Which Statement Best Describes The Ph Of Pure Water

Hey there, fellow science curious cats! Today we're diving into something that sounds a little… well, chemically, but I promise it's way more interesting than it sounds. We're talking about the pH of pure water. Yep, that stuff you guzzle by the gallon, or maybe just use to brew your morning coffee (no judgment here!). It’s got a secret score, and understanding it is actually pretty neat. Think of it like water’s personality quiz, and we’re about to find out what its best descriptor is.
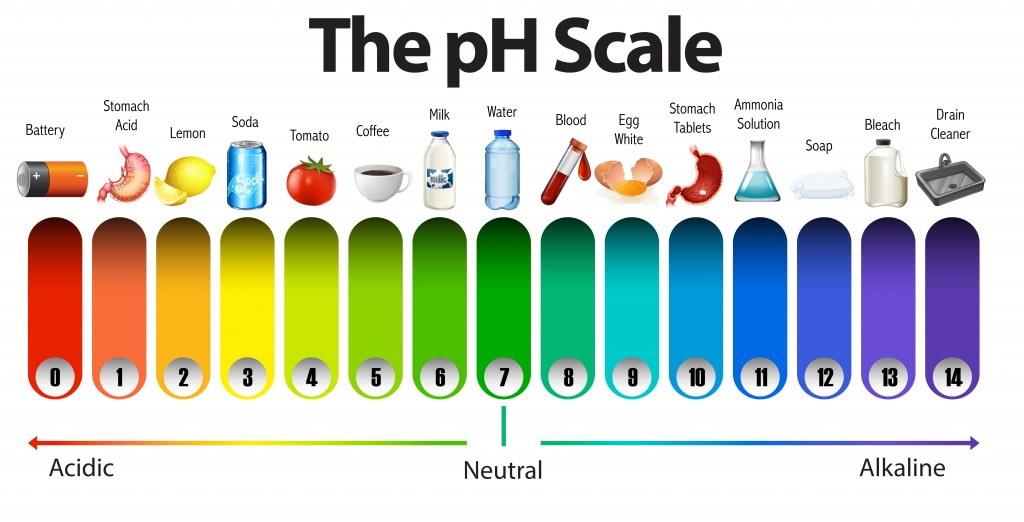
So, what's this whole "pH" thing, anyway? Don't let the fancy letters intimidate you! pH is basically a scale that tells us if something is acidic, neutral, or alkaline (sometimes people say "basic," but let's stick with alkaline for a bit of fun flair, shall we?). Imagine a seesaw, but instead of kids, it's about hydrogen ions. The more hydrogen ions bouncing around, the more acidic something is. Less hydrogen ions? More alkaline. And right in the middle, where it's perfectly balanced, is our buddy, neutral.
The pH scale runs from 0 to 14. If something is super acidic, like lemon juice or battery acid (don't taste that, please!), it’s going to be down on the lower end, closer to 0. If it's super alkaline, like drain cleaner (again, resist the urge to experiment!), it's going to be up on the higher end, closer to 14. Think of it as a spectrum of zinginess, from ouch, sour to whoa, slippery.
Now, where does our humble, everyday pure water fit into this dramatic spectrum? This is where things get really interesting, and honestly, a little bit magical. Pure water, the stuff that’s been stripped of all its extra minerals and goodies (and yes, even the little bits of dissolved air that make tap water taste like… well, tap water), is the ultimate definition of balance. It’s like the zen master of the liquid world.
So, when we ask, "Which statement best describes the pH of pure water?" we're looking for that perfect middle ground. We’re looking for the Goldilocks of liquids – not too sour, not too slippery, but just right. And what is that "just right" number on the pH scale? Drumroll, please… it’s a perfect 7.
Yes, a pH of 7 is the sweet spot for pure water. This means it's neither acidic nor alkaline. It's completely, utterly, and beautifully neutral. It’s like the Swiss Army knife of substances, versatile and essential, without leaning to one extreme or the other. It’s the friend who doesn't pick sides in an argument, just calmly observes. And honestly, who doesn't love that kind of stable energy?
Let’s break this down a little more, just to make sure it sinks in. Think about what makes something acidic. It’s all about those free hydrogen ions (H+). In acidic solutions, there are lots of them. They're practically throwing a party and getting everyone riled up. This is what gives acids their characteristic sour taste and their ability to react with other things.
On the flip side, in alkaline solutions, there are fewer hydrogen ions and more of something called hydroxide ions (OH-). These guys are the more laid-back members of the molecular party. They tend to make things feel a bit… well, slippery. Think of soap – that's alkaline! It’s great for cleaning, but you definitely wouldn't want to drink it. It’s the opposite end of the spectrum from a sour lemon drop.
But pure water? It’s like the peacemaker. In pure water, the number of hydrogen ions and hydroxide ions is exactly the same. They're in perfect equilibrium, holding hands and dancing a beautiful, balanced waltz. This perfect balance is what defines its neutrality. It’s the epitome of "just chilling."

