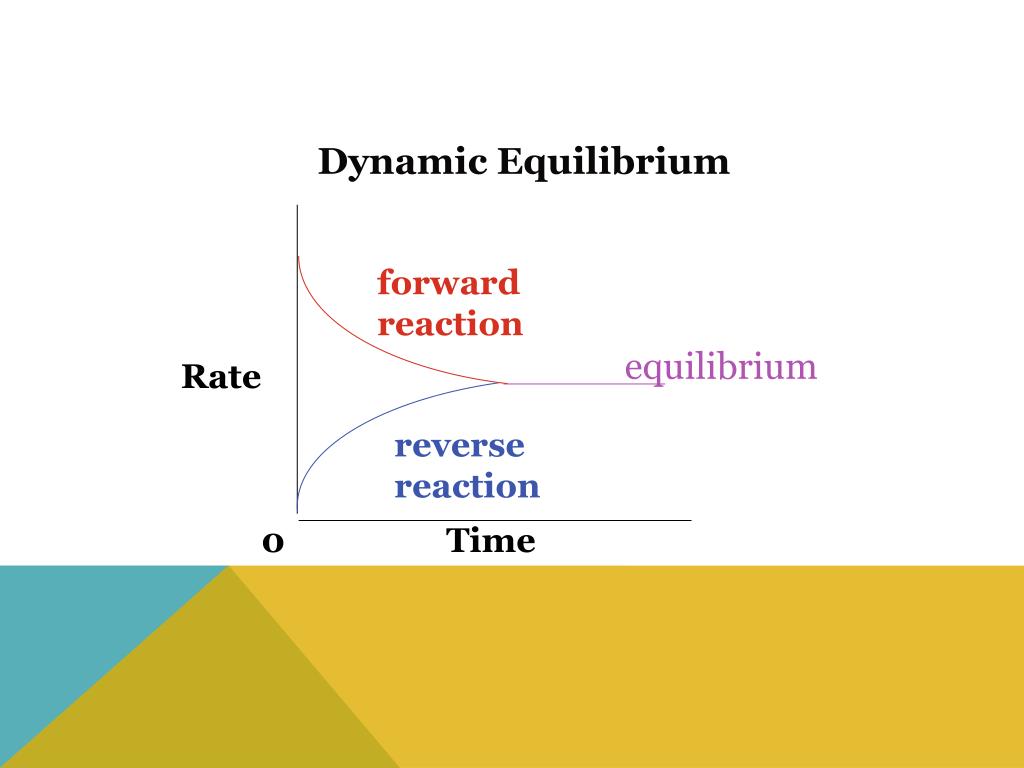
So, when we say a reaction is in dynamic equilibrium, it means that the forward reaction (reactants turning into products) is happening at the exact same speed as the reverse reaction (products turning back into reactants). It's like two equally strong tug-of-war teams pulling on a rope. The rope isn't moving left or right, but you can bet your bottom dollar that both teams are giving it their all!
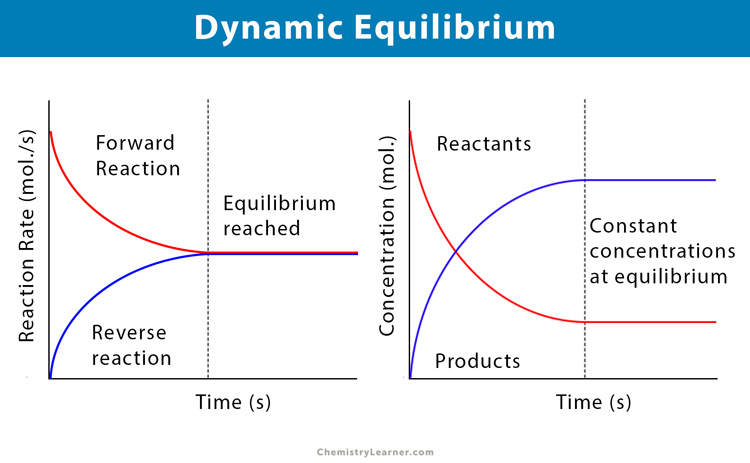
Here's where it gets super cool. Even though the amounts of reactants and products stay constant, the action is still going on! Molecules are still bumping into each other, bonds are still breaking and forming. It’s like a busy highway at rush hour. Cars are zooming everywhere, but the traffic jam (the number of cars on the road) stays relatively the same. It’s a hive of activity, just perfectly synchronized.
Let's try another fun example. Imagine you have a really popular YouTube channel. You’re posting awesome videos (that’s your reactants becoming products – viewers watching your videos). But, simultaneously, some people are unsubscribing (that's your products becoming reactants – viewers deciding they’ve seen enough). If your channel is in dynamic equilibrium, it means the number of new subscribers you’re getting each day is exactly the same as the number of people unsubscribing each day. So, your total subscriber count stays steady, even though there's a constant churn of people coming and going. It's a testament to your amazing content, of course!
What if someone tells you, "Oh, equilibrium means everything just stops!"? You can playfully chuckle and say, "Not so fast, my friend! That's a static equilibrium, which is like a sleeping cat. Wonderful in its own way, but dynamic equilibrium is the cat that's chased a laser pointer all day and is still full of zoomies, just perfectly balanced!"
So, to sum it up in a way that will make you feel like a chemistry rockstar: a reaction in dynamic equilibrium is when the rate of the forward reaction is perfectly matched by the rate of the reverse reaction. It looks like nothing's changing on the outside, but on the inside, it's a full-on, perfectly balanced chemical rave! It's the ultimate chemical harmony, where everything is in motion but perfectly still. Isn't that just the coolest thing ever? Keep that enthusiasm going, because understanding this is a major win!
The statement that correctly describes a reaction in dynamic equilibrium is one where the forward reaction rate equals the reverse reaction rate. This means that even though the reaction is still happening in both directions, the overall amounts of reactants and products remain constant. It's the chemical equivalent of a perfectly balanced seesaw, with equal effort on both sides!
So next time you see something that appears unchanging, remember the hidden dance of dynamic equilibrium. It’s a beautiful, energetic, and utterly fascinating aspect of the universe. Embrace the balance, embrace the buzz!