Now, a little disclaimer, because science is rarely that simple, right? In the real world, the water you drink from your tap, or the water in a stream, or even the water in your fancy-pants Brita filter, is rarely 100% pure. Tap water, for example, often has minerals dissolved in it, like calcium and magnesium. These minerals can actually nudge the pH up a little, making it slightly alkaline. That’s usually a good thing, as it can make the water taste a bit nicer and potentially even have some health benefits.
And if you're a keen gardener, you might have noticed that different plants prefer different soil pH levels. That's because the soil's pH affects how easily plants can absorb nutrients. It’s all connected, isn't it? Even the ground beneath our feet is playing a pH game!
But when we talk about pure water, the theoretical, pristine, absolutely nothing-else-in-it kind of water, we're talking about that ideal pH of 7. It’s the benchmark. It’s the starting point. It’s the blank canvas before any other ingredients are added.

So, if you were given a multiple-choice question (which, let's be honest, is way more fun than a pop quiz in real life), and one of the options was "a pH of 7, indicating a neutral substance," that would be your winning ticket! Another option might be "acidic," which would be wrong because water isn't making your tongue pucker. Another might be "alkaline," which is also wrong because it's not going to feel like a bar of soap. And then there might be some wacky options like "depends on the weather" or "tastes like blueberries." Those are definitely wrong, and frankly, a little silly!
The beauty of pure water's neutrality is its incredible versatility. Because it's not pushing the scale in either direction, it's able to dissolve a huge variety of substances. It's the ultimate solvent. It's the universal cleaner, the base for so many reactions, and the very essence of life. Without water's neutral pH, life as we know it simply wouldn't exist. It would be like trying to build a house with all the wrong tools – a recipe for disaster!
Think about it: your blood has a very specific, slightly alkaline pH that needs to be maintained for you to stay alive and kicking. Your stomach, on the other hand, is super acidic (pH 1.5-3.5!) to help digest your food. It's amazing how different environments in our bodies have drastically different pH levels, and how water plays a role in all of them.

Even in chemistry experiments, pure water is often used as a baseline to measure the acidity or alkalinity of other solutions. It's the control group, the standard against which everything else is compared. It’s the ultimate reference point, the one we always go back to. It’s the reliable friend who’s always there to lend an objective ear.
So, let’s recap our little pH adventure. We learned that pH is a scale measuring acidity and alkalinity. We learned that 0 is super acidic, 14 is super alkaline, and 7 is right in the middle – neutral. And the star of our show, pure water, is the perfect example of neutrality with its pH of 7. It’s balanced, it’s stable, and it’s essential for everything.
Isn’t it amazing how something as simple and everyday as water can have such a fundamental property that impacts so much? It’s like finding out your favorite comfy t-shirt is actually a superhero in disguise. The quiet, unassuming hero of the chemical world!
So, the next time you take a sip of water, or even just see a puddle after the rain, take a moment to appreciate its perfectly balanced pH. It’s a little reminder that sometimes, the most powerful things are the ones that are perfectly in sync. It’s a testament to the beauty of balance in our world, and a gentle nudge to find our own sense of equilibrium. Go forth and embrace the neutral, my friends, because sometimes, it’s the most brilliant place to be!
